Abstract
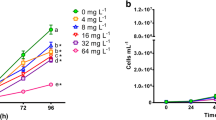
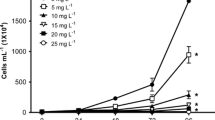
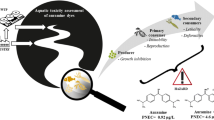
Aquatic pollution caused by dyes has increased together with the growth of activities using colorants such as the textile, leather, food, and agrochemicals industries. Because most popular azo dyes are synthesized from benzidine, a carcinogenic compound, a threat to aquatic biota could be expected. The use of single species for toxicity assessment provides limited data, so a battery of test organisms, including representatives of different trophic levels such as algae, zooplankters, and fish, could undoubtedly provide more information. Therefore, our study was aimed at evaluating the toxic effect of the azo dye Direct blue 15 (DB15) on a battery of bioassays using a primary producer (Pseudokirchneriella subcapitata), a primary consumer (Ceriodaphnia dubia), and a secondary consumer (Danio rerio). P. subcapitata was more sensitive to DB15 (IC50 = 15.99 mg L−1) than C. dubia (LC50: 450 mg L−1). In the algae exposed to DB15, chlorophyll-a and -b were significantly increased, and carotenoids were reduced. The concentrations of protein, carbohydrates, and lipids per cell in P. subcapitata exposed to all DB15 concentrations were significantly higher than that measured in control. At 25 mg L−1 of DB15, survival, total progeny, and the number of released clutches were significantly decreased, and the start of reproduction was delayed in C. dubia. DB15 did not induce lethal or sublethal effects in D. rerio embryos at any of the tested concentrations from 24 to 72 h post-fertilization (hpf), but from 96 to 144 hpf, the larvae exposed to 100 and 500 mg L−1 developed yolk sac edema, curved tail, and skeletal deformations. After 144 hpf, DB15 produced a significant increase in embryos without a heartbeat, as the concentration of dye raised. The textile-used, azo dye DB15, caused toxic effects of different magnitude on microalgae, cladocerans, and zebrafish embryos; for this reason, the discharge of this colorant into waterbodies should be regulated to prevent environmental impacts.






Similar content being viewed by others
References
Abe FR, Mendonca JN, Moraes LAB, de Oliveira GAR, Gravato C, Soares AMVM, de Oliveira DP (2017) Toxicological and behavioral responses as a tool to assess the effects of natural synthetic dyes on zebrafish early life. Chemosphere 178:282–290. https://doi.org/10.1016/j.chemosphere.2017.03.030
Bae JS, Freeman HS (2007) Aquatic toxicity evaluation of new direct dyes to the Daphnia magna. Dyes Pigments 73:81–85. https://doi.org/10.1016/j.dyepig.2005.10.015
Bafana A, Sivanesan SD, Tapan C (2011) Azo dyes: past, present and the future. Environ Rev 19:350–371. https://doi.org/10.1139/a11-018
Balapure K, Bhatt N, Madamwar D (2015) Mineralization of reactive azo dyes present in simulated textile waste water using down flow microaerophilic fixed film bioreactor. Bioresour Technol 175:1–7. https://doi.org/10.1016/j.biortech.2014.10.040
Blaise C, Férard JF (2005) Small-scale freshwater toxicity investigations. vol. 1–Toxicity test methods. Springer, Berlin, Germany, p 422
