Abstract
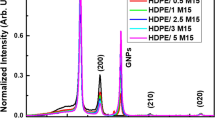
The non-isothermal degradation kinetics of polymer nanocomposites composed of high-density polyethylene (HDPE) as polymer matrix and aluminum nitride (AlN) as nanofiller has been studied at variable concentration of (nano) AlN. Field emission scanning electron microscopy was used to examine the microstructure of the HDPE polymer and the dispersion of AlN (nano) particles in it. The non-isothermal degradation kinetics of AlN (nano)/HDPE composites has been investigated by thermogravimetric analysis (TG) at multiple heating rates between 5 and 20 °C min−1. This study confirmed that the thermal stability of the filled nanocomposites is improved as compared to pristine HDPE. Thermal transport barrier effect is also enhanced with the increase in concentration of AlN (nano) loading. Dependence of activation energy \( (E_{\text{a}} ) \) on reaction extent (α) of the degradation process is estimated in light of ‘model-free’ methods (viz. Flynn–Wall–Ozawa and Starink) and advanced isoconversional approach. Ozawa’s generalized time ‘θ’ combination of the differential master plots ‘\( f(\alpha ) = ({\text{d}}\alpha /{\text{d}}\theta ) \)’ and combined [integral (θ) and differential \( ({\text{d}}\alpha /{\text{d}}\theta ) \)] master plots ‘\( Z(\alpha ) = ({\text{d}}\alpha /{\text{d}}\theta )\theta \)’ have been employed to investigate the appropriate thermal degradation mechanism(s) for AlN (nano)/HDPE composites. The results on ‘FZ master plots’ show that thermal decomposition of pristine HDPE follows two-dimensional diffusion mechanism, while AlN (nano)/HDPE composites undergo more complex degradation schematics, being dependent on the applied heating rates.












Similar content being viewed by others
References
Hussain F, Hojjati M, Okamoto M, Gorga RE. Review: polymer-matrix nanocomposites, processing, manufacturing, and application: an overview. J Compos Mater. 2006;40:1511–75.
Thostenson ET, Li C, Chou TW. Nanocomposites in context. Compos Sci Technol. 2005;65:491–516.
Olewnik E, Garman K, Czerwinski W. Thermal properties of new composites based on nanoclay, polyethylene and polypropylene. J Therm Anal Calorim. 2010;101:323–9.
Richard AV, John FM. Polymer nanocomposites with prescribed morphology: going beyond nanoparticle—filled polymers. Chem Mater. 2007;19:2736–51.
Paul DR, Robeson LM. Polymer nanotechnology: nanocomposites. Polymer. 2008;49:3187–204.
Gonzalez-Benito J, Olmos D. Efficient dispersion of nanoparticles in the thermoplastic polymers. Plast Res online Soc Plast Eng. 2010;. doi:10.1002/spepro.002566.
Chrissafis K, Bikiaris D. Can nanoparticles really enhance thermal stability of polymers? Part I: an overview on thermal decomposition of addition polymers. Thermochim Acta. 2011;523:1–24.
Bikiaris D. Can nanoparticles really enhance thermal stability of polymers? Part II: an overview on thermal decomposition of polycondensation polymers. Thermochim Acta. 2011;523:25–45.
Mofokeng JP, Luyt AS. Morphology and thermal degradation studies of melt-mixed poly(hydroxybutyrate-co-valerate) (PHBV)/poly(e-caprolactone) (PCL) biodegradable polymer blend nanocomposites with TiO2 as filler. J Mater Sci. 2015;50:3812–24.
Omrani A, Rostami AA, Ravar F. Advanced isoconversional and master plot analyses on solid-state degradation kinetics of a novel nanocomposite. J Therm Anal Calorim. 2013;111:677–83.
Park JW, Oh SC, Lee HP, Kim HT, Yoo KO. A kinetic analysis of thermal degradation of polymers using a dynamic method. Polym Degrad Stab. 2000;67:535–40.
Chrissafis K, Paraskevopoulos KM, Pavlidou E, Bikiaris D. Thermal degradation mechanism of HDPE nanocomposites containing fumed silica nanoparticles. Thermochim Acta. 2009;485:65–71.
Sanchez-Jimenez PE, Perez-Maqueda LA, Perejon A, Criado JM. Generalized kinetic master plots for the thermal degradation of polymers following a random scission mechanism. J Phys Chem A. 2010;114:7868–76.
Al-Salem SM, Lettieri P. Kinetic study of high density polyethylene (HDPE) pyrolysis. Chem Eng Res Des. 2010;88:1599–606.
