Abstract
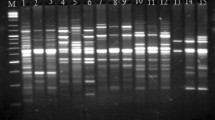
The two species of yellow catfish, Horabagrus brachysoma and H. nigricollaris are categorized as ‘endangered’ and ‘critically endangered’ respectively in their wild habitat. Proper knowledge of genetic structure and variability of these endangered species are highly essential for the management, conservation and improvement of fish stocks. Therefore, genetic variation and phylogenetic relationships between these species of yellow catfish sampled from Chalakkudy River in the hot spot of biodiversity-Western Ghats region, Kerala, India were analyzed by using Random amplified polymorphic DNA (RAPD) and microsatellite markers. 85 RAPD and five microsatellites loci were detected to analyze the genetic variation and phylogenetic relationships among these species. Out of 85 RAPD loci produced only 52.94% were polymorphic whereas in microsatellite, all 5 loci were polymorphic (100%). Species-specific RAPD bands were found in both species studied. In microsatellite, the number of alleles across the five loci ranged from 1 to 8. The observed heterozygosities in H. brachysoma and H. nigricollaris were 0.463 and 0.443, respectively. Here, both RAPD and microsatellite methods reported a low degree of gene diversity and lack of genetic heterogeneity in both species of Horabagrus which strongly emphasize the need of fishery management, conservation and rehabilitation of these species.



Similar content being viewed by others
References
Pethiyagoda R, Kottelat M (1994) Three new species of fishes of the genera Osteochilichthys, Travancoria and Horabagrus from the Chalakudy River, Kerala, India. J South Asian Nat Hist 1:97–116
Talwar PK, Jhingram AG (1991) Inland fishes of India and adjacent countries, vol 1. AA Balkema, Rotterdam
de Pinna MCC (1993) Higher-level Phylogeny of Siluriformes (Teleostei, Ostariophysi), with a new classification of the order. PhD Thesis. University of New York, New York
Hardman M (2005) The phylogenetic relationships among non-diplomystid catfishes as inferred from mitochondrial cytochrome b sequences; the search for the ictalurid sister taxon (Otophysi: Siluriformes). Mol Phylogenet Evol 37:700–720
Sullivan JP, Lundberg JG, Hardman M (2006) A phylogenetic analysis of the major groups of catfishes (Teleostei: Siluriformes) using rag1 and rag2 nuclear gene sequences. Mol Phylogenet Evol 41:636–662
Muneer PM (2006) Molecular genetic characterization of endemic yellow catfish, Horabagrus brachysoma (Gunther). PhD Thesis. Cochin University of Science and Technology, Cochin, p 225
Muneer PM, Gopalakrishnan A, Musammilu KK, Mohindra V, Lal KK, Basheer VS, Lakra WS (2009) Genetic variation and population structure of endemic yellow catfish, Horabagrus brachysoma (Bagridae) among three populations of Western Ghat region using RAPD and microsatellite markers. Mol Biol Rep 36:1779–1791
Ponniah AG, Gopalakrishnan A (2000) Endemic fish diversity of the Western Ghats. National Bureau of Fish Genetic Resources, NBFGR NATP Publication 1, Lucknow
Menon AGK (1999) Check list —fresh water fishes of India, Rec. Zool. Surv. India, Occas pap no 175, p 366
Muneer PM, Gopalakrishnan A, Lal KK, Mohindra V (2007) Population genetic structure of endemic and endangered yellow catfish, Horabagrus brachysoma, using allozyme markers. Biochem Genet 45:637–645
Prasad G, Anvar Ali PH, Raghavan R (2008) Threatened fishes of the world: Horabagrus nigricollaris (Pethiyagoda and Kottelat, 1994) (Bagridae). Environ Biol Fish 82:109–110
CAMP (1998) Conservation Assessment and Management Plan (CAMP) for freshwater fishes of India 1997, Zoo Outreach Organization (ZOO) and National Bureau of Fish Genetic Resources (NBFGR), Lucknow
Muneer PM, Gopalakrishnan A, Basheer VS, Lakra WS (2008) Identification of RAPD markers in endemic yellow catfish, Horabagrus brachysoma (Gunther, 1864). Asian Fish Sci 21:293–304
