Abstract
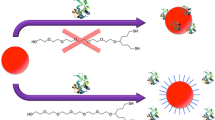
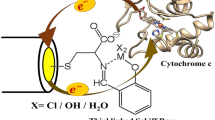
Controlled electrostatic physisorption of horse heart cytochrome c (Cyt c) onto nanostructured gold surfaces was investigated using Quartz-Crystal Microbalance measurements in planar gold surfaces with or without functionalization using a self-assembled monolayer (SAM) of the alkanethiol mercaptoundecanoic acid (MUA). MUA is a useful functionalization ligand for gold surfaces, shedding adsorbed biomolecules from the excessive electron density of the metal. A parallel analysis was conducted in the corresponding curved surfaces of 15 nm gold nanoparticles (AuNPs), using zeta-potential and UV– visible spectroscopy. Atomic Force Microscopy of both types of functionalized gold surfaces with a MUA SAM, allowed for visualization of Cyt c deposits on the nanostructured gold surface. The amount of Cyt c adsorbed onto the gold surface could be controlled by the solution pH. For the assays conducted at pH 4.5, when MUA SAM- functionalized planar gold surfaces are positive or neutral, and Cyt c has a positive net charge, only 13 % of the planar gold surface area was coated with protein. In contrast, at pH 7.4, when MUA SAM-functionalized planar gold surfaces and Cyt c have opposite charges, a protein coverage of 28 % could be observed implying an adsorption process strongly governed by electrostatic forces. Cyt c adsorption on planar and curved gold surfaces are found to be greatly favored by the presence of a MUA-capping layer. In particular, on the AuNPs, the binding constant is three times larger than the binding constant obtained for the original citrate-capped AuNPs.







Similar content being viewed by others
References
Aubin-Tam ME, Hamad-Schifferli K (2005) Gold nanoparticle cytochrome c complexes: the effect of nanoparticle ligand charge on protein structure. Langmuir 21(26):12080–12084
Bain CD, Whitesides GM (1988) Molecular-level control over surface order in self-assembled monolayer films of thiols on gold. Science 240(4848):62–63
Banci L, Bertini I, Rosato A, Varani G (1999) Mitochondrial cytochromes c: a comparative analysis. J Biol Inorg Chem 4(6):824–837
Baptista P, Doria G, Henriques D, Pereira E, Franco R (2005) Colorimetric detection of eukaryotic gene expression with DNA-derivatized gold nanoparticles. J Biotechnol 119(2):111–117
Baptista P, Pereira E, Eaton P, Doria G, Miranda A, Gomes I, Quaresma P, Franco R (2008) Gold nanoparticles for the development of clinical diagnosis methods. Anal Bioanal Chem 391:943–950
Brewer SH, Glomm WR, Johnson MC, Knag MK, Franzen S (2005) Probing BSA binding to citrate-coated gold nanoparticles and surfaces. Langmuir 21(20):9303–9307
Bunz UHF, Rotello VM (2010) Gold nanoparticle-fluorophore complexes: sensitive and discerning “noses” for biosystems sensing. Angewandte Chemie-Int Ed 49(19):3268–3279
Carmody WR (1961) Easily prepared wide range buffer series. J Chem Educ 38(11):559. doi:10.1021/ed038p559
Chah S, Hammond MR, Zare RN (2005) Gold nanoparticles as a colorimetric sensor for protein conformational changes. Chem Biol 12(3):323–328
de la Fuente JM, Penades S (2006) Glyconanoparticles: types, synthesis and applications in glycoscience, biomedicine and material science. Biochim Biophys Acta 1760(4):636–651
De M, Rana S, Akpinar H, Miranda OR, Arvizo RR, Bunz UHF, Rotello VM (2009) Sensing of proteins in human serum using conjugates of nanoparticles and green fluorescent protein. Nat Chem 1(6):461–465
Domingues MM, Santiago PS, Castanho MA, Santos NC (2008) What can light scattering spectroscopy do for membrane-active peptide studies? J Pept Sci 14(4):394–400. doi:10.1002/psc.1007
Doria G, Baumgartner BG, Franco R, Baptista PV (2010) Optimizing Au-nanoprobes for specific sequence discrimination. Colloid Surf B 77(1):122–124
