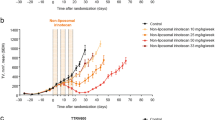
Summary
The (6-maleimidocaproyl)hydrazone derivative of doxorubicin (INNO-206) is an albumin-binding prodrug of doxorubicin with acid-sensitive properties that is being assessed clinically. The prodrug binds rapidly to circulating serum albumin and releases doxorubicin selectively at the tumor site. This novel mechanism may provide enhanced antitumor activity of doxorubicin while improving the overall toxicity profile. Preclinically, INNO-206 has shown superior activity over doxorubicin in a murine renal cell carcinoma model and in breast carcinoma xenograft models. In this work, we compared the antitumor activity of INNO-206 and doxorubicin at their respective maximum tolerated doses in three additional xenograft models (breast carcinoma 3366, ovarian carcinoma A2780, and small cell lung cancer H209) as well as in an orthotopic pancreas carcinoma model (AsPC-1). INNO-206 showed more potent antitumor efficacy than free doxorubicin in all tumor models and is thus a promising clinical candidate for treating a broad range of solid tumors.





Similar content being viewed by others
References
Kratz F, Warnecke A, Schmid B, Chung D-E, Gitzel M (2006) Prodrugs of anthracyclines in cancer chemotherapy. Curr Med Chem 13:477–523
Kratz F, Muller IA, Ryppa C, Warnecke A (2008) Prodrug strategies in anticancer chemotherapy. ChemMedChem 3:20–53, doi:10.1002/cmdc.200700159
Allen TM, Cullis PR (2004) Drug delivery systems: entering the mainstream. Science 303:1818–1822, doi:10.1126/science.1095833
Warnecke A, Fichtner I, Garmann D, Jaehde U, Kratz F (2004) Synthesis and biological activity of water-soluble maleimide derivatives of the anticancer drug carboplatin designed as albumin-binding prodrugs. Bioconjug Chem 15:1349–1359, doi:10.1021/bc049829j
Warnecke A, Fichtner I, Sass G, Kratz F (2007) Synthesis, cleavage profile, and antitumor efficacy of an albumin-binding prodrug of methotrexate that is cleaved by plasmin and cathepsin B. Arch Pharm (Weinheim) 340:389–395, doi:10.1002/ardp.200700025
Warnecke A, Kratz F (2003) Maleimide-oligo(ethylene glycol) derivatives of camptothecin as albumin-binding prodrugs: synthesis and antitumor efficacy. Bioconjug Chem 14:377–387, doi:10.1021/bc0256289
Kratz F, Warnecke A, Scheuermann K, Stockmar C, Schwab J, Lazar P, Drückes P, Esser N, Drevs J, Rognan D, Bissantz C, Hinderling C, Folkers G, Fichtner I, Unger C (2002) Probing the cysteine-34 position of endogenous serum albumin with thiol-binding doxorubicin derivatives: improved efficacy of an acid-sensitive doxorubicin derivative with specific albumin-binding properties compared to that of the parent compound. J Med Chem 45:5523–5533, doi:10.1021/jm020276c
Kratz F, Muller-Driver R, Hofmann I, Drevs J, Unger C (2000) A novel macromolecular prodrug concept exploiting endogenous serum albumin as a drug carrier for cancer chemotherapy. J Med Chem 43:1253–1256, doi:10.1021/jm9905864
Mansour AM, Drevs J, Esser N, Hamada FM, Badary OA, Unger C, Fichtner I, Kratz F (2003) A new approach for the treatment of malignant melanoma: enhanced antitumor efficacy of an albumin-binding doxorubicin prodrug that is cleaved by matrix metalloproteinase 2. Cancer Res 63:4062–4066
