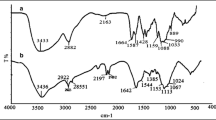
Abstract
The monomer, 4-acrylamidobenzenesulfonamide (ABS), was synthesized via reaction of acryloyl chloride with 4-aminobenzenesulfonamide in acetone at 0 °C. This monomer was then grafted onto chitosan using solution containing 2% acetic acid and mixture of K2S2O8 and Na2SO3 as the redox promoter. An optimal G% of 150% was obtained when the process is conducted at 60 °C for 3 h employing 3.0 × 10−3 M K2S2O8 and 1.5 × 10−3 M Na2SO3. The graft copolymers, chitosan-g-poly(4-acrylamidobenzenesulfonamide), were characterized by using FTIR, XRD, and SEM. The results were shown that the crystallinity of chitosan is enhanced by increasing the monomer content through the grafting process. Potential Antimicrobial activities of the permethyl ammonium salt forms of chitosan and its grafted copolymers against selected microorganisms were evaluated. The results show that the graft copolymers display better inhibitory effects on the growth of bacteria and some fungi than does chitosan.








Similar content being viewed by others
References
Muzzarelli RAA (1977) Chitin. Pergamon Press, Oxford
Jennings JA, Bumgardner JD (2016) Production 1 of chitosan-based hydrogels for biomedical application, Chapter in chitosan based biomaterials, fundamentals, Woodhead, Chapter 12. 1:295–319
Muzzarelli RAA (1996) Chitin enzymology. Ed., AtecEdizioni, Grottammare, Italy, vol. 2
Roberts GAF (1992) Chitin chemistry. Macmillan, London
Jennings JA, Bumgardner JD (2016) The role of nanotechnology and chitosan based biomaterials for tissue engineering and therapeutic delivery, in Chitosan biomaterials, Woodhead, chapter 1, 2:1–29
Agnihotri SA, Mallikarjuna NN, Aminabhavi TM (2004) Recent advances on chitosan-based micro and nanoparticles in drug delivery. Control Release 100:5–28
Rokhade AP, Aminabhavi TM (2007) Synthesis and characterization of semi-interpenetrating polymer network microspheres of acrylamyde grafted dextran and chitosan for the controlled release of acyclovir. CarbohydPolym 67:605–613
Angadi SC, Manjeshwar TM, Aminabhavi TM (2010) Interpenetrating polymer network blend microspheres of chitosan and hydroyethyl cellulose for controlled release of isoniazid. Int J Biol Macromol 47:171–179
Sankararamakrishnan N, Sanghi R (2006) Preparation and characterization of a novel xanthated chitosan. Carbohyd Polym 66:160–167
Elkholy SS, Khalil KD, Elsabee MZ (2006) Homogeneous and Hetrogeneous grafting of chitosan with 4-vinyl pyridine. Appl Polym Sci 99:3308–3317
Al Sagheer FA, Khalil KD, Ibrahim EI (2013) Synthesis and characterization of chitosan-g-poly(2-(furan-2-carbonyl)-acrylonitrile): grafting of chitosan via novel monomer prepared by Baylius-Hillman reaction. Eur Polym J 49:1662–1672
Elkholy SS, Khalil KD, Elsabee MZ (2007) Grafting of vinyl acetate onto chitosan and biocidal activity of the graft copolymers. J Appl Polym Sci 103:1651–1663
Elkholy SS, Khalil KD, Elsabee MZ (2011) Grafting of acryloylcyanoacetohydrazide onto Chitosan. J Polym Res 18:459–467
Khedr MA, Waly AI, Hafez AI, Ali H, Gadallah H (2012) Synthesis and Kinetic Study of Chitosan – g- Poly (acrylic acid) nanoparticles.(Part I). Aust J Basic Appl Sci 6(6):174–182
Lv P, Bin Y, Li Y, Chen R, Wang X, Zhao B (2009) Studies on graft copolymerization of chitosan with acrylonitrile by the redox system. Polymer 50:5675–5680
Liu Y, Li Y, Lv J, Wu G, Li J (2005) Graft copolymerization of methyl methacrylate onto chitosan initiated by potassium Ditelluratocuprate(III). J Mol Sci Part A Pure Appl Chem 42:1169–1180
Pourjavadi A, Mahdavinia GR, Zohuriaan-Mehr MJ, Omidian H (2003) Modified chitosan. 1. Optimized CAN-induced synthesis of chitosan-graft-polyacrylonitrile. J App Poly Sci 88:2048–2054
Khalil KD, Ibrahim EI, Al-Sagheer FA (2014) Synthesis and characterization of chitosan-g-poly (2-cyano-1-(pyridin-3-yl) allyl acrylate) copolymer using a novel monomer prepared via Morita Baylis–Hillman reaction. Polym Inter 63:2042–2051
Naguib HF, Al Sagheer FA, Ali AM, Elsabee MZ (1996) Polymerization behavior of a new vinyl thiourea derivatives. Eur Polym J 32(8):985–991
Al Sagheer FA, Ali AM, Reyed MA, Elsabee MZ (1997) Preparation and polymerization of vinyl monomers containing a heterocyclic pendant group. Polym Int 44:88–94
Ali AM, Al Sagheer FA, Elsabee MZ (1999) Study of the polymerization of solid vinyl monomers using differential scanning calorimetry. Polym Test 18(4):313–319
Al-Sagheer FA, Khalil KD, Ibrahim EI (2014) Crystallinity, antimicrobial activity and dyeing properties of chitosan-g-poly(N-acryloylmorpholine) copolymer. EurPolym J 58:164–172
