Abstract
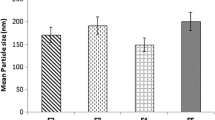
In this study, poly ethylene glycol 100 stearate (PEG 100-S) was used to prepare coated solid lipid nanoparticles with loading levothyroxine sodium (levo-loaded PEG 100-S-coated SLNs) by microemulsification technique. Evaluation of the release kinetic of prepared colloidal carriers was conducted. The particle size and zeta potential of levo-loaded PEG 100-S-coated SLNs have been measured to be 187.5 nm and −23.0 mV, respectively, using photon correlation spectroscopy (PCS). Drug entrapment efficiency (EE) was calculated to be 99 %. Differential scanning calorimetry indicated that the majority of drug loaded in PEG 100-S-coated SLNs were in amorphous state which could be considered desirable for drug delivery. The purpose of this study was to develop a new nanoparticle system, consisting lipid nanoparticles coated with PEG 100-S. The modification procedure led to a reduction in the zeta potential values, varying from −40.0 to −23.0 mV for the uncoated and PEG-coated SLNs, respectively. Stability results of the nanoparticles in gastric and intestinal media show that the low pH of the gastric medium is responsible for the critical aggregation and degradation of the uncoated lipid nanoparticles. PEG 100-S-coated SLNs were more stable due to their polymer coating layer which prevented aggregation of SLNs. Consequently, it is possible that the PEG surrounds the particles reducing the attachment of enzymes and further degradation of the triglyceride cores. Shape and surface morphology of particles were determined by transition electron microscopy and scanning electron microscopy that revealed spherical shape of nanoparticles. In vitro drug release of PEG 100-S-coated SLNs was characterized using diffusion cell which showed a controlled release for drug.





Similar content being viewed by others
References
Abdelbary G, Fahmy RH (2009) Diazepam-loaded solid lipid nanoparticles: design and characterization. AAPS Pharm SciTech 10(1):211–219
Barzegar-Jalali M, Adibkia K, Valizadeh H, Siahi-Shadbad MR, Nokhodchi A, Omidi Y, Mohammadi G, Hallaj-Nezhad S, Hasan M (2008) Kinetic analysis of drug release from nanoparticles. J Pharm Pharm Sci 11(1):167–177
Bhaskar K, Anbu J, Ravichandiran V, Venkateswarlu V, Rao YM (2009) Lipid nanoparticles for transdermal delivery of flurbiprofen: formulation, in vitro, ex vivo and in vivo studies. Lipids Health Dis 8(6):1–15
Blakesley VA (2005) Current methodology to assess bioequivalence of levothyroxine sodium products is inadequate. AAPS J 7(1):122–128
Bocca C, Caputo O, Cavalli R, Gabriel L, Miglietta A, Gasco MR (1998) Phagocytic uptake of fluorescent stealth and non-stealth solid lipid nanoparticles. Int J Pharm 175(2):185–193
Cavalli R, Gasco MR, Chetoni P, Burgalassi S, Saettone MF (2002) Solid lipid nanoparticles (SLN) as ocular delivery system for tobramycin. Int J Pharm 238(1–2):241–245
Cavalli R, Bargoni A, Podio V, Muntoni E, Zara GP, Gasco MR (2003) Duodenal administration of solid lipid nanoparticles loaded with different percentages of tobramycin. J Pharm Sci 92(5):1085–1094
Chimmiri P, Rajalakshmi R, Mahitha B, Ramesh G, Noor Ahmed VH (2012) Solid lipid nanoparticles: a novel carrier for cancer therapy. Int J Biol Pharm Res 3(3):405–413
Collier JW, Shah RB, Bryant AR, Habib MJ, Khan MA, Faustino PJ (2011) Development and application of a validated HPLC method for the analysis of dissolution samples of levothyroxine sodium drug products. J Pharm Biomed Anal 54(3):433–438
Dadashzadeh S, Derakhshandeh K, Shirazi FH (2008) 9-Nitrocamptothecin polymeric nanoparticles: cytotoxicity and pharmacokinetic studies of lactone and total forms of drug in rats. Anticancer Drugs 19(8):805–811
Damge C, Michel C, Aprahamian M, Couvreur P, Devissaguet JP (1990) Nanocapsules as carriers for oral peptide delivery. J Control Release 13(2–3):233–239
Derakhshandeh K, Erfan M, Dadashzadeh S (2007) Encapsulation of 9-nitrocamptothecin, a novel anticancer drug, in biodegradable nanoparticles: factorial design, characterization and release kinetics. Eur J Pharm Biopharm 66(1):34–41
