Abstract
Recently, the isolation of chitin nano-crystals (ChNCs) via green methods has received great interests in the scientific research community. In this study, one-pot and eco-friendly process to isolate ChNCs from Metapenaeus ensis shell via solid maleic acid hydrolysis was proposed. It was found that the highest ChNC yield can be reached to 10.42% when at the condition of 75% acid concentration and 110 °C for 3 h, and that the as-prepared ChNC suspensions exhibited extraordinary colloidal stability for 7 months. The corresponding length of ChNCs was 170.6 nm and the carboxylic group content was as high as 0.173 mmol/g. ChNCs were then applied as the efficient and green supports of silver nanoparticles (Ag NPs) and found that ChNCs can help generate Ag NPs with tiny and uniform particle size. The study paves a great potential way to isolate ChNCs from marine crustacean with high colloidal stability and the potential application in the preparation of uniform noble metal nanoparticles.
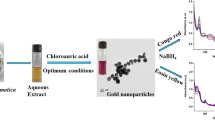
Graphic abstract
A one-pot facile and environmental friendly approach to isolate ChNCs from Chitin (Metapenaeus ensis shell as the marine biomass precursors) with high colloidal stability via solid maleic acid hydrolysis is proposed.










Similar content being viewed by others
References
Agasti N, Kaushik NK (2018) Synthesis and characterization of stearic acid capped silver nanoparticles: pH dependent stabilization and colorimetric detection of Hg(II) in water. Adv Mater Lett 9(1):53–57. https://doi.org/10.5185/amlett.2018.1756
Akhlaghi SP, Berry RC, Tam KC (2013) Surface modification of cellulose nanocrystal with chitosan oligosaccharide for drug delivery applications. Cellulose 20(4):1747–1764. https://doi.org/10.1007/s10570-013-9954-y
Aklog YF, Nagae T, Izawa H, Morimoto M, Saimoto H, Ifuku S (2016) Preparation of chitin nanofibers by surface esterification of chitin with maleic anhydride and mechanical treatment. Carbohydr Polym 153:55–59. https://doi.org/10.1016/j.carbpol.2016.07.060
An XY, Wen YB, Cheng D, Zhu XH, Ni YH (2016a) Preparation of cellulose nano-crystals through a sequential process of cellulase pretreatment and acid hydrolysis. Cellulose 23(4):2409–2420. https://doi.org/10.1007/s10570-016-0964-4
An XY, Cheng D, Dai L, Wang BB, Ocampo HJ, Nasrallah J, Jia X, Zou JJ, Long YD, Ni YH (2017a) Synthesis of nano-fibrillated cellulose/magnetite/titanium dioxide (NFC@Fe3O4@TNP) nanocomposites and their application in the photocatalytic hydrogen generation. Appl Catal B 206:53–64. https://doi.org/10.1016/j.apcatb.2017.01.021
An XY, Long YD, Ni YH (2017) Cellulose nanocrystal/hexadecyltrimethylammonium bromide/silver nanoparticle composite as a catalyst for reduction of 4-nitrophenol. Carbohydr Polym 156:253–258. https://doi.org/10.1016/j.carbpol.2016.08.099
An XY, Wen YB, Almujil A, Cheng D, Li JG, Jia X, Zou JJ, Ni YH (2016) Nano-fibrillated cellulose (NFC) as versatile carriers of TiO2 nanoparticles (TNPs) for photocatalytic hydrogen generation. RSC Adv 6(92):89457–89466. https://doi.org/10.1039/C6RA21042B
Azuma K, Ifuku S (2016) Nanofibers based on chitin: a new functional food. Pure Appl Chem 88(6):605. https://doi.org/10.1515/pac-2016-0504
Azuma K, Nishihara M, Shimizu H, Itoh Y, Takashima O, Osaki T, Itoh N, Imagawa T, Murahata Y, Tsuka T, Izawa H, Ifuku S, Minami S, Saimoto H, Okamoto Y, Morimoto M (2015) Biological adhesive based on carboxymethyl chitin derivatives and chitin nanofibers. Biomaterials 42:20–29. https://doi.org/10.1016/j.biomaterials.2014.11.043
Barikani M, Oliaei E, Seddiqi H, Honarkar H (2014) Preparation and application of chitin and its derivatives: a review. Iran Polym J 23(4):307–326. https://doi.org/10.1007/s13726-014-0225-z
