Abstract
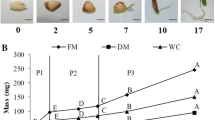
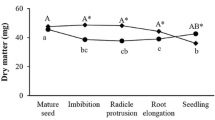
Amino acids, polyamines, 3-indoleacetic acid (IAA), abscisic acid (ABA), buffer-soluble protein and starch contents and dry matter accumulation were analyzed in megagametophytes containing developing embryos during seed development in Pinus taeda. The highest total amino acids and polyamine contents occurred at the cotyledonary stage, followed by a significant decrease in the mature seed. Free polyamines exhibited higher levels than conjugated ones, with putrescine being the predominant type until the cotyledonary stage, and spermidine at the mature seed stage. IAA content increased continually from the globular stage reaching the maximum at the cotyledonary stage, followed by a decrease in the mature seed. The highest ABA level occurred at the globular stage, followed by a continuous reduction until stabilization at the pre-cotyledonary stage. Buffer-soluble protein and starch contents, and dry matter increased progressively during development, reaching their maximum values at the mature stage.
Similar content being viewed by others
References
von Arnold S., Egertsdotter U., Ekberg I., Gupta P.K., Mo H. and Nörgaard J. 1995. Somatic embryogenesis in Norway spruce (Picea abies). In: Jain S.M., Gupta P.K. and Newton R.J. (eds), Somatic Embryogenesis in Woody Plants, Vol. 3. Kluwer Academic Publishers, Dordrecht, pp. 17–36.
Astarita L.V., Floh E.I.S. and Handro W. 2003a. Changes in IAA, tryptophan and activity of soluble peroxidase associated with zygotic embryogenesis in Araucaria angustifolia (Brazilian pine). Plant Growth Regul. 39: 113–118.
Astarita L.V., Floh E.I.S. and Handro W. 2004. Free amino acids, protein and water content changes associated with seed development in Araucaria angustifolia. Biol. Plantarum 47: 53–59.
Astarita L.V., Handro W. and Floh E.I.S. 2003b. Changes in polyamines content associated with zygotic embryogenesis in the Brazilian pine, Araucaria angustifolia (Bert.). O. Ktze. Rev. Bras. Bot. 26: 163–168.
Attree S.M. and Fowke L.C. 1993. Embryogeny of gymnosperms: advances in synthetic seed technology of conifers. Plant Cell Tiss. Org. Cult. 35: 1–35.
Bais H.P. and Ravishankar G.A. 2002. Role of polyamines in the ontogeny of plants and their biotechnological applications. Plant Cell Tiss. Org. Cult. 69: 1–34.
Bandurski R.S., Cohen J.D., Slovin J.P. and Reinecke D.M. 1995. Hormone biosynthesis and metabolism. In: Davies P.J. (ed.), Plant Hormones. Kluwer Academic Publishers, Dordrecht, pp. 39–65.
Bewley J.D. and Black M. 1994. Seeds: Physiology of Development and Germination, 2nd Ed. Plenum Publishing, New York, 445 pp.
Bouchereau A., Aziz A., Larher F. and Martin-Tanguy J. 1999. Polyamines and environmental challenges: recent development. Plant Sci. 140: 103–125.
Bradford M.B. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Chiwocha S. and von Aderkas P. 2002. Endogenous levels of free and conjugated forms of auxin, cytokinins and abscisic acid during seed development in Douglas fir. Plant Growth Regul. 36: 191–200.
Coruzzi G. and Last R. 2000. Amino acids. In: Bicochemistry, Buchanan B.B., Gruissem W. and Jones R.L. (eds), Molecular Biology of Plants. American Society of Plant Physiologists, Rockville, pp. 358–410.
Feirer R.P. 1995. The biochemistry of conifer embryo development: amino acids, polyamines, and storage proteins. In: Jain S.M., Gupta P.K. and Newton R.J. (eds), Somatic Embryogenesis in Woody Plants, Vol. 1. Kluwer Academic Publishers, Dordrecht, pp. 317–336.