Busquet F, Strecker R, Rawlings JM, Belanger SE, Braunbeck T, Carr GJ, Cenijn P, Fochtman P, Gourmelon A, Hübler N, Kleensang A, Knöbel M, Kussatz C, Legler J, Lillicrap A, Martínez-Jerónimo F, Polleichtner C, Rzodeczko H, Salinas E, Schneider KE, Scholz S, van den Brandhof EJ, van der Ven LT, Walter-Rohde S, Weigt S, Witters H, Halder M (2014) OECD validation study to assess intra- and inter-laboratory reproducibility of the zebrafish embryo toxicity test for acute aquatic toxicity testing. Regul Toxicol Pharmacol 69(3):496–511. https://doi.org/10.1016/j.yrtph.2014.05.018
Chia MA, Musa RI (2014) Effect of indigo dye effluent on the growth, biomass, production and phenotypic plasticity of Scenedesmus quadricauda (Chlorococcales). Acad Bras Cienc 86(1):419–428. https://doi.org/10.1590/0001-3765201420130225
Copaciu F, Opris O, Coman V, Ristoiu D, Niinemets Ü, Copolovici L (2013) Diffuse water pollution by anthraquinone and azo dyes in environment importantly alters foliage volatiles, carotenoids and physiology in wheat (Triticum aestivum). Water Air Soil Pollut 224:1478. https://doi.org/10.1007/s11270-013-1478-4
Croce R, Ciná F, Lombardo A, Crispeyn G, Cappelli CI, Vian M, Maiorana S, Benfenati E, Baderna D (2017) Aquatic toxicity of several textile dye formulations: acute and chronic assays with Daphnia magna and Raphidocelis subcapitata. Ecotoxicol Environ Saf 144:79–87. https://doi.org/10.1016/j.ecoenv.2017.05.046
D’Alessandro EB, Antoniosi Filho NR (2016) Concepts and studies on lipid and pigments of microalgae: a review. Renew Sust Energ Rev 58:832–841
de Luna LA, Silva TH, Nogueira RF, Kummrow F, Umbuzeiro GA (2014) Aquatic toxicity of dyes before and after photo-Fenton treatment. J Hazard Mater 276:332–338. https://doi.org/10.1016/j.jhazmat.2014.05.047
Du M, Zhang D, Yan C, Zhang X (2012) Developmental toxicity evaluation of three hexabromocyclododecane diastereoisomers on zebrafish embryos. Aquat Toxicol 112–113:1–10
Dubois M, Gilles K, Hamilton JK, Rebers PA, Smith F (1951) A colorimetric method for the determination of sugars. Nature 168(4265):167. https://doi.org/10.1038/168167a0
Einicker-Lamas M, Mezian GA, Fernandes TB, Silva FLS, Guerra F, Miranda K, Attias M, Oliveira MM (2002) Euglena gracilis as a model for the study of Cu2+ and Zn2+ toxicity and accumulation in eukaryotic cells. Environ Pollut 120:779–786. https://doi.org/10.1016/S0269-7491(02)00170-7
Environment Canada and Health Canada (2009) Screening assessment for the challenge: five disazo dyes
Environment Canada and Health Canada (2013a) Draft screening assessment-aromatic azo and benzidine-based substance grouping-certain azo disperse dyes
Environment Canada and Health Canada (2013b) Draft screening assessment-aromatic azo and benzidine-based substance grouping- certain azo solvent dyes
Environmental Hazard and Risk Assessment of Organic Colorants (ETAD) (1992) Including life-cycle analysis. Materials for ETAD seminar, May 22, Denmark
Ferraz ER, Umbuzeiro GA, de-Almeida G, Caloto-Oliveira A, Chequer FM, Zanoni MV, Oliveira DP (2011) Differential toxicity of Disperse Red 1 and Disperse Red 13 in the Ames test, HepG2 cytotoxicity assay, and Daphnia acute toxicity test. Environ Toxicol 26(5):489–497. https://doi.org/10.1002/tox.20576.
