Abstract
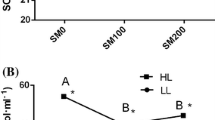
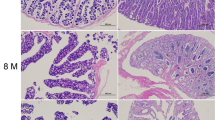
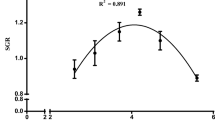
Selenium (Se), an essential component of deiodinases (DIOs), regulates the contents of thyroid hormones and thus improves animal growth. To explore the influences of selenium supplementation on fish growth metabolism, a total of 270 healthy grass carp (Ctenopharyngodon idella) were divided into three groups and feed three graded dietary selenium (0.141, 0.562, and 1.044 mg Se/kg) levels. The results showed that after 60-day feeding, dietary selenium improved the final body weight and specific growth rate (SGR) of grass carp. The hepatic DIO activities in selenium-supplemented groups were higher than those in control group. A significant increase in triiodothyronine (T3), free triiodothyronine (FT3), and thyroid-stimulating hormone (TSH) levels was accompanied by a decrease in the contents of thyroxine (T4) and free thyroxine (FT4) in selenium-supplemented groups. The histopathological observation of thyroid suggested that selenium deficiency resulted in hypertrophy of follicular epithelial cells. Moreover, the gene relative expression levels of dio1, dio2, and dio3 showed an increasing trend with the rising concentration of dietary selenium. The transcription levels of HPT axis-related genes (crh, tsh-β, ttr, tr-s, tpo, nis) and GH/IGF1-related genes (gh, ghr, igf1, igf1r) were significantly upregulated in selenium-supplemented groups. No significant differences in the above indicators were observed between 0.562 and 1.044 mg Se/kg diet group except T3 content and dio1 relative expression ratio. These results indicate that dietary selenium supplementation improves the hepatic DIO activities and thyroid hormone metabolism and regulates the transcription levels of HPT and GH/IGF axis-related genes, which may be responsible for the growth promotion in grass carp.








Similar content being viewed by others
Data availability
The datasets generated during the analysis in the current study are available from the corresponding author on reasonable request.
References
Abd El-Kader MF, Fath El-Bab AF, Abd-Elghany MF, Abdel-Warith AA, Younis EM, Dawood MAO (2020) Selenium nanoparticles act potentially on the growth performance, hemato-biochemical indices, antioxidative, and immune-related genes of European seabass (Dicentrarchus labrax). Biol Trace Elem Res 15
Arthur JR, Morrice PC, Beckett GJ (1988) Thyroid hormone concentrations in selenium deficient and selenium sufficient cattle. Res Vet Sci 45:122–123
Ashouri S, Keyvanshokooh S, Salati AP, Johari SA, Pasha-Zanoosi H (2015) Effects of different levels of dietary selenium nanoparticles on growth performance, muscle composition, blood biochemical profiles and antioxidant status of common carp (Cyprinus carpio). Aquaculture 446:25–29
Bang P, Ahlsén M, Berg U, Carlsson-Skwirut C (2001) Free insulin-like growth factor I: are we hunting a ghost. Horm Res 55:84–93
Behne D, Kyriakopoulos A, Gessner H, Walzog B, Meinhold H (1992) Type I iodothyronine deiodinase activity after high selenium intake, and relations between selenium and iodine metabolism in rats. J Nutr 122:1542–1546
Bermano G, Nicol F, Dyer JA, Sunde RA, Beckett GJ, Arthur JR, Hesketh JE (1995) Tissue-specific regulation of selenoenzyme gene expression during selenium deficiency in rats. Biochem J 311:425–430
Brown SB, Adams BA, Cyr DG, Eales JG (2004) Contaminant effects on the teleost fish thyroid. Environ Toxicol Chem 23:1680–1701
Chadio SE, Kotsampasi BM, Menegatos JG, Zervas GP, Kalogiannis DG (2006) Effect of selenium supplementation on thyroid hormone levels and selenoenzyme activities in growing lambs. Biol Trace Elem Res 109:145–154