Shivanandan R (2004) Genetic profile of endangered and endemic yellow catfish. Horabagrus nigricollaris using RAPD and Microsatellite markers. MSc Dissertation. Periyar University, Tamil Nadu, India, p 69
Welsh J, McClelland M (1990) Fingerprinting genomes using PCR with arbitrary primers. Nucl Acids Res 18:7213–7218
Williams JG, Kubelik AR, Livak KJ, Rafalski JA, Tingey SV (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucl Acids Res 18:6531–6535
Clark AG, Lanigan CM (1993) Prospects for estimating nucleotide divergence with RAPDs. Mol Biol Evol 10:1096–1111
Bardakci F, Skibinski DO (1994) Application of the RAPD technique in tilapia fish: species and subspecies identification. Heredity 73:117–123
Borowsky R, Wilkens H (2002) Mapping a cave fish genome: polygenic systems and regressive evolution. J Hered 93:19–21
Callejas C, Ochando MD (2002) Phylogenetic relationships among Spanish Barbus species (Pisces, Cyprinidae) shown by RAPD markers. Heredity 89:36–43
Klinbunga S, Boonyapakdee A, Pratoomchat B (2000) Genetic diversity and Species-diagnostic markers of Mud Crabs (Genus Scylla) in Eastern Thailand Determined by RAPD Analysis. Mar Biotechnol (NY) 2:180–187
Khedkar GD, Reddy AC, Mann P, Ravinder K, Muzumdar K (2009) Clarias batrachus (Linn.1758) population is lacking genetic diversity in India. Mol Biol Rep 37:1355–1362
Shifat R, Begum A, Khan H (2003) Use of RAPD fingerprinting for discriminating two populations of Hilsa shad (Tenualosa ilisha Ham.) from inland rivers of Bangladesh. J Biochem Mol Biol 36:462–467
Saini A, Dua A, Mohindra V, Lakra WS (2010) Molecular discrimination of six species of Bagrid catfishes from Indus river system using randomly amplified polymorphic DNA markers. Mol Biol Rep. doi:10.1007/s11033-010-9960-1
Liu Z, Tan G, Li P, Dunham RA (1999) Transcribed dinucleotide microsatellites and their associated genes from channel catfish Ictalurus punctatus. Biochem Biophys Res Commun 259:190–194
Liu ZJ, Li P, Argue B, Dunham RA (1998) Inheritance of RAPD markers in channel catfish (Ictalurus punctatus), blue catfish (I. furcatus) and their F1, F2 and backcross hybrids. Anim Genet 29:58–62
Kovacs B, Egedi S, Bartfai R, Orban L (2000) Male-specific DNA markers from African catfish (Clarias gariepinus). Genetica 110:267–276
Tautz D (1989) Hypervariability of simple sequences as a general source for polymorphic DNA markers. Nucl Acids Res 17:6463–6471
Zardoya R, Vollmer DM, Craddock C, Streelman JT, Karl S, Meyer A (1996) Evolutionary conservation of microsatellite flanking regions and their use in resolving the phylogeny of cichlid fishes (Pisces: Perciformes). Proc Biol Sci 263:1589–1598
Kato M, Yoshida M (1995) Nucleotide sequence of a highly repetitive element isolated from Opsariichthys uncirostris (Osteichthyes). O uncirostris repetitive sequence. Mol Biol Rep 21:85–86
Knapik EW, Goodman A, Ekker M, Chevrette M, Delgado J, Neuhauss S, Shimoda N, Driever W, Fishman MC, Jacob HJ (1998) A microsatellite genetic linkage map for zebrafish (Danio rerio). Nat Genet 18:338–343
Elmesiry GE, Okai S, Hokabe S, Minoshima S, Sugiyama S, Yoshino T, Ohtani T, Shimizu N, Kato M (2005) Isolation and characterization of simple repeat sequences from the yellow fin sea bream Acanthopagrus latus (Sparidae). Mol Biol Rep 32:117–126
Primmer CR, Veselov AJ, Zubchenko A, Poututkin A, Bakhmet I, Koskinen MT (2006) Isolation by distance within a river system: genetic population structuring of Atlantic salmon, Salmo salar, in tributaries of the Varzuga River in northwest Russia. Mol Ecol 15:653–666
Hatanaka T, Henrique-Silva F, Galetti P M Jr (2006) Population substructuring in a migratory freshwater fish Prochilodus argenteus (Characiformes, Prochilodontidae) from the Sao Francisco River. Genetica 126:153–159