Gifford D.J. 1988. An electrophoretic analysis of the seed proteins form Pinus monticola and eight other species of pine. Can. J. Bot. 66: 1808–1812.
Groot S.P.C., Vanyperen I.I. and Karssen C.M. 1991. Strongly reduced levels of endogenous abscisic-acid in developing seeds of tomato mutant sitiens do not influence in vivo accumulation of dry-matter and storage proteins. Physiol. Plantarum 81: 73–78.
Guerra M.P., Silveira V., Santos A.L.W., Astarita L.V. and Nodari R.O. 2000. Somatic embryogenesis in Araucaria angustifolia (Bert) O. Kuntze. In: Jain S.M., Gupta P.K. and Newton R.J. (eds), Somatic Embryogenesis in Woody Plants, Vol. 6. Kluwer Academic Publishers, Dordrecht, pp. 457–478.
Hakman I. 1993. Embryology in Norway spruce (Picea abies). An analysis of the composition of seed storage proteins and deposition of storage reserves during seed development and somatic embryogenesis. Physiol. Plantarum 87: 148–159.
Kapik R.H., Dinus R.J. and Dean J.F.D. 1995. Abscisic acid and zygotic embryogenesis in Pinus taeda. Tree Physiol. 15: 485–490.
King J.E. and Gifford D.J. 1997. Amino acid utilization in seeds of loblolly pine during germination and early seedling growth. Plant Physiol. 113: 1125–1135.
Kojima K. 1996. Changes of abscisic acid, indole-3-acetic acid and gibberellin-like substances in the flowers and development fruilets of citrus cultivar 'Hyuganatsu'. Sci. Hort. 65: 263–272.
Kong L. and Yeung E.C. 1995. Effects of AgNO3 and PEG on white spruce (Picea glauca) somatic embryo maturation: enhancing cotyledonary embryo formation and endogenous ABA content. Physiol. Plantarum 93: 298–304.
Kong L., Attree S.M. and Fowke L.C. 1997. Changes of endogenous hormone levels in developing seeds, zygotic embryos and megagametophytes in Picea glauca. Physiol. Plantarum 101: 23–30.
Kong L., Attree S.M. and Fowke L.C. 1998. Effects of polyethylene glycol and methylglyoxal bis(guanylhydrazone) on endogenous polyamine levels and somatic embryo maturation in white spruce (Picea glauca). Plant Sci. 133: 211–220.
Lelu M.A., Bastien C., Drugeault A., Gouez M.L. and Klimaszewska K. 1999. Somatic embryogenesis and plantlet development in Pinus sylvestris and Pinus pinaster on medium with and without growth regulators. Physiol. Plantarum 105: 719–728.
McCready R.M., Guggolz J., Silviera V. and Owens H.S. 1950. Determination of starch and amylose in vegetables. Anal. Chem. 22: 1156–1158.
Merkle S.A., Parrot W.A. and Flinn B.S. 1995. Morphogenic aspects of somatic embryogenesis. In: Thope T.A. (ed.), In Vitro Embryogenesis in Plants. Kluwer Academic Publishers, Dordrecht, pp. 155–203.
Michalczuk L., Cooke T.D. and Cohen J.D. 1992. Regulation of indole-3-acetic acid biosynthetic pathways in carrot cell cultures. Plant Physiol. 100: 1346–1356.
Minocha R., Dale R.S., Cathie R., Steele K.D. and Minocha S.C. 1999. Polyamine levels during the development of zygotic and somatic embryos of Pinus radiata. Physiol. Plantarum 105: 155–164.
Neill S.J., Desikan R. and Hancock J. 2003. Nitric oxide signalling in plants. New Physiol. 159: 11–35.
Pullman G.S. and Buchanan M. 2003. Loblolly pine (Pinus taeda L.): stage-specific elemental analyses of zygotic embryo and female gametophyte tissue. Plant Sci. 164: 943–954.
Pullman G.S., Montello P., Cairney J., Xu N. and Feng X. 2003. Loblolly pine (Pinus taeda L.) somatic embryogenesis: maturation improvements by metal analyses of zygotic and somatic embryos. Plant Sci. 164: 955–969.