Chen Q, Yu L, Yang L, Zhou B (2012) Bioconcentration and metabolism of decabromodiphenyl ether (BDE-209) result in thyroid endocrine disruption in zebrafish larvae. Aquat Toxicol 110–111:141–148
Chiamolera MI, Wondisford FE (2009) Minireview: thyrotropin-releasing hormone and the thyroid hormone feedback mechanism. Endocrinology 150:1091–1096
Dalir-Naghadeh B, Rezaei SA (2008) Assessment of serum thyroid hormone concentrations in lambs with selenium deficiency myopathy. Am J Vet Res 69:659–663
De Azevedo FM, Lanes CF, Almeida DV, Proietti MC, Marins LF (2007) The effect of GH overexpression on GHR and IGF-I gene regulation in different genotypes of GH-transgenic zebrafish. Comp Biochem Physiol Genom Proteonomics 2:228–233
De Groef B, Vander GS, Darras VM, Kühn ER (2006) Role of corticotropin-releasing hormone as a thyrotropin-releasing factor in non-mammalian vertebrates. Gen Comp Endocrinol 146:62–68
Dhingra S, Bansal MP (2006) Hypercholesterolemia and tissue-specific differential mRNA expression of type-1 5’-iodothyronine deiodinase under different selenium status in rats. Biol Res 39:307–319
Dhingra S, Singh U, Bansal MP (2003) Protective role of selenium status on T3/T4 kinetics in rats under hyperlipidemia. Indian J Biochem Biophys 40:260–264
Duan C (1988) Nutritional and developmental regulation of insulin-like growth factors in fish. J Nutr 128:306S-314S
Dunn JT, Dunn AD (2001) Update on intrathyroidal iodine metabolism. Thyroid 11:407–414
Eales JG, Brown SB (1993) Measurement and regulation of thyroidal status in teleost fish. Rev Fish Biol Fisher 3:299–347
Farchi-Pisanty O, Hackett PB, Moav B (1995) Regulation of fish growth hormone transcription. Mol Mar Biol Biotechnol 4:215–223
Gaber MM (2009) Efficiency of selenium ion inclusion into common carp (Cyprinus carpio L.) diets. Afr J Agric Res 4:348–353
Gahete MD, Córdoba-Chacón J, Lantvit DD, Ortega-Salas R, Sanchez-Sanchez R, Pérez-Jiménez F, López-Miranda J, Swanson SM, Castaño JP, Luque RM, Kineman RD (2014) Elevated GH/IGF-I promotes mammary tumors in high-fat, but not low-fat, fed mice. Carcinogenesis 35:2467–2473
Gao Y, Liu Z, Jia D, Hu Q, Li L, Tang R, Li D (2020) Acute microcystin-LR exposure interfere thyroid hormones homeostasis in adult zebrafish (Danio rerio). Chemosphere 243:125258
Golstein J, Corvilain B, Lamy F, Paquer D, Dumont JE (1988) Effects of a selenium deficient diet on thyroid function of normal and perchlorate treated rats. Acta Endocrinol 118:495–502
Guo H, Lin W, Hou J, Wang L, Zhang D, Wu X, Li D (2018) The protective roles of dietary selenium yeast and tea polyphenols on growth performance and ammonia tolerance of juvenile Wuchang bream (Megalobrama amblycephala). Front Physiol 9:1371
Han D, Xie S, Liu M, Xiao X, Liu H, Zhu X, Yang Y (2011) The effects of dietary selenium on growth performances, oxidative stress and tissue selenium concentration of gibel carp (Carassius auratus gibelio). Aquacult Nutr 17:e741–e749
Holt RI, Erotokritou-Mulligan I, Guha N, Stow M, Bassett E, Bartlett C, Sonksen PH (2012) An overview of the GH-2004 project: catching the growth hormone cheats. Growth Horm IGF Research 22:S19
Hooper SE, Backus R, Amelon S (2018) Effects of dietary selenium and moisture on the physical activity and thyroid axis of cats. J Anim Physiol Anim Nutr 102:495–504
Pérez-Sánchez J (2000) The involvement of growth hormone in growth regulation, energy homeostasis and immune function in the gilthead sea bream (Sparus aurata): a short review. Fish Physiol Biochem 22(2):135–144
