Abstract
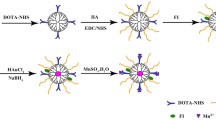
In this study, we designed and synthesized [19F]FDG-(2-deoxy-2-[fluorine-19]fluoro-d-glucose) conjugated iron oxide magnetic nanoparticles ([19F]FDG-MNPs) for hybrid imaging and hyperthermia treatment. MNPs were synthesized, silica coated, and fabricated with TEOS (tetraethyl orthosilicate). They were then covered with hyaluronic acid (HA) to enhance their bioavailability. The modified MNPs were conjugated with [19F]FDG and optically labeled with ICG (indocyanine green). The in vitro bioaffinities were surveyed in MCF7 and PC3 cell lines. In vivo bioaffinies were determined using Sprague–Dawley rats.














Similar content being viewed by others
References
Varani M, Galli F, Auletta S, Signore A (2018) Radiolabelled nanoparticles for cancer diagnosis. Clin Transl Imaging 6(4):271–292
Ren X, Chen H, Yang V, Sun D (2014) Iron oxide nanoparticle-based theranostics for cancer imaging and therapy. Front Chem Sci Eng 8(3):253–264
Medine EI, Ünak P, Sakarya S, Özkaya F (2011) Investigation of in vitro efficiency of magnetic nanoparticle-conjugated 125I-uracil glucuronides in adenocarcinoma cells. J Nanoparticle Res 13(10):4703–4715
Mi Y, Shao Z, Vang J, Kaidar-Person O, Wang AZ (2016) Application of nanotechnology to cancer radiotherapy. Cancer Nanotechnol 7(1):11
Kim GG, Lee JY, Choi PS, Vyas CK, Yang SD, Hur MG, Park JH (2018) Synthesis and evaluation of triphenylphosphonium conjugated 18F-labeled silica nanoparticles for PET imaging. J Radioanal Nucl Chem 316(3):1099–1106
Laurent S, Morteza M (2011) Superparamagnetic iron oxide nanoparticles: promises for diagnosis and treatment of cancer. Int J Mol Epidemiol Genet 2(4):367
Wang AY, Kuo CL, Lin JL, Fu CM, Wang YF (2010) Study of magnetic ferrite nanoparticles labeled with 99mTc-pertechnetate. J Radioanal Nucl Chem 284:405–413
Toksöz F, Ünak P, Medine Eİ, Sakarya S, Ünak G, Timur S (2013) 18FDG conjugated magnetic nanoparticle probes: synthesis and in vitro investigation on MCF7 breast cancer cells. J Radioanal Nucl Chem 295(3):1789–1796
Nosrati S, Shanehsazzadeh S, Yousefnia H, Gholami A, Grüttner C, Jalilian AR, Hosseini RH, Lahooti A (2016) Biodistribution evaluation of 166Ho–DTPA–SPION in normal rats. J Radioanal Nucl Chem 307(2):1559–1566
Shanehsazzadeh S, Oghabian MA, Lahooti A, Abdollahi M, Abolghasem Haeri S, Amanlou M, Daha FJ, Allen BJ (2013) Estimated background doses of [67Ga]-DTPA-USPIO in normal Balb/c mice as a potential therapeutic agent for liver and spleen cancers. Nucl Med Commun 34:915–925
Shanehsazzadeh S, Oghabian MA, Daha FJ, Amanlou M, Allen BJ (2013) Biodistribution of ultra small superparamagnetic iron oxide nanoparticles in BALB mice. J Radioanal Nucl Chem 295:1517–1523
Zhao Y, Yao Q, Tan H, Wu B, Hu P, Wu P, Gu Y, Zhang C, Cheng D, Shi H (2014) Design and preliminary assessment of 99mTc-labeled ultrasmall superparamagnetic iron oxide-conjugated bevacizumab for single photon emission computed tomography/magnetic resonance imaging of hepatocellular carcinoma. J Radioanal Nucl Chem 299:1273–1280
