Abstract
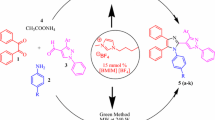
Optimization of Radziszewski's four-component reaction employing a microwave-assisted protocol, led to a small library of 48 imidazoles with a success rate of 65% (conversion > 45%). All three diversity points of the four-component reaction were varied. Aromatic and aliphatic inputs were successfully implemented and mono-, di-, tri- and tetrasubstituted imidazoles with various substitution patterns were synthesized. Furthermore, unsymmetrical diketones could successfully be used which improved the intrinsic diversity of the method significantly. If the unsymmetrical diketone 1,2-phenylpropanedione (R1 and R2) was used two regioisomers were formed. Depending on the type of amine (R4) and aldehyde (R3) applied, regioselectivity was modest to good. Based on these results, a reaction mechanism is proposed.
Similar content being viewed by others
Abbreviations
- MW:
-
microwave
- MCR:
-
multicomponent reaction
- EWG:
-
electron withdrawing group
References
Kitbunnadaj, R., Zuiderveld, O.P., Christophe, B., Hulscher, S., Menge, W.M.P.B., Gelens, E., Snip, E., Bakker, R.A., Celanire, S., Gillard, M., Talaga, P., Timmerman, H. and Leurs, R., Identification of 4-(1H-imidazol-4(5)-ylmethyl)pyridine (immethridine) as a novel, potent, and highly selective histamine H-3 receptor agonist, J. Med. Chem., 10 (2004) 2414–2417.
Adams, J.L., Boehm, J.C., Gallagher, T.F., Kassis, S., Webb, E.F., Hall, R., Sorenson, M., Garigipati, R., Griswold, D.E. and Lee, J.C., Pyrimidinylimidazole inhibitors of p38: Cyclic N-1 imidazole substituents enhance p38 kinase inhibition and oral activity, Bioog. Med. Chem. Lett., 11 (2001) 2867–2870.
Dinsmore, C.J., Williams, T.M., O'Neill, A.B., Liu, D., Rands, E., Culberson, J.C., Lobell, R.B., Koblan, K.S., Kohl, N.E., Gibbs, J.B., Oliff, A.I., Graham, S.L. and Hartman, G.D., Imidazole-containing diarylether and diarylsulfone inhibitors of farnesyl-protein transferase Bioog. Med. Chem. Lett., 9 (1999) 3301–3306.
Khanna, I.K., Weier, R.M., Yu, Y., Xu, X.D., Kosyk, F.J., Collins, P.W., Koboldt, C.M., Veenhuizen, A.W., Perkins, W.E., Casler, J.J., Masferrer, J.L., Zhang, Y.Y., Gregory, S.A., Seibert, K. and Isakson, P.C., 1,2-Diarylimidazoles as potent, cycloogygenase-2 selective, and orally active antiinflammatory agents, J. Med. Chem., 40 (1997) 1634–1647.
Revesz, L., Bonne, F. and Makavou, P., Vicinal bromostannanes as novel building blocks for the preparation of di- and trisubstituted imidazoles, Tetrahedron Lett., 39 (1998) 5171–5174.
Zhang, C., Woiwode, T.F., Short, K.S. and Mjalli, A.M.M., Synthesis of tetrasubstituted imidazoles via a-(N-acyl-N-alkylamino)-b-ketoamides on Wang resin, Tetrahedron Lett., 37 (1996) 751–754.
Orru, R.V.A. and de Greef, M., Recent advances in solution-phase multicomponent reactions for the synthesis of heterocyclic compounds, Synthesis, 10 (2003) 1471–1499.
Radziszewski, B., Ber., 15 (1882) 1493.
Drefahl, G. and Herma, H., Ubersuchungen uber stilbene 28. stilbenyl-imidazole, Chem. Ber., 93 (1960) 486.
Maduskuie Jr., T.P., Wilde, R.G., Billheimer, J.T., Cromley, D.A., germain, S., Gillies, P.J., Higley, C.A., Johnson, A.L., Pennev, P., Shimshick, E.J. and Wexler, R.R., Design, synthesis, and structure-activity relationship studies for a new imidazole series of J774 macrophages specific acyl-CoA: Cholesterol acyltransferase (ACAT) inhibitors., J. Med. Chem., 38 (1995) 1067–1083.