Kawakami Y, Seoka M, Miyashita S, Kumai H, Ohta H (2006) Characterization of transthyretin in the Pacific bluefin tuna. Thunnus Orientalis Zoolog Sci 23(5):443–448
Khan KU, Zuberi A, Nazir S, Ullah I, Jamil Z, Sarwar H (2017a) Synergistic effects of dietary nano selenium and vitamin C on growth, feeding, and physiological parameters of mahseer fish (Tor putitora). Aquacult Rep 5:70–75
Khan KU, Zuberi A, Fernandes JBK, Ullah I, Sarwar H (2017b) An overview of the ongoing insights in selenium research and its role in fish nutrition and fish health. Fish Physiol Biochem 43:1689–1705
Kitajima T, Chiba Y (2013) Selenomethionine metabolism and its toxicity in yeast. Biomol Concepts 4:611–616
Köhrle J (2000) The deiodinase family: selenoenzymes regulating thyroid hormone availability and action. Cell Mol Life Sci 57:1853–1863
Köhrle J, Gärtner R (2009) Selenium and thyroid. Best Pract Res Cl En 23:815–827
Lin S, Wang C, Tan S, Liang Y, Yao H, Zhang Z, Xu S (2014) Selenium deficiency inhibits the conversion of thyroidal thyroxine (T4) to triiodothyronine (T3) in chicken thyroids. Biol Trace Elem Res 161:263–271
Liu Y, Wang J, Fang X, Zhang H, Dai J (2011) The thyroid-disrupting effects of long-term perfluorononanoate exposure on zebrafish (Danio rerio). Ecotoxicology 20:47–55
Liu Y, Lo L, Chan W (2000) Temporal expression and T3 induction of thyroid hormone receptors alpha1 and beta1 during early embryonic and larval development in zebrafish (Danio rerio). Mol Cell Endocrinol 159:187–195
Liu Z, Li D, Hu Q, Tang R, Li L (2016) Effects of exposure to microcystin-LR at environmentally relevant concentrations on the metabolism of thyroid hormones in adult zebrafish (Danio rerio). Toxicon 124:15–25
Liu Z, Li D, Wang Y, Guo W, Gao Y, Tang R (2015a) Waterborne exposure to microcystin-LR causes thyroid hormone metabolism disturbances in juvenile Chinese rare minnow (Gobiocypris rarus). Environ Toxicol Chem 34:2033–2040
Liu Z, Tang R, Li D, Hu Q, Wang Y (2015b) Subacute microcystin-LR exposure alters the metabolism of thyroid hormones in juvenile zebrafish (Danio Rerio). Toxins 7:337–352
Liu Z, Tang R, Yin X, Tong N, Li D (2015c) Microcystin-LR alters the gene transcription and activities of iodothyronine deiodinases in the hepatic cells of grass carp (Ctenopharyngodon idella). J Biochem Mol Toxicol 29:305–310
Liu L, Liang X, Li J, Fang J, Alam M (2018) Effects of dietary selenium on growth performance and oxidative stress in juvenile grass carp (Ctenopharyngodon idellus). Aquacult Nutr 24
Livak K, Schmittgen T (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Manchado M, Infante C, Asensio E, Planas J, Cañavate J (2008) Thyroid hormones down-regulate thyrotropin beta subunit and thyroglobulin during metamorphosis in the flatfish Senegalese sole (Solea senegalensis Kaup). Gen Comp Endocrinol 155:447–455
Meinhold H, Campos-Barros A, Walzog B, Köhler R, Müller F, Behne D (1993) Effects of selenium and iodine deficiency on type I, type II and type III iodothyronine deiodinases and circulating thyroid hormones in the rat. Exp Clin Endocrinol 101:87–93
Moav B, McKeown B (1992) Thyroid hormone increases transcription of growth hormone mRNA in rainbow trout pituitary. Horm Metab Res 24:10–14
Morgado I, Santos C, Jacinto R, Power D (2007) Regulation of transthyretin by thyroid hormones in fish. Gen Comp Endocrinol 152:189–197
Mousa R, Notis D, Metanis N (2017) Selenium and selenocysteine in protein chemistry. Angew Chem Int Ed Engl 56:15818–15827
Müller E, Locatelli V, Cocchi D (1999) Neuroendocrine control of growth hormone secretion. Physiol Rev 79:511–607