Aras O, Pearce G, Watkins AJ, Nurili F, Medine EI, Guldu Kozgus O, Tekin V, Wong J, Ma X, Ting R, Unak P, Akin O (2018) An in-vivo pilot study into the effects of FDG-mNP in cancer in mice. PLoS ONE 13(8):e0202482
Watkins AJ, Pearce G, Unak P, Guldu KO, Yasakci V, Akin O, Aras O, Wong J, Ma X (2018) Tissue morphology and gene expression characterisation of transplantable adenocarcinoma bearing mice exposed to fluorodeoxyglucose-conjugated magnetic nanoparticles. J Biomed Nanotechnol 14:1979–1991
Golovin YI, Gribanovsky SL, Golovin DY, Klyachko NL, Majouga AG, Master AM, Sokolsky M, Kabanov AV (2015) Towards nanomedicines of the future: Remote magneto-mechanical actuation of nanomedicines by alternating magnetic fields. J Control Release 219(43–60):7
Bañobre-López M, Antonio T, Jose R (2013) Magnetic nanoparticle-based hyperthermia for cancer treatment. Rep Pract Oncol Radiother 18(6):397–400
Thomas RG, Moon MJ, Lee H, Sasikala ARK, Kim CS, Park IK, Jeong YY (2015) Hyaluronic acid conjugated superparamagnetic iron oxide nanoparticle for cancer diagnosis and hyperthermia therapy. Carbohydr Polym 131:439–446
Longmire M, Choyke PL, Kobayashi H (2008) Clearance properties of nano-sized particles and molecules. Nanomedicine (Lond) 3:703–717
Del Campo A, Sen T, Lellouche JP, Bruce IJ (2005) Multifunctional magnetite and silica-magnetite nanoparticles: synthesis, surface activation and applications in life sciences. J Magn Magn Mater 293(1):33–40
Jain TK, Morales MA, Sahoo SK, Leslie-Pelecky DL, Labhasetwar V (2005) Iron oxide nanoparticles for sustained delivery of anticancer agents. Mol Pharm 2(3):194–205
Mok H, Miqin Z (2013) Superparamagnetic iron oxide nanoparticle-based delivery systems for biotherapeutics. Expert Opin Drug Deliv 10(1):73–87
Uthaman S, Lee SJ, Cherukula K, Cho CS, Park IK (2015) Polysaccharide-coated magnetic nanoparticles for imaging and gene therapy. BioMed Res Int. https://doi.org/10.1155/2015/959175
Laurent S, Forge D, Port M, Roch A, Robic C, Vander Elst L, Muller RN (2008) Magnetic iron oxide nanoparticles: synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem Rev 108(6):2064–2110
Cowman MK, Lee HG, Schwertfeger KL, McCarthy JB, Turley EA (2015) The content and size of hyaluronan in biological fluids and tissues. Front Immunol 6:261
El-Dakdouki MH, Zhu DC, El-Boubbou K, Kamat M, Chen J, Li W, Huang X (2012) Development of multifunctional hyaluronan-coated nanoparticles for imaging and drug delivery to cancer cells. Biomacromol 13(4):1144–1151
Chan LC, Cox BG (2007) Kinetics of amide formation through carbodiimide/N-hydroxybenzotriazole (HOBt) couplings. J Org Chem 72(23):8863–8869
Hafid M, Dalili W, Yazami SI, Merzouk N (2010) Growth and characterization of Fe3O4 nanoparticles in silica matrix. Phase Transit 83(8):627–633
Wu W, He Q, Chen H, Tang J, Nie L (2007) Sonochemical synthesis, structure and magnetic properties of air-stable Fe3O4/Au nanoparticles. Nanotechnology 18(14):145609
Carraway KL, Triplett RB (1970) Reaction of carbodiimides with protein sulfhydryl groups. BBA Protein Struct 200(3):564–566
Babu P, Sinha S, Surolia A (2007) Sugar-quantum dot conjugates for a selective and sensitive detection of lectins. Bioconjug Chem 18(1):146–151