Derakhshandeh K, Soheili M, Dadashzadeh S, Saghiri R (2010) Preparation and in vitro characterization of 9-nitrocamptothecin loaded long circulating nanoparticles for delivery of to cancer. Int J Nanomedicine 5(1):1–9
Desai MP, Labhasetwar V, Amidon GL, Levy RL (1996) Gastrointestinal uptake of biodegradable microparticles: effect of particle size. Pharm Res 13(12):1838–1845
Ekambaram P, Sathali AAH, Priyanka K (2012) Solid lipid nanoparticles: a review. Sci Rev Chem Commun 2(1):80–102
Fang YP, Wu PC, Huang YB, Tzeng CC, Chen YL, Hung YH, Tsai MJ, Tsai YH (2012) Modification of polyethylene glycol onto solid lipid nanoparticles encapsulating a novel chemotherapeutic agent (PK-L4) to enhance solubility for injection delivery. Int J Nanomedicine 7:4995–5005
Florence AT (1997) The oral absorption of micro- and nanoparticulates: neither exceptional nor unusual. Pharm Res 14(3):259–266
Garcia-Fuentes M, Torres D, Alonso MJ (2002) Design of lipid nanoparticles for the oral delivery of hydrophilic macromolecules. Colloids Surf B 27(2–3):159–168
Garcia-Fuentes M, Torres D, Alonso MJ (2005a) Design and characterization of a new drug nanocarrier made from solid–liquid lipid mixtures. J Colloid Interf Sci 285(2):590–598
Garcia-Fuentes M, Torres D, Alonso MJ (2005b) New surface-modified lipid nanoparticles as delivery vehicles for salmon calcitonin. Int J Pharm 296(1–2):122–132
Garcia-Fuentes M, Prego C, Torres D, Alonso MJ (2005c) A comparative study of the potential of solid glyceride nanostructures coated with chitosan or poly(ethylene glycol) as carriers for oral calcitonin delivery. Eur J Pharm Sci 25(1):133–143
Gref R, Luck M, Quellec P, Marchand M, Dellacherie E, Harnisch S, Blunk T, Muller RH (2000) ‘Stealth’ corona-core nanoparticles surface modified by polyethylene glycol (PEG): influences of the corona (PEG chain length and surface density) and of the core composition on phagocytic uptake and plasma protein adsorption. Colloids Surf B 18(3–4):301–313
Gregorini A, Ruiz ME, Volonté MG (2013) A derivative UV spectrophotometric method for the determination of levothyroxine sodium in tablets. J Anal Chem 68(6):510–515
Hans ML, Lowman AM (2002) Biodegradable nanoparticles for drug delivery and targeting. Curr Opin Solid State Mater Sci 6(4):319–327
Heurtault B, Saulnier P, Pech B, Proust JE, Benoit JP (2003) Physico-chemical stability of colloidal lipid particles. Biomaterials 24(23):283–300
Hu FQ, Jiang SP, Du YZ, Yuan H, Ye YQ, Zeng S (2005) Preparation and characterization of stearic acid nanostructured lipid carriers by solvent diffusion method in an aqueous system. Colloids Surf B 45(3–4):167–173
Jani PU, Halbert GW, Langridge J, Florence AT (1990) Nanoparticle uptake by the rat gastrointestinal mucosa: quantitation and particle size dependency. J Pharm Pharmacol 42(12):821–826
Kashanian S, Azandaryani HA, Derakhshandeh K (2011) New surface modified solid lipid nanoparticles by using N-glutaryl phosphatidyl ethanolamine as outer shell. Int J Nanomedicine 6:1–9
Khalil R, Kassem M, Elbary AA, ElRidi M, AbouSamra M (2013) Preparation and characterization of nystatin-loaded solid lipid nanoparticles for topical delivery. Int J Pharm Sci Res 4(6):2292–2300
Khan S (2012) Solid lipid nanoparticles: a review. World J Pharm Pharm Sci 1(1):96–115
Lai F, Sinico C, De LA, Zaru M, Müller RH, Fadda AM (2007) SLN as a topical delivery system for Artemisia arborescens essential oil: in vitro antiviral activity and skin permeation study. Int J Nanomedicine 2(3):419–425
Li XW, Lin XH, Zheng L, Yu L, Lv FF, Zhang Q, Liu W (2008) Effect of poly (ethylene glycol) stearate on the phase behavior of monocaprate/tween80/water system and characterization of poly (ethylene glycol) stearate-modified solid lipid nanoparticles. Colloids Surf A 317(1–3):352–359
Lilja JJ, Laitinen K, Neuvonen PJ (2005) Effects of grapefruit juice on the absorption of levothyroxine. Brit J Clin Pharmacol 60(3):337–341
Liu J, Gong T, Wang C, Zhong Z, Zhang Z (2007) Solid lipid nanoparticles loaded with insulin by sodium cholate-phosphatidylcholine-based mixed micelles: preparation and characterization. Int J Pharm 340(1–2):153–162