Richard D., Lescot M., Inzé D. and De Veylder L. 2002. Effect of auxin, cytokinin, and sucrose on cell cycle gene expression in Arabidopsis thaliana cell suspension cultures. Plant Cell Tiss. Org. Cult. 69: 167–176.
Rock C.D. and Quatrano R.S. 1995. The role of hormones during seed development. In: Davies P.J. (ed.), Plant Hormones. Kluwer Academic Publishers, Dordrecht, pp. 671–697.
dos Santos A.L.W., Silveira V., Steiner N., Vidor M. and Guerra M.P. 2002. Somatic embryogenesis in parana pine (Araucaria angustifolia (Bert.) O. Kuntze. Braz. Arch. Biol. Tec. 45: 97–106.
Shoeb F., Yadav J.S., Bajaj S. and Rajam M.V. 2001. Polyamines as biomarkers for plant regeneration capacity: improvement of regeneration by modulation of polyamine metabolism in different genotypes of Indica rice. Plant Sci. 160: 1229–1235.
Silveira V., Floh E.I.S., Handro W. and Guerra M.P. 2004. Effect of plant growth regulators on the cellular growth and levels of intracellular protein, starch and polyamines in embryogenic suspension cultures of Pinus taeda. Plant Cell Tiss. Org. Cult. 69: 233–249.
Silveira V., Steiner N., dos Santos A.L.W., Nodari R.O. and Guerra M.P. 2002. Biotechnology tolls in Araucaria angustifolia conservation and improvement: inductive factors affecting somatic embryogenesis. Crop Breeding Appl. Biotechnol. 3: 463–470.
Sokal R.R. and Rohlf F.J. 1995. Biometry. 3rd edn. Freeman and Co, New York.
Stals H. and Inzé D. 2001. When plant cells decide to divide. Trends Plant Sci. 8: 359–364.
Stone S.L. and Gifford D.J. 1997. Structural and biochemical changes in loblolly pine (Pinus taeda L.) seeds during germination and early seedling growth.I.Storage protein reserves. Int. J. Plant Sci. 158: 727–737.
Stone S.L. and Gifford D.J. 1999. Structural and biochemical changes in loblolly pine (Pinus taeda L.) seeds during germination and early seedling growth.I.Storage triacylglycerols and carbohydrates. Int. J. Plant Sci. 160: 663–671.
Todd C.D., Cooke J.E.K., Mullen R.T. and Gifford D.J. 2001. Regulation of loblolly pine (Pinus taeda L.)arginase in developing seedling tissue during germination and post-germinative growth. Plant Mol. Biol. 45: 555–565.
Todd C.D. and Gifford D.J. 2002. The role of the megagame-tophyte in maintaining loblolly pine (Pinus taeda L.) seedling arginase gene expression in vitro. Planta 215: 110–118.
Todd C.D. and Gifford D.J. 2003. Loblolly pine arginase responds to arginine in vitro. Planta 217: 610–615.
Tun N.N., Holk A. and Scherer G.F.E. 2001. Rapid increase of NO release in plant cell cultures induced by cytokinin. FEBS Lett. 509: 174–176.
Xu N., Coulter K.M. and Bewley J.D. 1990. Abscisic-acid and osmoticum prevent germination of developing alfalfa embryos, but only osmoticum maintains the synthesis of developmental proteins. Planta 1982: 382–390.
Wendehenne D., Pugin A., Klessig D.F. and Durner J. 2001. Nitric oxide: comparative synthesis and signaling in animal and plant cells. Trends Plant Sci. 6: 177–183.
Wise M.J. and Tunnacliffe A. 2004. POPP the question: what do LEA proteins do? Trends Plant Sci. 9: 13–17.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Silveira, V., Balbuena, T.S., Santa-Catarina, C. et al. Biochemical changes during seed development in Pinus taeda L.. Plant Growth Regulation 44, 147–156 (2004). https://doi.org/10.1023/B:GROW.0000049410.63154.ed
Issue Date:
DOI: https://doi.org/10.1023/B:GROW.0000049410.63154.ed