Naderi M, Mousavi S, Safahieh A, Ghatrami E, Zargham D (2014) Effects of 4-nonylphenol on balance of steroid and thyroid hormones in sexually immature male yellowfin seabream (Acanthopagrus latus). Environ Toxicol 29:459–465
Orozco A, Valverde-R C (2005) Thyroid hormone deiodination in fish. Thyroid 15:799–813
Patino R, Wainscott M, Cruz-Li E, Balakrishnan S, McMurry C, Blazer V, Anderson T (2003) Effects of ammonium perchlorate on the reproductive performance and thyroid follicle histology of zebrafish. Environ Toxicol Chem 22:1115–1121
Power D, Llewellyn L, Faustino M, Nowell M, Björnsson BT, Einarsdottir I, Sweeney G (2001) Thyroid hormones in growth and development of fish. Comp Biochem Physiol C Toxicol Pharmacol 130:447–459
Rayman MP (2000) The importance of selenium to human health. Lancet 356:233–241
Rayman MP (2012) Selenium and human health. Nutr Rev 379:1256–1268
Reinecke M (2010) Influences of the environment on the endocrine and paracrine fish growth hormone-insulin-like growth factor-I system. J Fish Biol 76:1233–1254
Ribeiro AR, Ribeiro L, Sæle O, Dinis MT, Moren M (2012a) Iodine and selenium supplementation increased survival and changed thyroid hormone status in Senegalese sole (Solea senegalensis) larvae reared in a recirculation system. Fish Physiol Biochem 38:725–734
Ribeiro AR, Ribeiro L, Saele Ø, Hamre K, Dinis MT, Moren M (2012b) Selenium supplementation changes glutathione peroxidase activity and thyroid hormone production in Senegalese sole (Solea senegalensis) larvae. Aqua Nutr 18:559–567
Rider SA, Davies SJ, Jha AN, Fisher AA, Knight J, Sweetman JW (2009) Supra-nutritional dietary intake of selenite and selenium yeast in normal and stressed rainbow trout (Oncorhynchus mykiss): implications on selenium status and health responses. Aquaculture 295:282–291
Sebastián EA, Alicia F, María JM, Gabriel BL, Jaume P, Björn TB, Silvia Z, Manuel C (2000) Long-term feeding of a maintenance ration affects the release of Igf-1 and leptin, and delays maturation in a male teleost fish (Dicentrarchus labrax L). Aquaculture 527:735467
Shamblott MJ, Cheng CM, Bolt D, Chen TT (1995) Appearance of insulin-like growth factor mRNA in the liver and pyloric ceca of a teleost in response to exogenous growth hormone. Proc Natl Acad Sci 92:6943–6946
Simó-Mirabet P, Perera E, Calduch-Giner JA, Afonso JM, Pérez-Sánchez J (2018) Co-expression analysis of sirtuins and related metabolic biomarkers in juveniles of gilthead sea bream (Sparus aurata) with differences in growth performance. Front Physiol 9:608
Shi X, Liu C, Wu G, Zhou B (2009) Waterborne exposure to PFOS causes disruption of the hypothalamus-pituitary-thyroid axis in zebrafish larvae. Chemosphere 77:1010–1018
Stéphanie, Fontagné-Dicharry, Vincent Véron, Larroquet L, Godin S, Kaushik SJ (2020) Effect of selenium sources in plant-based diets on antioxidant status and oxidative stress-related parameters in rainbow trout juveniles under chronic stress exposure. Aquaculture 735684
Sun CF, Tao Y, Jiang XY, Zou SM (2011) IGF binding protein 1 is correlated with hypoxia-induced growth reduce and developmental defects in grass carp (Ctenopharyngodon idellus) embryos. Gen Comp Endocrinol 172:409–415
Triggiani V, Tafaro E, Giagulli VA, Sabbà C, Resta F, Licchelli B, Guastamacchia E (2009) Role of iodine, selenium and other micronutrients in thyroid function and disorders. Endocr Metab Immune Disord Drug Targets 9:277–294
Valcavi R, Zini M, Portioli I (1992) Thyroid hormones and growth hormone secretion. J Endocrinol Invest 15:313–330
Walpita CN, Grommen SV, Darras VM, Vander GS (2007) The influence of stress on thyroid hormone production and peripheral deiodination in the Nile tilapia (Oreochromis niloticus). Gen Comp Endocrinol 150:18–25