Graeser R, Chung DE, Esser N, Moor S, Schachtele C, Unger C, Kratz F (2008) Synthesis and biological evaluation of an albumin-binding prodrug of doxorubicin that is cleaved by prostate-specific antigen (PSA) in a PSA-positive orthotopic prostate carcinoma model (LNCaP). Int J Cancer 122:1145–1154, doi:10.1002/ijc.23050
Kratz, F (2008) Albumin as a drug carrier: Design of prodrugs, drug conjugates and nanoparticles. J. Control. Release 132:171–183
Kratz F, Beyer U (1998) Serum proteins as drug carriers of anticancer agents: a review. Drug Deliv 5:281–299
Kratz F (2007) DOXO-EMCH (INNO-206): the first albumin-binding prodrug of doxorubicin to enter clinical trials. Expert Opin Investig Drugs 16:855–866, doi:10.1517/13543784.16.6.855
Kratz F, Ehling G, Kauffmann HM, Unger C (2007) Acute and repeat-dose toxicity studies of the (6-maleimidocaproyl)hydrazone derivative of doxorubicin (DOXO-EMCH), an albumin-binding prodrug of the anticancer agent doxorubicin. Hum Exp Toxicol 26:19–35, doi:10.1177/0960327107073825
Lebrecht D, Geist A, Ketelsen UP, Haberstroh J, Setzer B, Kratz F, Walker UA (2007) The 6-maleimidocaproyl hydrazone derivative of doxorubicin (DOXO-EMCH) is superior to free doxorubicin with respect to cardiotoxicity and mitochondrial damage. Int J Cancer 120:927–934, doi:10.1002/ijc.22409
Unger C, Haring B, Medinger M, Drevs J, Steinbild S, Kratz F, Mross K (2007) Phase I and pharmacokinetic study of the (6-maleimidocaproyl)hydrazone derivative of doxorubicin. Clin Cancer Res 13:4858–4866, doi:10.1158/1078-0432.CCR-06-2776
Graeser R, Bornmann C, Esser N, Ziroli V, Jantscheff P, Unger C, Hopt UT, Schaechtele C, von Dobschuetz E, Massing U (2008) Anti-metastatic effects of liposomal gemcitabine and empty liposomes in an orthotopic mouse model of pancreatic cancer. Pancreas (in press)
Kratz F, Roth T, Fichtner I, Schumacher P, Fiebig HH, Unger C (2000) In vitro and in vivo efficacy of acid-sensitive transferrin and albumin doxorubicin conjugates in a human xenograft panel and in the MDA-MB-435 mamma carcinoma model. J Drug Target 8:305–318, doi:10.3109/10611860008997908
Trail PA, Willner D, Lasch SJ, Henderson AJ, Greenfield RS, King D, Zoeckler ME, Braslawsky GR (1992) Antigen-specific activity of carcinoma-reactive BR64-doxorubicin conjugates evaluated in vitro and in human tumor xenograft models. Cancer Res 52:5693–5700
Noble S, Goa KL (1997) Gemcitabine. A review of its pharmacology and clinical potential in non-small cell lung cancer and pancreatic cancer. Drugs 54:447–472
Hotz HG, Reber HA, Hotz B, Yu T, Foitzik T, Buhr HJ, Cortina G, Hines OJ (2003) An orthotopic nude mouse model for evaluating pathophysiology and therapy of pancreatic cancer. Pancreas 26:89–98, doi:10.1097/00006676-200305000-00020
Edinger M, Sweeney TJ, Tucker AA, Olomu AB, Negrin RS, Contag CH (1999) Noninvasive assessment of tumor cell proliferation in animal models. Neoplasia 1:303–310, doi:10.1038/sj.neo.7900048
Maeda H, Wu J, Sawa T, Matsumura Y, Hori K (2000) Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release 65:271–284, doi:10.1016/S0168-3659(99)00248-5
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Graeser, R., Esser, N., Unger, H. et al. INNO-206, the (6-maleimidocaproyl hydrazone derivative of doxorubicin), shows superior antitumor efficacy compared to doxorubicin in different tumor xenograft models and in an orthotopic pancreas carcinoma model. Invest New Drugs 28, 14–19 (2010). https://doi.org/10.1007/s10637-008-9208-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-008-9208-2