Gillies JM, Prenant C, Chimon GN, Smethurst GJ, Dekker BA, Zweit J (2006) Microfluidic technology for PET radiochemistry. Appl Radiat Isot 64(3):333–336
Subramanian M, Pearce G, Guldu OK, Tekin V, Miaskowski A, Aras O, Unak P (2016) A pilot study into the use of FDG-mNP as an alternative approach in neuroblastoma cell hyperthermia. IEEE Trans Nanobioscience 15(6):517–525
Yilmaz T, Unak P, Muftuler FZB, Medine EI, Ichedef C, Kilcar AY (2015) Magnetic nanoparticle-conjugated and radioiodinated-DESG: in vitro and in vivo efficiency investigation. J Radioanal Nucl Chem 303(1):63–69
Guldu OK, Unak P, Medine EI, Barlas FB, Muftuler FZB, Timur S (2015) Radioiodinated magnetic nanoparticles conjugated with moxifloxacin: synthesis and in vitro biological affinities. Int J Polym Mater Polym Biomater 64(5):253–259
Namgung R, Zhang Y, Fang QL, Singha K, Lee HJ, Kwon IIK, Jeong YY, Park I, Son SJ, Kim WJ (2011) Multifunctional silica nanotubes for dual-modality gene delivery and MR imaging. Biomaterials 32(11):3042–3052
Roonasi P, Holmgren A (2009) A Fourier transform infrared (FTIR) and thermogravimetric analysis (TGA) study of oleate adsorbed on magnetite nano-particle surface. Appl Surf Sci 255(11):5891–5895
Andrade AL, Valente MA, Ferreira JM, Fabris JD (2012) Preparation of size-controlled nanoparticles of magnetite. J Magn Magn Mater 324(10):1753–1757
Chen B, Miller RJ, Dhal PK (2014) Hyaluronic acid-based drug conjugates: state-of-the-art and perspectives. J Biomed Nanotechnol 10(1):4–16
Choi KY, Yoon HY, Kim JH, Bae SM, Park RW, Kang YM, Kim IS, Kwon IC, Choi K, Jeong SY, Kim K et al (2011) Smart nanocarrier based on PEGylated hyaluronic acid for cancer therapy. ACS Nano 5(11):8591–8599
Yang XY, Li YX, Li M, Zhang L, Feng LX, Zhang N (2013) Hyaluronic acid-coated nanostructured lipid carriers for targeting paclitaxel to cancer. Cancer Lett 334(2):338–345
Choi KY, Saravanakumar G, Park JH, Park K (2012) Hyaluronic acid-based nanocarriers for intracellular targeting: interfacial interactions with proteins in cancer. Colloids Surf B Biointerfaces 99:82–94
Veiman KL, Künnapuu K, Lehto T, Kiisholts K, Pärn K, Langel Ü, Kurrikoff K (2015) PEG shielded MMP sensitive CPPs for efficient and tumor specific gene delivery in vivo. J Control Release 209:238–247
Lokeshwar VB, Selzer MG (2008) Hyalurondiase: both a tumor promoter and suppressor. Semin Cancer Biol 18(4):281–287
Cheng G, Zheng SY (2014) Construction of a high-performance magnetic enzyme nanosystem for rapid tryptic digestion. Sci Rep 4:6947
Garg PK, Alston KL, Welsh PC, Zalutsky MR (1996) Enhanced binding and inertness to dehalogenation of α-melanotropic peptides labeled using N -succinimidyl 3-iodobenzoate. Bioconjug Chem 7(2):233–239
Unak P, Cetinkaya B (2005) Absorbed dose estimates at the cellular level for 131I. Appl Radiat Isot 62(6):861–869
Dagdeviren K, Unak P, Bekis R, Biber FZ, Akdurak S, Ulker O, Ergur B, Ertay T, Durak H (2007) Radioiodinated magnetic targeted carriers (131I-MTC). J Radioanal Nucl Chem 273(3):635–639
Ma SY, Nam YR, Jeon J, Rho JK, Lee DE, Choi DS, Jang BS, Park SH (2015) Simple and efficient radiolabeling of hyaluronic acid and its in vivo evaluation via oral administration. J Radioanal Nucl Chem 305:139–145
