Abstract
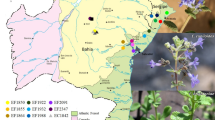
Duckweed, with rapid growth rate and high starch content, is a new alternate feedstock for bioethanol production. The genetic diversity among 27 duckweed populations of seven species in genus Lemna and Spirodela from China and Vietnam was analyzed by ISSR-PCR. Eight ISSR primers generating a reproducible amplification banding pattern had been screened. 89 polymorphic bands were scored out of the 92 banding patterns of 16 Lemna populations, accounting for 96.74% of the polymorphism. 98 polymorphic bands of 11 Spirodela populations were scored out of 99 banding patterns, and the polymorphism was 98.43%. The genetic distance of Lemna varied from 0.127 to 0.784, and from 0.138 to 0.902 for Spirodela, which indicated a high level of genetic variation among the populations studied. The unweighted pair group method with arithmetic average (UPGMA) cluster analysis corresponded well with the genetic distance. Populations from Sichuan China grouped together and so did the populations from Vietnam, which illuminated populations collected from the same region clustered into one group. Especially, the only one population from Tibet was included in subgroup A2 alone. Clustering analysis indicated that the geographic differentiation of collected sites correlated closely with the genetic differentiation of duckweeds. The results suggested that geographic differentiation had great influence on genetic diversity of duckweed in China and Vietnam at the regional scale. This study provided primary guidelines for collection, conservation, characterization of duckweed resources for bioethanol production etc.



Similar content being viewed by others
References
Crawford DJ, Landolt E, Les DH, Kimball RT (2006) Speciation in duckweeds (Lemnaceae): phylogenetic and ecological inferences. Aliso 22:231–242
Landolt E (1996) Duckweeds (Lemnaceae): morphological and ecological characteristics and their potential for recycling of nutrients. In: 2nd international conference on ecological engineering for wastewater treatment. Recycling the resource, 5–6, pp 289–296
Les DH, Landolt E, Crawford DJ (1997) Systematics of the Lemnaceae (duckweeds): inferences from micromolecular and morphological data. Plant Syst Evol 204:161–177. doi:10.1007/BF00989203
Wu ZY (2004) Flora Reipublicae Popularis Sinicae, vol 13. Science Press, Beijing, p. 207
Marín CM, Oron G (2007) Boron removal by the duckweed Lemna gibba: a potential method for the remediation of boron-polluted waters. Water Res 41:4579–4584. doi:10.1016/j.watres.2007.06.051
Myriam KB, Joaquim AF, Cristina N, Prasad MNV, Helena F (2009) Ecophysiological tolerance of duckweeds exposed to copper. Aquat Toxicol 91:1–9. doi:10.1016/j.aquatox.2008.09.009
Culley DDJ, Rejmankova E, Kvet J, Feye JB (1981) Production, chemical quality, and use of duckweed (Lemnaceae) in aquaculture, waster management, and animal feeds. J World Maric Soc 12:27–29. doi:10.1111/j.1749-7345.1981.tb00273.x
Krishna KCB, Polprasert C (2008) An integrated kinetic model for organic and nutrient removal by duckweed-based wastewater treatment (DUBWAT) system. Ecol Eng 34:243–250. doi:10.1016/j.ecoleng.2008.08.013
Oron G (1994) Duckweed culture for wastewater renovation and biomass production. Agric Water Manag 26:27–40. doi:10.1016/0378-3774(94)90022-1
Chen Y, Yablonski M, Ernst E, Stomp AM, Cheng JJ (2007) Duckweed: an alternative starch source for bioethanol production. In: ASABE regional annual conference, May 31–June 1
Cheng JJ, Stomp AM (2009) Growing duckweed to recover nutrients from wastewaters and for production of fuel ethanol and animal feed. Clean Soil Air Water 37:17–26. doi:10.1002/clen.200800210
Wang Y, Zhang Y, Yang B, Chen S (2010) Characterization of SSU5C promoter of a rbcS gene from duckweed (Lemna gibba). Mol Biol Rep (Published online: 16 Nov 2010). doi:10.1007/s11033-010-0395-5
Heller J (1996) Physic nut. Jatropha curcas L. Promoting the conservation and use of underutilized and neglected crop. International Plant Genetic Resources Institute, Rome
Han YC, Teng CZ, Chang FH (2007) Analyses of genetic relationships in Nelumbo nucifera using nuclear ribosomal ITS sequence data, ISSR and RAPD markers. Aquat Bot 87:141–146. doi:10.1016/j.aquabot.2007.04.005
Li WG, Wang BR, Wang JB (2006) Lack of genetic variation of an invasive clonal plant Eichhornia crassipes in China revealed by RAPD and ISSR markers. Aquat Bot 84:176–180. doi:10.1016/j.aquabot.2005.09.008
Triest L (2008) Molecular ecology and biogeography of mangrove trees towards conceptual insights on gene flow and barriers: a review. Aquat Bot 89:138–154. doi:10.1016/j.aquabot.2007.12.013
Zietkiewicz E, Rafalski A, Labuda D (1994) Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 20:176–183. doi:10.1006/geno.1994.1151