Liu W, Hu M, Liu W, Xue C, Xu H, Yang XL (2008) Investigation of the carbopol gel of solid lipid nanoparticles for the transdermal iontophoretic delivery of triamcinolone acetate. Int J Pharm 364(1):135–141
Lowe PL, Temple CS (1994) Calcitonin and insulin in isobutyl cyanoacrylate nanocapsules: protection against proteases and effect on intestinal absorption. J Pharm Pharmacol 46(7):547–552
Muller RH, Mader K, Gohla S (2000) Solid lipid nanoparticles (SLN) for controlled drug delivery—are view of the state of the art. Eur J Pharm Biopharm 50(1):161–177
Parhi R, Suresh P (2010) Production of solid lipid nanoparticles—drug loading and release mechanism. J Chem Pharm Res 2(1):211–227
Prego C, Torres D, Fernandez-Megia E, Novoa-Carballal R, Quiñoá E, Alonso MJ (2006) Chitosan–PEG nanocapsules as new carriers for oral peptide delivery. Effect of chitosan pegylation degree. J Controlled Release 111(3):299–308
Rapaka RS, Roth J, Brine GA, Prasad VK (1982) A simple HPLC method for the dissolution studies on levothyroxine sodium tablets. Int J Pharm 12(4):285–294
Sarmento B, Martins S, Ferreira D, Souto EB (2007) Oral insulin delivery by means of solid lipid nanoparticles. Int J Nanomedicine 2(4):743–749
Shah RB, Bryant A, Collier J, Habib MJ, Khan MA (2008) Stability indicating validated HPLC method for quantification of levothyroxine with eight degradation peaks in the presence of excipients. Int J Pharm 360(1–2):77–82
Singhvi G, Singh M (2011) Review: in vitro drug release characterization models. Int J Pharm Stud Res 2(1):77–84
Souto EB, Wissing SA, Barbosa CM, Müller RH (2004) Development of a controlled release formulation based on SLN and NLC for topical clotrimazole delivery. Int J Pharm 278(1):71–77
Subedi RK, Kang KW, Choi HK (2009) Preparation and characterization of solid lipid nanoparticles loaded with doxorubicin. Eur J Pharm Sci 37(3–4):508–513
Svensson J, Ericsson UB, Nilsson P, Olsson C, Jonsson B, Lindberg B, Ivarsson S (2006) Levothyroxine treatment reduces thyroid size in children and adolescents with chronic autoimmune thyroiditis. J Clin Endocrinol Metab 91(5):1729–1734
Uner M, Yener G (2007) Importance of solid lipid nanoparticles (SLN) in various administration routes and future perspectives. Int J Nanomedicine 2(3):289–300
Vila A, Sanchez A, Tobıo M, Calvo P, Alonso MJ (2002) Design of biodegradable particles for protein delivery. J Controlled Release 78(1–3):15–24
Vila A, Sanchez A, Evora C, Soriano I, Vila Jato JL, Alonso MJ (2004) PEG-PLA nanoparticles as carriers for nasal vaccine delivery. J Aerosol Med 17(2):174–185
Volpato NM, Silva RL, Brito APP, Gonçalves JCS, Vaisman M, Noël F (2004) Multiple level C in vitro/in vivo correlation of dissolution profiles of two l-thyroxine tablets with pharmacokinetics data obtained from patients treated for hypothyroidism. Eur J Pharm Sci 21(5):655–660
Yang SC, Lu LF, Cai Y, Zhu JB, Liang BW, Yang CZ (1999) Body distribution in mice of intravenously injected camptothecin solid lipid nanoparticles and targeting effect on the brain. J Control Release 59(3):299–307
Zara GP, Bargoni A, Cavalli R, Fundaro A, Vighetto D, Gasco MR (2002) Pharmaco- kinetics and tissue distribution of idarubicin-loaded solid lipid nanoparticles after duodenal administration to rats. J Pharm Sci 91(5):1324–1333
Zhang Q, Yie G, Li Y, Yang Q, Nagai T (2000) Studies on the cyclosporine A loaded stearic acid nanoparticles. Int J Pharm 200(2):153–159
Zhang N, Ping QN, Huang GH, Xua WF (2005) Investigation of lectin-modified insulin liposomes as carriers for oral administration. Int J Pharm 294(1–2):247–259
Zhang C, Gu C, Peng F, Liu W, Wan J, Xu H, Lam CW, Yang X (2013) Preparation and optimization of triptolide-loaded solid lipid nanoparticles for oral delivery with reduced gastric irritation. Molecules 18(11):13340–13356
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kashanian, S., Rostami, E. PEG-stearate coated solid lipid nanoparticles as levothyroxine carriers for oral administration. J Nanopart Res 16, 2293 (2014). https://doi.org/10.1007/s11051-014-2293-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-014-2293-6