Zabihi O, Omrani A, Rostami AA. Thermo-oxidative degradation kinetics and mechanism of the system epoxy nano-composite reinforced with nano-Al2O3. J Therm Anal Calorim. 2012;108:1251–60.
Sánchez-Jiménez PE, Pérez-Maqueda LA, Perejón A, Criado JM. Generalized master plots as a straightforward approach for determining the kinetic model: the case of cellulose pyrolysis. Thermochim Acta. 2013;552:54–9.
Rajeshwari P, Dey TK. Structural and thermal properties of HDPE/n-AlN polymer nano-composites. J Therm Anal Calorim. 2014;118:1513–30.
Rajeshwari P. Kinetic analysis of the non-isothermal degradation of high-density polyethylene filled with multi-wall carbon nanotubes. J Therm Anal Calorim. 2016;123:1523–44.
Ozawa T. A new method of analyzing thermogravimetric data. Bull Chem Soc Jpn. 1965;38:1881–6.
Flynn JH, Wall LA. A quick direct method for the determination of activation energy from thermogravimetric data. J Polym Sci B Polym Lett. 1966;4:323–8.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29:1702–6.
Friedman HL. Kinetic and gaseous products of thermal decomposition of polymers. J Macromol Sci A. 1967;1:57–79.
Starink MJ. The determination of activation energy from linear heating rate experiments: a comparison of the accuracy of isoconversional methods. Thermochim Acta. 2003;404:163–76.
Akahira T, Sunose T. Method of determining activation deterioration constant of electrical insulating materials. Res Rep Chiba Inst Technol (Sci Technol). 1971;16:22–31.
Vyazovkin S, Dollimore D. Linear and nonlinear procedures in isoconversional computations of the activation energy of non-isothermal reactions in solids. J Chem Inf Comput Sci. 1996;36:42–5.
Vyazovkin S. Modification of the integral isoconversional method to account for variation in the activation energy. J Comput Chem. 2001;2:178–83.
Coats AW, Redfern JP. Kinetics parameters from thermogravimetric data. Nature. 1964;201:68–9.
Vyazovkin S, Wight CA. Model-free and model-fitting approaches to kinetic analysis of isothermal and non-isothermal data. Thermochim Acta. 1999;340–341:53–68.
Malek J. The kinetic analysis of non-isothermal data. Thermochim Acta. 1992;200:257–69.
Criado JM, Malek J, Ortega A. Applicability of the master plots in kinetic analysis of a non-isothermal rate. Thermochim Acta. 1989;147:377–85.
Gotor FJ, Criado JM, Malek J, Koga N. Kinetic analysis of solid-state reactions: the universality of master plots for analyzing isothermal and non-isothermal experiments. J Phys Chem A. 2000;104:10777–82.
Turmanova SCh, Genieva SD, Dimitrova AS, Vlaev LT. Non-isothermal degradation kinetics of filled with rise husk ash polypropene composites. Express Polym Lett. 2008;2:133–46.
Montero B, Ramirez C, Rico M, Barral L, Diez J, Lopez J. Effect of an epoxy octasilsesquioxane on the thermodegradation of an epoxy/amine system. Polym Int. 2010;59:112–8.
Chen Y, Wang Q. Thermal oxidative degradation kinetics of flame-retarded polypropylene with intumescent flame-retardant master batches in situ prepared in twin-screw extruder. Polym Degrad Stab. 2007;92:280–91.
Chrissafis K, Paraskevopoulos KM, Tsiaoussis I, Bikiaris D. Comparative study of the effect of different nanoparticles on the mechanical properties, permeability, and thermal degradation mechanism of HDPE. J Appl Polym Sci. 2009;114:1606–18.
Koga N. Kinetic analysis of thermo-analytical data by extrapolating to infinite temperature. Thermochim Acta. 1995;258:145–59.
Koga N, Criado JM. Kinetic analyses of solid-state reactions with a particle-size distribution. J Am Ceram Soc. 1998;81:2901–9.
Malek J, Mitsuhashi T, Criado JM. Kinetic analysis of solid state processes. J Mater Res. 2001;16:1862–71.
Criado JM, Perez-Maqueda LA, Gotor FJ, Malek J, Koga N. A unified theory for the kinetic analysis of solid state reactions under any thermal pathway. J Therm Anal Calorim. 2003;72:901–6.
Malek J, Koga N, Perez-Maqueda LA, Criado JM. The Ozawa’s generalized time concept and YZ-master plots as a convenient tool for kinetic analysis of complex processes. J Therm Anal Calorim. 2013;113:1437–46.