Xiao C, Liu Z, Li D, Refaey MM, Tang R, Li L, Zhang X (2017) Acute nitrite exposure alters the metabolism of thyroid hormones in grass carp (Ctenopharyngodon idellus). Chemosphere 186:974–982
Xie L, Chen S, Yao C, Li D, Li L, Tang R (2019) Nitrite induces endoplasmic reticulum stress and associates apoptosis of liver cells in grass carp (Ctenopharyngodon idella). Aquaculture
Xie L, Yan W, Li J, Yu L, Wang J, Li G, Steinman AD (2015) Microcystin-RR exposure results in growth impairment by disrupting thyroid endocrine in zebrafish larvae. Aquat Toxicol 164:16–22
Yan W, Zhou Y, Yang J, Li S, Hu D, Wang J, Chen J, Li G (2012) Waterborne exposure to microcystin-LR alters thyroid hormone levels and gene transcription in the hypothalamic-pituitary-thyroid axis in zebrafish larvae. Chemosphere 87:1301–1307
Yang X, Xie J, Wu T, Yue G, Chen J, Zhao R (2007) Hepatic and muscle expression of thyroid hormone receptors in association with body and muscle growth in large yellow croaker (Pseudosciaena crocea). Gen Comp Endocrinol 151:163–171
Yoshiura Y, Sohn YC, Munakata A, Kobayashi M, Aida K (1999) Molecular cloning of the cdna encoding the β subunit of thyrotropin and regulation of its gene expression by thyroid hormones in the goldfish (Carassius auratus). Fish Physiol Biochem 21:201–210
Yu L, Chen M, Liu Y, Gui W, Zhu G (2013) Thyroid endocrine disruption in zebrafish larvae following exposure to hexaconazole and tebuconazole. Aquat Toxicol 138–139:35–42
Yu L, Deng J, Shi X, Liu C, Yu K, Zhou B (2010) Exposure to DE-71 alters thyroid hormone levels and gene transcription in the hypothalamic-pituitary-thyroid axis of zebrafish larvae. Aquat Toxicol 97:226–233
Zeng X, Sun H, Huang Y, Liu J, Yu L, Liu C, Wang J (2018) Effects of environmentally relevant concentrations of tris (2-butoxyethyl) phosphate on growth and transcription of genes involved in the GH/IGF and HPT axes in zebrafish (Danio rerio). Chemosphere 212:376–384
Zhang X, Tian H, Wang W, Ru S (2013) Exposure to monocrotophos pesticide causes disruption of the hypothalamic-pituitary-thyroid axis in adult male goldfish (Carassius auratus). Gen Comp Endocrinol 193:158–166
Zhao H, Xia J, Zhang X, He X, Li L, Tang R, Li D (2018) Diet affects muscle quality and growth traits of grass carp (Ctenopharyngodon idellus): a comparison between grass and artificial feed. Front Physiol 9:283
Zheng L, Feng L, Jiang WD, Wu P, Tang L, Kuang SY, Zeng YY, Zhou XQ, Liu Y (2018) Selenium deficiency impaired immune function of the immune organs in young grass carp (Ctenopharyngodon idella). Fish Shellfish Immunol 77:53–70
Acknowledgements
This research was supported by the National Key R&D Program of China (No. 2019YFD0900301), the Earmarked Fund for China Agriculture Research System (CARS-45-24), and the National Natural Science Foundation (No.31502140).
Author information
Authors and Affiliations
Contributions
PinMa, Zhenyi Hu, and RongTang: concept and design of the experiments and wrote the manuscript. LiLi and Dapeng Li: edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The experimental procedure used in this study had been approved by the Institutional Animal Care and Use Committee (IACUC) of Huazhong Agricultural University, China.
Competing interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Ma, P., Hu, Z., Li, L. et al. Dietary selenium promotes the growth performance through growth hormone–insulin-like growth factor and hypothalamic–pituitary–thyroid axes in grass carp (Ctenopharyngodon idella). Fish Physiol Biochem 47, 1313–1327 (2021). https://doi.org/10.1007/s10695-021-00974-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-021-00974-1