Liverton, N.J., Butcher, J.W., Clayborne, C.F., Claremon, D.A., Libby, B.E., Nguyen, K.T., Pitzenberger, S.M., Selnick, H.G., Smith, G.R., Tebben, A., Vacca, J.P., Varga, S.L., Agarwal, L., Dancheck, K., Forsyth, A.J., Fletcher, D.S., Frantz, B., Hanlon, W.A., Harper, C.F., Hofsess, S.J., Kostura, M., Lin, J., Luell, S., O'Neill, E.A., Orevillo, C.J., Pang, M., Parsons, J., Rolando, A., Sahly, Y., Visco, D.M. and O'Keefe, S.J., Design and synthesis of potent, selective and orally active bioavailable tetrasubtituted imidazole inhibitors of p38 mitogen-activated protein, J. Med. Chem., 42 (1999) 2180–2190.
Santos, J., Mintz, E.A., Zehnder, O., Bosshard, C., Bu, X.R. and Günter, P., New class of imidazoles incorporated with thiophenevinyl conjunction pathway for robust nonlinear optical chromophores, Tetrahedron Lett., 42 (2001) 805–808.
Davies, J.R., Kane, P.D. and Moody, C.J., N-H insertion reactions of rhodium carbenoids. Part 5: A convenient route to 1,3-azoles, Tetrahedron, 60 (2004) 3967–3977.
Wolkenberg, S.E., Wisnoski, D.D., Leister, W.H., Wang, Y., Zhao, Z. and Lindsley, C.W., Efficient synthesis of imidazoles from aldehydes and 1,2-diketones using microwave irradiation, Org. Lett., 6 (2004) 1453–1456.
Xu, Y., Liu, Y.Z., Rui, L. and Guo, Q.X., One-pot synthesis of tetra-substituted imidazoles on silicagel under microwave irradiation, Heterocycles, 63 (2004) 87.
Balalaie, S., Hashemi, M.M. and Akhbari, M., A novel one-pot synthesis of tetrasubstituted imidazoles under solvent-free conditions and microwave irradiation, Tetrahedron Lett., 44 (2003) 1709–1711.
Usyatinsky, A.Y. and Khmelnitsky, Y.L., Microwave-assisted synthesis of substituted imidazoles on a solid support under solvent-free conditions, Tetrahedron Lett., 41 (2000) 5031–5034.
Loupy, A., Petit, A., Hamelin, J., Texier-Boullet, F., Jacquault, P. and Mathe, D., New solvent-free organic synthesis using focused microwaves, Synthesis, (1998) 1213, and references cited herein.
Lew, A., Krutzik, P.O., Hart, M.E. and Chamberlin, R. Increasing rates of reaction: Microwave assisted organic synthesis for combinatorial chemistry, J. Comb. Chem., 4 (2002) 95–105 and references cited herein.
Perreux, L., Loupy, A. and Volatron, F., Solvent free preparation of amides from acids and primary amines under microwave irradiation, Tetrahedron, 58 (2002) 2155–2162.
Goretski, C, Krlej, A., Steffens, C. and Ritter, H., Green polymer chemsitry: Microwave-assisted single-step synthesis of various (meth)acrylamides and poly(meth)acrylamides directly from (meth)acrylic acid, Macromol. Rapid Commun., 25 (2004) 513–516.
The isolated yield was determined by work-up via the following procedure. After the reaction, the reaction mixture was taken up in DCM. Water was added and the water layer was neutralized with Na2CO3. The layers were separated and the water layer was extracted twice more with DCM. The combined DCM layers were dried over Na2SO4 and reduced in vacuo. The crude mixture was analysed by 1H-NMR and was found to contain 71% of the expected imidazole 1. Flash chromatography with DCM as the eluent gave imidazole 1 in 70% yield and 95% purity.
The use of the optimised conditions for the MCR to produce imidazole 1 in ethanol, toluene, acetonitrile, dioxane, t-butylmethylether, or dichloroethane instead of chloroform results in all cases in > 80% conversion to 1. However, chloroform gave the best results: The highest conversions were obtained and the least side products were formed.
Yan, B., New combichem QC standards guard the supply of compounds and ensure HTS results, Modern Drug Discovery, (2004) 30–34.
This was established by NOESY-NMR analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gelens, E., Kanter, F.J.J.D., Schmitz, R.F. et al. Efficient library synthesis of imidazoles using a multicomponent reaction and microwave irradiation. Mol Divers 10, 17–22 (2006). https://doi.org/10.1007/s11030-006-8695-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11030-006-8695-3