George B, Pancha I, Desai C, Choskshi K, Paliwal C, Ghosh T, Mishra S (2014) Effects of different media composition, light intensity and photoperiod on morphology and physiology of freshwater microalgae Ankistrodesmus falcatus –a potential strain for bio-fuel production. Bioresour Technol 171:367–374. https://doi.org/10.1016/j.biortech.2014.08.086
Gita S, Shukla SP, Saharan N, Prakash C, Deshmukhe G (2019) Toxic Effects of selected textile dyes on elemental composition, photosynthetic pigments, protein content and growth of a freshwater chlorophycean alga Chlorella vulgaris. Bull Environ Contam Toxicol 102:795–801. https://doi.org/10.1007/s00128-019-02599-w
Golka K, Kopps S, Myslak WZ (2004) Carcinogenicity of azo colorants: influence of solubility and bioavailability. Toxicol Lett 151(1):203–210. https://doi.org/10.1016/j.toxlet.2003.11.016
Greene JC, Baughman GL (1996) Effects of 46 dyes on population growth of freshwater green alga Selenastrum capricornutum. Text Chem Color 28(4):23–30
Herbomel P, Thisse B, Thisse C (1999) Ontogeny and behavior of early macrophages in the zebrafish embryo. Development 126:3735–3745
Hernández-Zamora M, Perales-Vela HV, Flores-Ortiz CM, Cañizares- Villanueva RO (2014) Physiological and biochemical responses of Chlorella vulgaris to Congo Red. Ecotoxicol Environ Saf 108:72–77. https://doi.org/10.1016/j.ecoenv.2014.05.030
Hernández-Zamora M, Martínez-Jerónimo F, Cristiani-Urbina E, Cañizares-Villanueva RO (2016) Congo red dye affects survival and reproduction in the cladoceran Ceriodaphnia dubia. Effects of direct and dietary exposure. Ecotoxicology 25(10):1832–1840. https://doi.org/10.1007/s10646-016-1731-x
Hernández-Zamora M, Martínez-Jerónimo F (2019) Congo red diversely affects organisms of different trophic levels: a comparative study with microalgae, cladocerans and zebrafish embryos. Environ Sci Pollut R 26:11743–11755. https://doi.org/10.1007/s11356-019-04589-1
Hill AJ, Teraoka H, Heideman W, Peterson RE (2005) Zebrafish as a model vertebrate for investigating chemical toxicity. Toxicol Sci 86(1):6–19. https://doi.org/10.1093/toxsci/kfi110
ISO. International Standard Organization (1996) Water quality – Determination of the inhibition of the mobility of Daphnia magna Straus (Cladocera, Crustacea) – Acute toxicity test. ISO 6341, 3rd Ed
Jang GH, Park IS, Lee SH, Huh TL, Lee YM (2009) Malachite green induces cardiovascular defects in developing zebrafish (Danio rerio) embryos by blocking VEGFR-2 signaling. Biochem Biophys Res Commun 382(3):486–491. https://doi.org/10.1016/j.bbrc.2009.01.118
Jena S, Acharya S, Mohapatra PK (2012) Variation in effects of four OP insecticides on photosynthetic pigment fluorescence of Chlorella Vulgaris Beij. Ecotox Environ Saf 80:111–117. https://doi.org/10.1016/j.ecoenv.2012.02.016
Joshi V, Katti P (2018) Developmental toxicity assay for food additive tartrazine using Zebrafish (Danio rerio) embryo cultures. Int J Toxicol 37(1):38–44. https://doi.org/10.1177/1091581817735227
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203:253–310. https://doi.org/10.1002/aja.1002030302
Lammer E, Carr GJ, Wendler K, Rawlings JM, Belanger SE, Braunbeck T (2009) Is the fish embryo toxicity test (FET) with the zebrafish (Danio rerio) a potential alternative for the fish acute toxicity test. Comp Biochem Physiol C 149:196–209. https://doi.org/10.1016/j.cbpc.2008.11.006
Levy JL, Stauber JL, Jolley DF (2007) Sensitivity of marine microalgae to copper: the effect of biotic factors on copper adsorption and toxicity. Sci Total Environ 387(1–3):141–154. https://doi.org/10.1016/j.scitotenv.2007.07.016