Nagaoka T, Ogihara Y (1997) Applicability of inter-simple sequence repeat polymorphisms in wheat for use as DNA markers in comparison to RFLP and RAPD markers. Theor Appl Genet 94:597–602. doi:10.1007/s001220050456
Tsumura Y, Ohba K, Strauss SH (1996) Diversity and inheritance of inter-simple sequence repeat polymorphisms in douglas-fir (Pseudotsuga menziesii) and sugi (Cryptomeria japonica). Theor Appl Genet 92:40–45. doi:10.1007/BF00222949
Godwin ID, Aitken EAB, Smith LW (1997) Application of inter simple sequence repeat (ISSR) markers to plant genetics. Electrophoresis 18:1524–1528. doi:10.1002/elps.1150180906
Chen YY, Zhou RC, Lin XD (2008) ISSR analysis of genetic diversity in sacred lotus cultivars. Aquat Bot 89:311–316. doi:10.1016/j.aquabot.2008.03.006
Grativol C, Lira CF, Hemerly AS, Ferreira PCG (2010) High efficiency and reliability of inter-simple sequence repeats (ISSR) markers for evaluation of genetic diversity in Brazilian cultivated Jatropha curcas L. accessions. Mol Biol Rep (published online: 27 Nov 2010). doi:10.1007/s11033-010-0547-7
Kumar RS, Parthiban KT, Rao MG (2009) Molecular characterization of Jatropha genetic resources through inter-simple sequence repeat (ISSR) markers. Mol Biol Rep 36:1951–1956. doi:10.1007/s11033-008-9404-3
Esselman EJ, Jianqiang L, Crawford DJ, Winduss JL, Wolfe AD (1999) Clonal diversity in the rare Calamagrostis porteri ssp. insperata (Poaceae): comparative results for allozymes and random amplified polymorphic DNA (RAPD) and inter simple sequence repeat (ISSR) markers. Mol Ecol 8:443–451. doi:10.1046/j.1365-294X.1999.00585.x
Fang DQ, Roose ML (1997) Identification of closely related citrus cultivars with inter-simple sequences repeat markers. Theor Appl Genet 95:408–417. doi:10.1007/s001220050577
Li JL, Wang GL, Bai ZY (2009) Genetic diversity of freshwater pearl mussel (Hyriopsis cumingii) in populations from the five largest lakes in China revealed by inter-simple sequence repeat (ISSR). Aquac Int 17:323–330. doi:10.1007/s10499-008-9204-8
Martirosyan EV, Ryzhova NN, Kochieva EZ, Skryabin KG (2009) Analysis of chloroplast rpS16 intron sequences in Lemnaceae. Mol Biol 43:32–38. doi:10.1134/S0026893309010051
Rahimmalek M, Bahreininejad B, Khorrami M, Tabatabaei BE (2009) Genetic variability and geographic differentiation in Thymus daenensis subsp. daenensis, an endangered medicinal plant, as revealed by inter simple sequence repeat (ISSR) markers. Biochem Genet 47:831–842. doi:10.1007/s10528-009-9281-z
Shahsavar AR, Izadpanah K, Tafazoli E, Tabatabaei BE (2007) Characterization of citrus germplasm including unknown variants by inter-simple sequence repeat (ISSR) markers. Sci Hortic 112:310–314. doi:10.1016/j.scienta.2006.12.039
Wang Xl, Zhao FJ, Hu ZM, Critchley AT, Morrell SL, Duan D (2008) Inter-simple sequence repeat (ISSR) analysis of genetic variation of Chondrus crispus populations from North Atlantic. Aquat Bot 88:154–159. doi:10.1016/j.aquabot.2007.10.001
Nei M (1972) Genetic distance between populations. Am Nat 106:282–283. doi:10.1086/282771
Bergmann BA, Cheng J, Classen J, Stomp AM (2000) In vitro selection of duckweed geographical isolates for potential use in swine lagoon effluent renovation. Bioresour Technol 73:13–20. doi:10.1016/S0960-8524(99)00137-6
Costa JC, Neto C, Arse′nio P, Capelo J (2009) Geographic variation among Iberian communities of the exotic halophyte Cotula coronopifolia. Bot Helv 119:53–61. doi:10.1007/s00035-009-0056-2
Dohzono I, Suzuki K (2010) Morphological and genetic differentiation in Isodon umbrosus by altitudinal variation in bumblebee pollinator assemblages. Plant Species Biol 25:20–29. doi:10.1111/j.1442-1984.2010.00268.x
Escudero M, Valcárce V, Vargas P, Luceño M (2009) Significance of ecological vicariance and long-distance dispersal in the diversification of Carex sect Spirostachyae (Cyperaceae). Am J Bot 96:2100–2114. doi:10.3732/ajb.0900134
Khalifa MB, Simon V, Marrakchi M, Fakhfakh H, Moury B (2009) Contribution of host plant resistance and geographic distance to the structure of Potato virus Y (PVY) populations in pepper in northern Tunisia. Plant Pathol 58:763–772. doi:10.1111/j.1365-3059.2009.02064.x
Cross JW (2006) Charms of Duckweed: an introduction to the smallest flowering plants. http://www.mobot.org/jwcross/duckweed/duckweed.htm. Accessed 17 Aug 2006
Acknowledgments
This research was supported by a grant from the National Key Technology R&D Program during the Twelfth Five-year Plan Period of China (2011BAD22B03).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xue, H., Xiao, Y., Jin, Y. et al. Genetic diversity and geographic differentiation analysis of duckweed using inter-simple sequence repeat markers. Mol Biol Rep 39, 547–554 (2012). https://doi.org/10.1007/s11033-011-0769-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-011-0769-3