Zheng X, Xing D, Zhou F, Wu B, Chen WR (2011) Indocyanine green-containing nanostructure as near infrared dual-functional targeting probes for optical imaging and photothermal therapy. Mol Pharm 8(2):447–456
McSheehy PM, Leach MO, Judson IR, Griffiths JR (2000) Metabolites of 2′-fluoro-2′-deoxy-d-glucose detected by 19F magnetic resonance spectroscopy in vivo predict response of murine RIF-1 tumors to 5-fluorouracil. Can Res 60(8):2122–2127
Pauwels EKJ, Ribeiro MJ, Stoot JHMB, McCready VR, Bourguignon M, Maziere B (1998) FDG accumulation and tumor biology. Nucl Biol Med 25(4):317–322
Wood TE, Dalili S, Simpson CD, Hurren R, Mao X, Saiz FS, Klip A (2008) A novel inhibitor of glucose uptake sensitizes cells to FAS-induced cell death. Mol Cancer Ther 7(11):3546–3555
Zahr AS, Davis CA, Pishko MV (2006) Macrophage uptake of core–shell nanoparticles surface modified with poly(ethylene glycol). Langmuir 22(14):8178–8185
Beduneau A, Ma Z, Grotepas CB, Kabanov A, Rabinow BE, Gong N, Mosley RL, Dou H, Boska MD, Gendelman HE (2009) Facilitated monocyte-macrophage uptake and tissue distribution of superparmagnetic iron-oxide nanoparticles. PLoS ONE 4(2):1–12
Nicolete R, dos Santos DF, Faccioli LH (2011) The uptake of PLGA micro or nanoparticles by macrophages provokes distinct in vitro inflammatory response. Int Immunopharmacol 11(10):1557–1563
Barbaro D, Bari LD, Gandin V, Evangelisti C, Vitulli G, Schiavi E, Marzano C, Ferretti AM, Salvadori PG (2015) Glucose-coated superparamagnetic iron oxide nanoparticles prepared by metal vapour synthesis are electively internalized in a pancreatic adenocarcinoma cell line expressing GLUT1 transporter. PLoS ONE 10(4):e0123159
Hu C, Niestroj M, Yuan D, Chang S, Chen J (2015) Treating cancer stem cells and cancer metastasis using glucose-coated gold nanoparticles. Int J Nanomedicine 10:2065
Gromnicova R, Davies HA, Sreekanthreddy P, Romero IA, Lund T, Roitt IM, Phillips JB, Male DK (2013) Glucose-coated gold nanoparticles transfer across human brain endothelium and enter astrocytes in vitro. PLoS ONE 8(12):e81043
He C, Jiang S, Jin H, Chen S, Lin G, Yao H, Wang X, Mi P, Ji Z, Lin Y, Lin Z et al (2016) Mitochondrial electron transport chain identified as a novel molecular target of SPIO nanoparticles mediated cancer-specific cytotoxicity. Biomaterials 83:102–114
Acknowledgements
The authors wish to thank Dr. Omer Aras for their assistance in the TEM Image of FDG MNPs. The work is MS thesis of Volkan Yasakçı from Graduate School of Natural and Applied Sciences and has been partially presented at EANM’17—Annual Congress of the European Association of Nuclear Medicine, October 21–25, 2017 in Vienna/Austria.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yasakci, V., Tekin, V., Guldu, O.K. et al. Hyaluronic acid-modified [19F]FDG-conjugated magnetite nanoparticles: in vitro bioaffinities and HPLC analyses in organs. J Radioanal Nucl Chem 318, 1973–1989 (2018). https://doi.org/10.1007/s10967-018-6282-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-018-6282-6