Fears KP, Creager SE, Latour RA (2008) Determination of the surface pK of carboxylic- and amine-terminated alkanethiols using surface plasmon resonance spectroscopy. Langmuir 24(3):837–843
Gomes I, Santos NC, Oliveira LMA, Quintas A, Eaton P, Pereira E, Franco R (2008) Probing surface properties of cytochrome c at Au bionanoconjugates. J Phys Chem C 112(42):16340–16347
Haiss W, Thanh NT, Aveyard J, Fernig DG (2007) Determination of size and concentration of gold nanoparticles from UV-vis spectra. Anal Chem 79(11):4215–4221. doi:10.1021/ac0702084
Haynes CA, Norde W (1994) Globular proteins at solid/liquid interfaces. Colloids Surf B 2(6):517–566. doi:10.1016/0927-7765(94)80066-9
Heering HA, Wiertz FGM, Dekker C, de Vries S (2004) Direct immobilization of native yeast Iso-1 Cytochrome c on bare gold: fast electron relay to redox enzymes and zeptomole protein-film voltammetry. J Am Chem Soc 126(35):11103–11112. doi:10.1021/ja046737w
Hulko M, Hospach I, Krasteva N, Nelles G (2011) Cytochrome C biosensor—a model for gas sensing. Sensors 11(6):5968–5980
Imabayashi S, Mita T, Kakiuchi T (2005) Effect of the electrostatic interaction on the redox reaction of positively charged cytochrome C adsorbed on the negatively charged surfaces of acid-terminated alkanethiol monolayers on a Au(111) electrode. Langmuir 21(4):1470–1474
Ipe BI, Shukla A, Lu HC, Zou B, Rehage H, Niemeyer CM (2006) Dynamic light-scattering analysis of the electrostatic interaction of hexahistidine-tagged cytochrome P450 enzyme with semiconductor quantum dots. ChemPhysChem 7(5):1112–1118
Jiang X, Jiang UG, Jin YD, Wang EK, Dong SJ (2005) Effect of colloidal gold size on the conformational changes of adsorbed cytochrome c: probing by circular dichroism, UV-visible, and infrared spectroscopy. Biomacromolecules 6(1):46–53
Kaufman ED, Belyea J, Johnson MC, Nicholson ZM, Ricks JL, Shah PK, Bayless M, Pettersson T, Feldoto Z, Blomberg E, Claesson P, Franzen S (2007) Probing protein adsorption onto mercaptoundecanoic acid stabilized gold nanoparticles and surfaces by quartz crystal microbalance and zeta-potential measurements. Langmuir 23(11):6053–6062
Keating CD, Kovaleski KM, Natan MJ (1998) Protein : colloid conjugates for surface enhanced Raman scattering: stability and control of protein orientation. J Phys Chem B 102(47):9404–9413
Kluck RM, BossyWetzel E, Green DR, Newmeyer DD (1997a) The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275(5303):1132–1136
Kluck RM, Martin SJ, Hoffman BM, Zhou JS, Green DR, Newmeyer DD (1997b) Cytochrome c activation of CPP32-like proteolysis plays a critical role in a Xenopus cell-free apoptosis system. EMBO J 16(15):4639–4649
Lin S, Jiang X, Wang L, Li G, Guo L (2011) Adsorption orientation of horse heart cytochrome c on a bare gold electrode hampers its electron transfer. J Phys Chem C 116(1):637–642. doi:10.1021/jp2063782
Lindman S, Lynch I, Thulin E, Nilsson H, Dawson KA, Linse S (2007) Systematic investigation of the thermodynamics of HSA adsorption to N-iso-propylacrylamide/N-tert-butylacrylamide copolymer nanoparticles. Effects of particle size and hydrophobicity. Nano Lett 7(4):914–920
Louie GV, Brayer GD (1990) High-resolution refinement of yeast Iso-1-cytochrome-C and comparisons with other eukaryotic cytochromes-C. J Mol Biol 214(2):527–555
Lundqvist M, Stigler J, Elia G, Lynch I, Cedervall T, Dawson KA (2008) Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. P Natl Acad Sci USA 105(38):14265–14270
Mandal HS, Kraatz HB (2007) Effect of the surface curvature on the secondary structure of peptides adsorbed on nanoparticles. J Am Chem Soc 129(20):6356–6357
Millo D, Bonifacio A, Ranieri A, Borsari M, Gooijer C, van der Zwan G (2007) pH-Induced changes in adsorbed cytochrome c. voltammetric and surface-enhanced resonance Raman characterization performed simultaneously at chemically modified silver electrodes. Langmuir 23(19):9898–9904