Lim SL, Chu WL, Phang SM (2010) Use of Chlorella vulgaris for bioremediation of Textile Wastewater. Bioresour Technol 101(19):7314–7322. https://doi.org/10.1016/j.biortech.2010.04.092
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Maguire RJ, Tkacz RJ (1991) Occurrence of dyes in the Yamaska River, Quebec. Water Pollut Res J Can New Ser 26(2):145–161
Mahalakshmi S, Lakshmi D, Menaga U (2015) Biodegradation of different concentration of dye (Congo red dye) by using green and blue green algae. Int J Environ Res 9(2):735–744
Malik, NH, Zain, H, Ali, N (2018) Organismic-level acute toxicology profiling of reactive azo dyes. Environ. Monit. Asess. 190–612. https://doi.org/10.1007/s10661-018-6986-7
Manjunatha B, Wei-bing P, Ke-chun L, Marigoudar AR, Xi-Qiang C, Xi-min W, Xue W (2014) The effects of henna (hair dye) on the embryonic development of zebrafish (Danio rerio). Environ Sci Pollut Res 21:10361–10367. https://doi.org/10.1007/s11356-014-2968-7
Martínez-Ruiz EB, Martínez-Jerónimo F (2015) Nickel has biochemical, physiological, and structural effects on the Green microalga Ankistrodesmus falcatus: an integrative study. Aquat Toxicol 169:27–36
Meireles G, Daam MA, Sanches ALM, Zanoni MVB, Soares AMVM, Gravato C, Oliveira DP (2018) Red disperse dyes (DR 60, DR 73 and DR 78) at environmentally realistic concentrations impact biochemical profile of early life stages of zebrafish (Danio rerio) Chem Biol Interact 292:94–100. https://doi.org/10.1016/j.cbi.2018.07.007
Ministry of Environment and Forest (1997) Government of India, Gazette on 26th March
Mohamed NA (1994) Application of algal ponds for wastewater treatment and algal production. M.Sc. Thesis, Faculty of Science, Cairo University, Bani-Sweef Branch
Novotný N, Dias A, Kapanen K, Malachová M, Vándrovcová M, Itävaara N, Lima N (2006) Comparative use of bacterial, algal and protozoan tests to study toxicity of azo and anthraquinone dyes. Chemosphere 63:1436–1442. https://doi.org/10.1016/j.chemosphere.2005.10.002
Oliveira GAR, Leme DM, de LaPuente J, Brito LB, Porredón C, Rodrigues LB, Brull N, Serret JT, Borrás M, Disner GR, Cestari MM, Oliveira DP (2018) A test battery for assessing the ecotoxic effects of textile dyes. Chem Biol Interact 291:171–179. https://doi.org/10.1016/j.cbi.2018.06.026
Organisation for Economic Co-operation and Development OECD (2004) Test No. 202: Daphnia sp. acute immobilisation test. OECD guidelines for the testing of chemicals (Section 2). OECD Publishing, Paris
Organisation for Economic Co-operation and Development OECD (2011) Test No. 201: freshwater alga and cyanobacteria, growth inhibition test. OECD guidelines for the testing of chemicals, Section 2. OECD Publishing, Paris
Organisation for Economic Co-operation and Development OECD (2013) Test no. 236: fish embryo acute toxicity (FET) test. OECD guidelines for the testing of chemicals. Paris, France
Øllgaard, H, Frost, L, Galster, J, Hansen, OC (1998) Survey of azo-colorants in Denmark: consumption, use, health and environmental aspects. Danish Environmental Protection Agency, Denmark
Parrott JL, Bartlett AJ, Balakrishnan VK (2016) Chronic toxicity of azo and anthracenedione dyes to embryo-larval fathead minnow. Environ Pollut 210:40–47
Puvaneswari N, Muthukrishnan J, Gunasekaran P (2006) Toxicity assessment and microbial degradation of azo dyes. Indian J Exp Biol 44(8):618–626
Rehman R, Mahmud T, Ejaz R, Rauf A, Mitu L (2017) Sorptive removal of Direct blue-15 dye from water using Camellia sinensis and Carica papaya leaves. Bulg Chem Comm 49(1):20–25