Aguero L, Guerrero-Ramirez LG, Katime I (2010) New family of functinalized monomers based on amines: a novel synthesis that exploits the nucleophilic substitution reaction. Mater SciAppl 1:103–108
Chinelatto MA, Agnelli JAM, Canevarolo SV (2014) Synthesis and characterization of copolymers from hindered amines and vinyl monomers. Polimers 24:30–36
Kong M, Chen XG, Xing K, Park HJ (2010) Antimicrobial properties of chitosan and mode of action: a state of the art review. Inter J Food Microbiol 144:51–63
Sashiwa H, Aiba S (2004) Chemically modified chitin and chitosan as biomaterials. Prog Polym Sci 29:887–908
Qi L, Xu Z, Jiang X, Hu C, Zou X (2004) Preparation and antibacterial activity of chitosan nanoparticles. Carbohydr Res 339:2693–2700
Mirzaei BE, Ramazani SAA, Shafiee M, Danaei M (2013) Studies on glutaraldehyde crosslinked chitosan hydrogel properties for drug delivery systems. Int J Polym Mater 62:605–611
Reddy NS, Rao AS, Chari MA, Kumar VR, Jyothy V, Himabindu V (2012) Synthesis and antibacterial activity of sulfonamide derivatives at C-8 alkyl chain of anacardic acid mixture isolated from a natural product cashew nut shell liquid (CNSL). J Chem Sci 124:723–730
Dogruer DS, Urlu S, Onkol T, Ozcelik B, Sahin MF (2010) Synthesis of some pyridazine derivatives carrying urea, thiourea, and sulfonamide moieties and their antimicrobial activity. Turk J Chem 34(1):57–65
Argyropoulou I, Geronikaki A, Vicini P, Zani F (2009) Synthesis and biological evaluation of sulfonamidethiazole and benzothiazole derivatives as antimicrobial agents. ARKIVOC vi:89–102
Alsughayer A, Elassar AA, Al Sagheer FA, Mustafa S (2012) Synthesis and characterization of polysulfanilamide and its copolymers: bioactivity and drug release. Pharmaceutical Chem J 46:418–428
Al Sagheer FA, Al Sughayer MA, Muslim S, Elsabee MZ (2009) Extraction and characterization of chitin and chitosan from marine sources in Arabian gulf. Carbohyd Polym 77:410–419
Ogawa K, Hirano S, Miyanishi T, Yui T, Watanabe T (1984) New polymorph of chitosan. Macromolecules 17:973–975
Wang SF, Shen L, Zhang WD, Tong YJ (2005) Preparation and mechanical properties of chitosan/carbon nanotubes composites. Biomacromolecules 6:3067–3072
Mun GA, Nurkeeva ZS, Dergunov SA, Nam IK, Maimakov TP, Shaikhutdinov EM, Lee SC, Park K (2008) Studies on graft copolymerization of 2-hydroxyethyl acrylate onto chitosan. React Funct Polym 68:389–395
Ding W, Lian Q, Samuels RJ, Polk MB (2003) Synthesis and characterization of a novel derivative of chitosan. Polymer 44:547–556
Jia Z, Shen D, Xu W (2001) Synthesis and antibacterial activities of quaternary ammonium salt of chitosan. Carbohydr Res 333:1–6
deBritto D, Assis OBG (2007) A novel method for obtaining a quaternary salt of chitosan. Carbohydr Polym 69(2):305–310
Henry RJ (1944). Bacteriol Rev 7(4):175–260
Isaacson D M, Kirschbaum J (1986) Assays of antimicrobial substances. In: Manual of industrial microbiology and biotechnology (Demain, A. L.; Solomon, N. A.; Eds), ASM, Washington, DC, pp. 410–435
Liu H, Du Y, Wang X, Sun L (2004) Chitosan kills bacteria through cell membrane damage. Int J Food Microbiol 95(2):147–155
Tayel AA, Moussa S, Wael F, Knittel D, Opwis K, Schollmeyer E (2010) Anticandidal action of fungal chitosan against Candida Albicans. Int J Biol Macromol 47(4):454–457
Seyfarth F, Schliemann S, Elsner P, Hipler UC (2008) Antifungal effect of high- and low-molecular-weight chitosan hydrochloride, carboxymethyl chitosan, chitosan oligosaccharide and N-acetyl-d-glucosamine against Candida albicans, Candida kruseiand Candida glabrata. Int J Pharm 353:139–148
Mourya VK, Inamdar NN (2009) Trimethyl chitosan and its applications in drug delivery. J Mat Sci: Mater Med 20:1057–1079
Acknowledgements
The authors wish to acknowledge financial support provided by the Kuwait University under the project SC03/10. They also express their appreciation for technical support from the E.M unit and the general facilities projects GS01/01, GS01/03, GS01/05, GS03/01, under the GFS program. Also, the authors thank the Nanoscopy Science Center at Kuwait University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 1842 kb)
Rights and permissions
About this article
Cite this article
Al-Sagheer, F., Khalil, K., Mahmoud, H. et al. Chitosan-g-poly(4-acrylamidobenzenesulfonamide) copolymers: synthesis, characterization, and bioactivity. J Polym Res 24, 230 (2017). https://doi.org/10.1007/s10965-017-1381-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-017-1381-6