Abstract
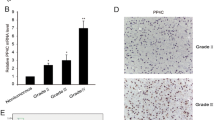
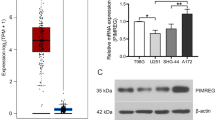
p53-induced protein with a RING-H2 domain (Pirh2), also known as Rchy1, is an ubiquitin E3 ligase that regulates the turnover and functionality of several proteins involved in cell proliferation and differentiation, cell cycle checkpoints, and cell death. However, it remains unclear whether aberrant expression of Pirh2 is involved in the development of glioma, a major type of primary brain tumor in adults. Western blot and immunohistochemical analyses showed that Pirh2 was highly expressed in glioma specimens, compared with normal brain tissues. High Pirh2 expression was positively correlated with higher tumor grade, as well as Ki-67 expression. Kaplan–Meier analysis revealed that patients with high Pirh2 expression had worsened prognosis, compared with those with low Pirh2 expression. Moreover, to determine whether Pirh2 could regulate malignant behavior of glioma cells, we transfected glioma cells with interfering RNA targeting Pirh2 to specifically silence Pirh2 expression. Mechanistically, our results indicated that knockdown of Pirh2 induced the apoptosis of glioma cells. In addition, depletion of Pirh2 diminished the expression of PCNA and cyclin D1 and led to cell cycle arrest at G1 phase. Taken together, these findings for the first time suggest that Pirh2 might play an important role in the regulation of glioma proliferation and apoptosis and thus serve as a promising therapeutic target in the treatment of glioma.




Similar content being viewed by others
References
Beitel LK et al (2002) Cloning and characterization of an androgen receptor N-terminal-interacting protein with ubiquitin-protein ligase activity. J Mol Endocrinol 29:41–60
Bohgaki M et al (2013) The E3 ligase PIRH2 polyubiquitylates CHK2 and regulates its turnover. Cell Death Differ 20:812–822. doi:10.1038/cdd.2013.7
Coppari E, Yamada T, Bizzarri AR, Beattie CW, Cannistraro S (2014) A nanotechnological, molecular-modeling, and immunological approach to study the interaction of the anti-tumorigenic peptide p28 with the p53 family of proteins. Int J Nanomed 9:1799–1813. doi:10.2147/IJN.S58465
Ferguson SD (2011) Malignant gliomas: diagnosis and treatment. Disease-a-month 57:558–569. doi:10.1016/j.disamonth.2011.08.020
Giaccia AJ, Kastan MB (1998) The complexity of p53 modulation: emerging patterns from divergent signals. Genes Dev 12:2973–2983
Gomez-Manzano C et al (1996) Adenovirus-mediated transfer of the p53 gene produces rapid and generalized death of human glioma cells via apoptosis. Cancer Res 56:694–699
Hakem A et al (2011) Role of Pirh2 in mediating the regulation of p53 and c-Myc. PLoS Genet 7:e1002360. doi:10.1371/journal.pgen.1002360
Halaby MJ, Hakem R, Hakem A (2013) Pirh2: an E3 ligase with central roles in the regulation of cell cycle, DNA damage response, and differentiation. Cell Cycle 12:2733–2737. doi:10.4161/cc.25785
Han L et al (2015) HOXB1 is a tumor suppressor gene regulated by miR-3175 in glioma. PLoS ONE 10:e0142387. doi:10.1371/journal.pone.0142387
Hattori T et al (2007) Pirh2 promotes ubiquitin-dependent degradation of the cyclin-dependent kinase inhibitor p27Kip1. Cancer Res 67:10789–10795. doi:10.1158/0008-5472.CAN-07-2033
Jarvius M et al (2013) Piperlongumine induces inhibition of the ubiquitin-proteasome system in cancer cells. Biochem Biophys Res Commun 431:117–123. doi:10.1016/j.bbrc.2013.01.017
Jung YS, Qian Y, Chen X (2011) The p73 tumor suppressor is targeted by Pirh2 RING finger E3 ubiquitin ligase for the proteasome-dependent degradation. J Biol Chem 286:35388–35395. doi:10.1074/jbc.M111.261537
Jung YS, Qian Y, Chen X (2012) Pirh2 RING-finger E3 ubiquitin ligase: its role in tumorigenesis and cancer therapy. FEBS Lett 586:1397–1402. doi:10.1016/j.febslet.2012.03.052
Jung YS, Qian Y, Yan W, Chen X (2013) Pirh2 E3 ubiquitin ligase modulates keratinocyte differentiation through p63. J Invest Dermatol 133:1178–1187. doi:10.1038/jid.2012.466
Karpel-Massler G et al (2016) Inhibition of deubiquitinases primes glioblastoma cells to apoptosis in vitro and in vivo. Oncotarget 7:12791–12805. doi:10.18632/oncotarget.7302
Kymionis GD, Kankariya VP, Plaka AD, Reinstein DZ (2012) Femtosecond laser technology in corneal refractive surgery: a review. J Refract Surg 28:912–920. doi:10.3928/1081597X-20121116-01
Leng RP et al (2003) Pirh2, a p53-induced ubiquitin-protein ligase, promotes p53 degradation. Cell 112:779–791
Li H et al (1999) Intracerebral adenovirus-mediated p53 tumor suppressor gene therapy for experimental human glioma. Clin Cancer Res 5:637–642
Lin Z et al (2012) USP22 antagonizes p53 transcriptional activation by deubiquitinating Sirt1 to suppress cell apoptosis and is required for mouse embryonic development. Mol Cell 46:484–494. doi:10.1016/j.molcel.2012.03.024
McClurg UL, Robson CN (2015) Deubiquitinating enzymes as oncotargets. Oncotarget 6:9657–9668. doi:10.18632/oncotarget.3922
Patel AP et al (2014) Single-cell RNA-seq highlights intratumoral heterogeneity in primary glioblastoma. Science 344:1396–1401. doi:10.1126/science.1254257
Reifenberger G, Liu L, Ichimura K, Schmidt EE, Collins VP (1993) Amplification and overexpression of the MDM2 gene in a subset of human malignant gliomas without p53 mutations. Cancer Res 53:2736–2739
Reifenberger J et al (1996) Analysis of p53 mutation and epidermal growth factor receptor amplification in recurrent gliomas with malignant progression. J Neuropathol Exp Neurol 55:822–831
Sheng Y et al (2008) Molecular basis of Pirh2-mediated p53 ubiquitylation. Nat Struct Mol Biol 15:1334–1342. doi:10.1038/nsmb.1521
Takano S (2016) Bevacizumab treatment for malignant glioma based on basic research and clinical trial. Gan to kagaku ryoho Cancer Chemother 43:694–701
Yan W, Jung YS, Zhang Y, Chen X (2014) Arsenic trioxide reactivates proteasome-dependent degradation of mutant p53 protein in cancer cells in part via enhanced expression of Pirh2 E3 ligase. PLoS ONE 9:e103497. doi:10.1371/journal.pone.0103497
Yu SC et al (2008) Isolation and characterization of cancer stem cells from a human glioblastoma cell line U87. Cancer Lett 265:124–134. doi:10.1016/j.canlet.2008.02.010
Zheng H et al (2008) p53 and Pten control neural and glioma stem/progenitor cell renewal and differentiation. Nature 455:1129–1133. doi:10.1038/nature07443
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Yifeng Bao and Xue Wu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Bao, Y., Wu, X., Yuan, D. et al. High Expression of Pirh2 is Associated with Poor Prognosis in Glioma. Cell Mol Neurobiol 37, 1501–1509 (2017). https://doi.org/10.1007/s10571-017-0481-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-017-0481-5