Ruyters G (1984) Effects of blue light on enzymes. Blue light effects in biological systems. Proc Life Sci 283–301
Rocha OP, Cesila CA, Christovam EM, Barros SB, Zanoni MV, de Oliveira DP (2017) Ecotoxicological risk assessment of the “Acid Black 210” dye. Toxicology 376:113–119. https://doi.org/10.1016/j.tox.2016.04.002
Rocha GS, Parrish CC, Lombardi AT, da GG, Melão M (2016) Copper affects biochemical and physiological responses of Selenastrum gracile (Reinsch). Ecotoxicology 25(8):1468–1477. https://doi.org/10.1007/s10646-016-1698-7
Rodrigues de Oliveira GA, de Lapuente J, Teixidó E, Porredón C, Borràs M, de Oliveira DP (2016) Textile dyes induce toxicity on zebrafish early life stages. Environ Toxicol Chem 35(2):429–434. https://doi.org/10.1002/etc.3202
Roy R, Fakhruddin ANM, Khatun R, Islam MS, Ahsan MA, Neger AJMT (2010) Characterization of textile industrial effluents and its effects on aquatic macrophytes and algae. Bangladesh J Sci Ind Res 45(1):79–84. https://doi.org/10.3329/bjsir.v45i1.5187
SCCNFP (2002) Opinion concerning the safety review of the use of certain azo-dyes in cosmetic products according to the cosmetic directive 76/768/EFC. Opinion expressed at the 19th plenary meeting
Scholz S, Fischer S, Gündel U, Küster E, Luckenbach T, Voelker D(2008) The zebrafish embryo model in environmental risk assessment—applications beyond acute toxicity testing Environ Sci Pollut Res Int 15:394–404. https://doi.org/10.1007/s11356-008-0018-z
Schroder GD, Rosslewandowski S, Davis EM (1991) Evaluation of the toxic effects of selected municipal wastewater effluents on aquatic invertebrates. Environ Technol 12:757–768. https://doi.org/10.1080/09593339109385067
Shan A, Harbin D (1988) Analyses of two azo dyes by high-performance liquid chromatography. J Chromatogr Sci 26:439–442
Seo YH, Lee Y, Jeon DY, Han JI (2015) Enhancing the light utilization efficiency of microalgae using organic dyes. Bioresour Technol 181:355–359. https://doi.org/10.1016/j.biortech.2015.01.031
Souza JP, Melo DC, Lombardi AT, Melão MG (2014) Effects of diet-borne cadmium on life history and secondary production of a tropical freshwater cladoceran. Ecotoxicology 23(9):1764–1773. https://doi.org/10.1007/s10646-014-1341-4
Stein JR (1973) Handbook of phycological methods. Culture methods and growth measurements. Cambridge University Press, London, p 7–24
Tigini V, Giansanti P, Mangiavillano A, Pannocchia A, Varese GC (2011) Evaluation of toxicity,genotoxicity and environmental risk of simulated textile and tannery wastewaters with a battery of biotests. Ecotoxicol Environ Saf 74(4):866–873. https://doi.org/10.1016/j.ecoenv.2010.12.001
Tsuruwaka Y, Konishi M, Shimada E (2015) Loss of wwox expression in zebrafish embryos causes edema and alters Ca2+ dynamics. PeerJ 3:1–12. https://doi.org/10.7717/peerj.727
U.S. Environmental Protection Agency (2002) Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms. EPA-821-R-02-012. 5th edn, Office of Research and Development, Cincinnati, OH
Umbuzeiro G, de A, Freeman HS, Warren SH, De Oliveira DP, Terao Y, Watanabe T, Claxton LD (2005) The contribution of azo dyes to the mutagenic activity of the Cristais River. Chemosphere 60:55–64. https://doi.org/10.1016/j.chemosphere.2004.11.100
Vacchi FI, Von der Ohe PC, Albuquerque AF, Vendemiatti JA, Azevedo CC, Honório JG, Silva BF, Zanoni MV, Henry TB, Nogueira AJ, Umbuzeiro GdeA (2016) Occurrence and risk assessment of an azo dye—the case of Disperse Red 1. Chemosphere 156:95–100. https://doi.org/10.1016/j.chemosphere.2016.04.121