Murgida DH, Hildebrandt P (2004) Electron-transfer processes of cytochrome c at interfaces. New insights by surface-enhanced resonance Raman spectroscopy. Acc Chem Res 37(11):854–861
Nakano K, Yoshitake T, Yamashita Y, Bowden EF (2007) Cytochrome c self-assembly on alkanethiol monolayer electrodes as characterized by AFM, IR, QCM, and direct electrochemistry. Langmuir 23(11):6270–6275
Phillips RL, Miranda OR, You CC, Rotello VM, Bunz UHF (2008) Rapid and efficient identification of bacteria using gold-nanoparticle - Poly(para-phenyleneethynylene) constructs. Angewandte Chemie-Int Ed 47(14):2590–2594
Rieder R, Bosshard HR (1980) Comparison of the binding-sites on cytochrome-C for cytochrome-C oxidase, cytochrome-Bc1, and cytochrome-C1 - differential acetylation of lysyl residues in free and complexed cytochrome-C. J Biol Chem 255(10):4732–4739
Scott RA, Mauk AG (1995) Cytochrome c: a multidisciplinary approach. University Science Books, Sausalito
Serro AP, Carapeto A, Paiva G, Farinha JPS, Colaço R, Saramago B (2011) Formation of an intact liposome layer adsorbed on oxidized gold confirmed by three complementary techniques: QCM-D, AFM and confocal fluorescence microscopy. Surface and Interface Analysis:n/a-n/a. doi:10.1002/sia.3820
Shang L, Wang YZ, Jiang JG, Dong SJ (2007) pH-dependent protein conformational changes in albumin : gold nanoparticle bioconjugates: a spectroscopic study. Langmuir 23(5):2714–2721
Sperling RA, Rivera gil P, Zhang F, Zanella M, Parak WJ (2008) Biological applications of gold nanoparticles. Chem Soc Rev 37(9):1896–1908
Srivastava S, Verma A, Frankamp BL, Rotello VM (2005) Controlled assembly of protein-nanoparticle composites through protein surface recognition. Adv Mater 17(5):617–621
Vargo ML, Gulka CP, Gerig JK, Manieri CM, Dattelbaum JD, Marks CB, Lawrence NT, Trawick ML, Leopold MC (2010) Distance dependence of electron transfer kinetics for azurin protein adsorbed to monolayer protected nanoparticle film assemblies. Langmuir 26(1):560–569
Vörös J (2004) The density and refractive index of adsorbing protein layers. Biophys J 87(1):553–561
Wang L, Waldeck DH (2008) Denaturation of cytochrome c and its peroxidase activity when immobilized on SAM films. J Phys Chem C 112(5):1351–1356
Wegerich F, Turano P, Allegrozzi M, Möhwald H, Lisdat F (2009) Cytochrome c mutants for superoxide biosensors. Anal Chem 81(8):2976–2984. doi:10.1021/ac802571h
Yin H, Zhou Y, Liu T, Cui L, Ai S, Qiu Y, Zhu L (2010) Amperometric nitrite biosensor based on a gold electrode modified with cytochrome c on Nafion and Cu-Mg-Al layered double hydroxides. Microchim Acta 171(3):385–392. doi:10.1007/s00604-010-0444-8
You CC, De M, Rotello VM (2005) Mono layer-protected nanoparticle-protein interactions. Curr Opin Chem Biol 9(6):639–646
Zhang D, Neumann O, Wang H, Yuwono VM, Barhoumi A, Perham M, Hartgerink JD, Wittung-Stafshede P, Halas NJ (2009) Gold nanoparticles can induce the formation of protein-based aggregates at physiological pH. Nano Lett 9(2):666–671
Acknowledgments
Financial support was provided by Fundação para a Ciência e a Tecnologia (FCT/MEC), Portugal, under Grants PEst-C/EQB/LA0006/2011 to MJF, PE, EP, and RF; and PTDC/CTM-NAN/112241/2009 to RF; and PEst-OE/QUI/UI0100/2011 to APS and BS. IG is a recipient of a FCT/MEC post-doctoral fellowship (SFRH/BPD/63850/2009). The authors thank the Centro de Materiais da Universidade do Porto (CEMUP), for allowing the use of the multimode AFM, and Prof. António Fernando Silva and CIQUP, Laboratório de Química Analítica, Faculdade de Ciências, Universidade do Porto for allowing the use of the PicoLE AFM.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gomes, I., Feio, M.J., Santos, N.C. et al. Controlled adsorption of cytochrome c to nanostructured gold surfaces. J Nanopart Res 14, 1321 (2012). https://doi.org/10.1007/s11051-012-1321-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-012-1321-7