Sheppard LM. Aluminum nitride: a versatile but challenging material. Am Ceram Soc Bull. 1990;69:1801–12.
Mroz TJ Jr. Annual materials review: aluminum nitride. Am Ceram Soc Bull. 1992;71:782–6.
Galli P, Vecellio G. Polyolefins: the most promising large-volume materials for the 21st century. J Polym Sci A Polym Chem. 2004;42:396–415.
Vyazovkin S, Burnham AK, Criado JM, Maqueda LA, Popescu C, Sbirrazzuoli N. ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta. 2011;520:1–19.
Doyle CD. Series approximations to the equation of thermo-gravimetric data. Nature. 1965;207:290–1.
Vyazovkin S. Evaluation of activation energy of thermally stimulated solid-state reactions under arbitrary variation of temperature. J Comput Chem. 1997;18:393–402.
Zabihi O, Khodabandeh A. Understanding of thermal/thermo-oxidative degradation kinetics of polythiophene nanoparticles. J Therm Anal Calorim. 2013;112:1507–13.
Senum GI, Yang RT. Rational approximations of the integral of the Arrhenius functions. J Therm Anal. 1977;11:445–7.
Annakutty KS, Kishore K. Novel polymeric flame retardant plasticizers for poly-vinyl chloride. Eur Polym J. 1993;29:1387–90.
Zabihi O, Khodabandeh A, Mostafavi SM. Preparation, optimization and thermal characterization of a novel conductive thermoset nanocomposite containing polythiophene nanoparticles using dynamic thermal analysis. Polym Degrad Stab. 2012;97:3–13.
Peng Z, Li SD, Huang MF, Xu K, Wang C, Li PW, Chen XG. Thermogravimetric analysis of methyl methacrylate-graft-natural rubber. J Appl Polym Sci. 2002;85:2952–5.
Doyle CD. Estimating thermal stability of experimental polymers by empirical thermogravimetric analysis. Anal Chem. 1961;33:77–9.
Chrissafis K. Kinetics of thermal degradation of polymers complementary use of isoconversional and model-fitting methods. J Therm Anal Calorim. 2009;95:273–83.
Ozawa T. Kinetic analysis of derivative curves in thermal analysis. J Therm Anal. 1970;2:301–24.
Ozawa T. Non-isothermal kinetics and generalized time. Thermochim Acta. 1986;100:109–18.
Simha R, Wall LA. Kinetics of chain depolymerisation. J Phys Chem. 1952;56:707–15.
Wall LA, Madorsky SL, Brown DW, Straus S, Simha R. The depolymerization of polymethylene and polyethylene. J Am Chem Soc. 1954;76:3430–7.
Tsuchiya Y, Sumi K. Thermal decomposition products of polyethylene. J Polym Sci A Polym Chem. 1968;6:415–24.
Tsuchiya Y, Sumi K. Thermal decomposition products of polypropylene. J Polym Sci A Polym Chem. 1969;7:1599–607.
Tsuchiya Y, Sumi K. Thermal decomposition products of polyisobutylene. J Polym Sci A Polym Chem. 1969;7:813–26.
Kiran E, Gillham JK. Pyrolysis-molecular weight chromatography: a new on-line system for analysis of polymers. II. Thermal decomposition of polyolefins: polyethylene, polypropylene, polyisobutylene. J Appl Polym Sci. 1976;20:2045–68.
Lattimer RP. Pyrolysis field ionization mass spectrometry of polyolefins. J Anal Appl Pyrolysis. 1995;31:203–25.
Peterson JD, Vyazovkin S, Wight CA. Kinetics of the thermal and thermo-oxidative degradation of polystyrene, polyethylene and poly(propylene). Macromol Chem Phys. 2001;202:775–84.
Murata K, Hirano Y, Sakata Y, Uddin MA. Basic study on a continuous flow reactor for thermal degradation of polymers. J Anal Appl Pyrolysis. 2002;65:71–90.
Sanchez-Jimenez PE, Perez-Maqueda LA, Perejon A, Criado JM. Combined kinetic analysis of thermal degradation of polymeric materials under any thermal pathway. Polym Degrad Stab. 2009;94:2079–85.
Acknowledgements
One of the authors (TKD) is grateful to the Department of Science & Technology, Government of India (New Delhi), for financial assistance in the form of a research project grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rajeshwari, P., Dey, T.K. Advanced isoconversional and master plot analyses on non-isothermal degradation kinetics of AlN (nano)-reinforced HDPE composites. J Therm Anal Calorim 125, 369–386 (2016). https://doi.org/10.1007/s10973-016-5406-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5406-x