Villegas-Navarro A, Romero-González MC, Rosas-López E, Domínguez-Aguilar R, Sachetin Marcal W (1999) Evaluation of Daphnia magna as an indicator of toxicity and treatment efficacy of textile wastewaters. Environ Int 25(5):619–624. https://doi.org/10.1016/S0160-4120(99)00034-3
Vimonses V, Lei S, Jin B, Chow CWK, Saint C (2009) Kinetic study and equilibrium isotherm analysis of Congo Red adsorption by clay materials. Chem Eng J 148:354–364. https://doi.org/10.1016/j.cej.2008.09.009
Walthall WK, Stark JD (1999) The acute and chronic toxicity of two xanthene dyes, fluorescein sodium salt and phloxine B, to Daphnia pulex. Environ Pollut 104:207–215. https://doi.org/10.1016/S0269-7491(98)00189-4
Wellburn AR (1994) The spectral determination of chlorophyll a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313. https://doi.org/10.1016/S0176-1617(11)81192-2
Wen YZ, Chen H, Shen CS, Zhao MR, Liu WP (2011) Enantio selectivity tuning of chiral herbicide dichlorprop by copper: roles of reactive oxygen species. Environ Sci Technol 45:4778–4784. https://doi.org/10.1021/es2003793
Wong K, Liu XJ, Lee AOK, Wong PK (2006) Effect of azo dyes on survivorship, oxygen consumption rate, and filtration rate of the freshwater cladoceran Moina macrocopa. Hum Ecol Risk Assess 12:89–300. https://doi.org/10.1080/10807030500531604
Yadav S, Saravanan PS, Renganathan S (2014) Accumulation of malachite green and cristal violet dye from synthetic effluents using single cell microalgae Chlamydomonas reinhardtii. Asian J Chem 26(15):4801–4806
Zaharia, C, Suteu, D (2012) Textile organic dyes—characteristics, polluting effects and separation/elimination procedures from industrial effluents—a critical overview. Organic Pollutants Ten Years After the Stockholm Convention. Environmental and Analytical Update, Dr. Tomasz Puzyn (Ed.), InTech. https://doi.org/10.5772/32373
Zaharia C, Suteau D (2013) Coal fly ash as adsorptive material for treatment of a real textile effluent: operating parameters and treatment efficiency. Environ Sci Pollut Res 20:2226–2235. https://doi.org/10.1007/s11356-012-1065-z
Zocolo GJ, dos Santos GP, Vendemiatti J, Vacchi FI, Umbuzeiro G, de A, Zanoni MVB (2015) Using SPE–LC–ESI–MS/MS analysis to assess disperse dyes in environmental water samples. J Chromatogr Sci 53:1257–1264. https://doi.org/10.1093/chromsci/bmu221
Zöllner N, Kirsch K (1962) Microdetermination of lipids by the sulphophospho vanillin reaction. Z die Gesamte Exp Med Einschl Exp Chir 135:545–561
Zucker E (1985) Hazard Evaluation Division—standard evaluation procedure: acute toxicity test for freshwater fish (540/9-85-006). USEPA Publication, Washington, USA
Acknowledgements
Miriam Hernández-Zamora received a postdoctoral scholarship from the Consejo Nacional de Ciencia y Tecnología, and thanks the Escuela Nacional de Ciencias Biológicas (IPN) for providing the support and facilities to conduct the present study. Fernando Martínez-Jerónimo acknowledges the Secretaría de Investigación y Posgrado - IPN, and the Comisión de Operación y Fomento de Actividades Académicas del IPN for the support provided.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hernández-Zamora, M., Martínez-Jerónimo, F. Exposure to the azo dye Direct blue 15 produces toxic effects on microalgae, cladocerans, and zebrafish embryos. Ecotoxicology 28, 890–902 (2019). https://doi.org/10.1007/s10646-019-02087-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-019-02087-1