Galbusera P, Van S, Matthysen E (2000) Cross-species amplicifcation of microsatellite primers in passerine birds. Conserv Genet 1:163–168
Volckaert FAM, Helleman BAS, Pouyaud L (1999) Nine polymorphic microsatellite markers in the South East Asian Catfishes Pangasius hypophthalmus and Clarias batrachus. Anim Genet 30:383
Taggart JB, Hynes RA, Prodohl PA, Ferguson A (1992) A simplified protocol for routine total DNA isolation from salmonid fishes. J Fish Biol 40:963–965
Yeh FC Yang RC, Boyle T (1999) POPGENE 32 —version 1.31. Population genetics software
Na-Nakorn U, Taniguchi N, Nugroho E, Seki S, Kamonrat W (1999) Isolation and characterization of microsatellite loci of Clarias macrocephalus and their application to genetic diversity study. Fish Sci 65:520–526
Galbusera P, Volckaert FA, Hellemans B, Ollevier F (1996) Isolation and characterization of microsatellite markers in the African catfish Clarias gariepinus (Burchell, 1822). Mol Ecol 5:703–705
Van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) MICRO-CHECKER: software for identifying and correcting genotying errors in microsatellite data. Mol Ecol Notes 4:535–538
Nei M (1987) Molecular evolutionary genetics. Columbia University Press, New York
Nei M (1978) Estimation of Average Heterozygosity and Genetic Distance from a Small Number of Individuals. Genetics 89:583–590
Usmani S, Tan SG, Siraj SS, Yusoff K (2003) Population structure of the Southeast Asian river catfish Mystus nemurus. Anim Genet 34:462–464
Jackson TR, Ferguson MM, Danzmann RG, Fishback AG, Ihssen PE, O’Connel M, Crease TJ (1998) Identification of two QTL influencing upper temperature tolerance in three rainbow trout (Oncorhynchus mykiss) half-sib families. Heredity 80:143–151
Chistiakov DA, Hellemans B, Volckaert FAM (2006) Microsatellites and their genomic distribution, evolution, function and applications: A review with special reference to fish genetics. Aquaculture 255:1–29
Araneda C, Neira R, Iturra P (2005) Identification of a dominant SCAR marker associated with colour traits in Coho salmon (Oncorhynchus kisutch). Aquaculture 247:67–73
Si-Fa L, Shou-Jie T, Wan-Qi C (2010) RAPD-SCAR markers for genetically improved NEW GIFT Nile Tilapia (Oreochromis niloticus niloticus L.) and their application in strain identification. Zool Res 31:147–153
Kauffman EJ, Gestl EE, Kim DJ, Walker C, Hite JM, Yan G, Rogan PK, Johnson SL, Cheng KC (1995) Microsatellite-centromere mapping in the zebrafish (Danio rerio). Genomics 30:337–341
Lee W-J, Kocher TD (1996) Microsatellite DNA markers for edited genetic mapping in the tilapia, Oreochromis niloticus. J Fish Biol 49:169–171
Waldbieser GC, Bosworth BG, Nonneman DJ, Wolters WR (2001) A microsatellite-based genetic linkage map for channel catfish, Ictalurus punctatus. Genetics 158:727–734
Perry GM, Ferguson MM, Sakamoto T, Danzmann RG (2005) Sex-linked quantitative trait loci for thermotolerance and length in the rainbow trout. J Hered 96:97–107
Kocher TD, Lee WJ, Sobolewska H, Penman D, McAndrew B (1998) A genetic linkage map of a cichlid fish, the tilapia (Oreochromis niloticus). Genetics 148:1225–1232
Acknowledgments
Indian Council of Agricultural Research–National Agricultural Technology Project (ICAR–NATP), which supported this study financially, is gratefully acknowledged. The authors are grateful to Dr. S.P. Singh (PI, NATP, NBFGR) for encouragement, support and guidance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muneer, P.M.A., Gopalakrishnan, A., Shivanandan, R. et al. Genetic variation and phylogenetic relationship between two species of yellow catfish, Horabagrus brachysoma and H. nigricollaris (Teleostei: Horabagridae) based on RAPD and microsatellite markers. Mol Biol Rep 38, 2225–2232 (2011). https://doi.org/10.1007/s11033-010-0352-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-010-0352-3