Bian H, Chen L, Dai H, Zhu JY (2017) Effect of fiber drying on properties of lignin containing cellulose nanocrystals and nanofibrils produced through maleic acid hydrolysis. Cellulose 24(10):4205–4216. https://doi.org/10.1007/s10570-017-1430-7
Bian H, Luo J, Wang RB, Zhou XL, Ni SZ, Shi R, Fang G, Dai H (2019) Recyclable and reusable maleic acid for efficient production of cellulose nanofibrils with stable performance. ACS Sustain Chem Eng 7:20022–20031. https://doi.org/10.1021/acssuschemeng.9b05766
Börjesson M, Westman G (2015) Crystalline nanocellulose - preparation, modification, and properties. Cellulose: fundamental aspects and current trends chapter: 7, Publisher: InTech, Editors: Dr. Matheus Poletto, 159–191. https://doi.org/10.5772/61899
Cetin NS, Tingaut P, Ozmen N, Henry N, Harper D, Dadmun M, Se`be G (2009) Acetylation of cellulose nanowhiskers with vinyl acetate under moderate conditions. Macromol Biosci 9:997–1003. https://doi.org/10.1002/mabi.200900073
Chen Y, Liu C, Chang PR, Cao X, Anderson DP (2009) Bionanocomposites based on pea starch and cellulose nanowhiskers hydrolyzed from pea hull fibre: effect of hydrolysis time. Carbohydr Polym 76(4):607–615. https://doi.org/10.1016/j.carbpol.2008.11.030
Cheng D, Wen YB, An XY, Zhu XH, Cheng XJ, Zheng LQ, Nasrallah JE (2016) Improving the colloidal stability of cellulose nano-crystals by surface chemical grafting with polyacrylic acid. J Bioresour Bioprod 1(3):114–119. https://doi.org/10.21967/jbb.v1i3.6
Clark GL, Smith AF (1936) X-ray diffraction studies of chitin, chitosan, and derivatives. J Phys Chem 40(7):863–879. https://doi.org/10.1021/j150376a001
Coates J (2006) Interpretation of infrared spectra, a practical approach. Encyclopedia of Analytical Chemistry. Wiley, Hoboken, pp 1–23. https://doi.org/10.1002/9780470027318$4a5606
Corrêa AC, Teixeira E, Pessan LA, Mattoso LHC (2010) Cellulose nanofibers from curaua fibers. Cellulose 17(6):1183–1192. https://doi.org/10.1007/s10570-010-9453-3
Dai L, Wang Y, Zou X, Chen Z, Liu H, Ni Y (2020) Ultrasensitive physical, bio, and chemical sensors derived from 1-, 2‐, and 3‐D nanocellulosic materials. Small 16:1906567. https://doi.org/10.1002/smll.201906567
Trachea D, Hussin MH, Mohamad Haafizc MK, Thakurd VK (2017) Recent progress in cellulose nanocrystals. Nanoscale 9(5):1763–1786. https://doi.org/10.1039/C6NR09494E
Edgar KJ, Arnold KM, Blount WW, Lawniczak JE, Lowman DW (1995) Synthesis and properties of cellulose acetoacetates. Macromolecules 28:4122–4128. https://doi.org/10.1021/ma00116a011
Fan Y, Saito T, Isogai A (2008) Chitin nanocrystals prepared by TEMPO-mediated oxidation of alpha chitin. Biomacromol 9:192–198. https://doi.org/10.1021/bm700966g
Fan Y, Saito T, Isogai A (2010) Individual chitin nano-whiskers prepared from partially deacetylated a-chitin by fibril surface cationization. Carbohydr Polym 79:1046–1051. https://doi.org/10.1016/j.carbpol.2009.10.044
Filson PB, Dawson-Andoh BE (2009) Sono-chemical preparation of cellulose nanocrystals from lignocellulose derived materials. Biores Technol 100:2259–2264. https://doi.org/10.1016/j.biortech.2008.09.062
Fujisawa S, Okita Y, Fukuzumi H, Saito T, Isogai A (2011) Preparation and characterization of TEMPO-oxidized cellulose nanofibril films with free carboxyl groups. Carbohydr Polym 84(1):579–583. https://doi.org/10.1016/j.carbpol.2010.12.029
Garcia-Garcia D, Balart R, Lopez-Martinez J, Ek M, Moriana R (2018) Optimizing the yield and physico-chemical properties of pine cone cellulose nanocrystals by different hydrolysis time. Cellulose 25(5):2925–2938. https://doi.org/10.1007/s10570-018-1760-0
Gao K, Guo Y, Niu Q, Fang H, Zhang L, Zhang Y, Wang L, Zhou L (2018) Effects of chitin nanofibers on the microstructure and properties of cellulose nanofibers/chitin nanofibers composite aerogels. Cellulose 25(8):4591–4602. https://doi.org/10.1007/s10570-018-1899-8
Goodrich JD, Winter WT (2007) α-Chitin nanocrystals prepared from shrimp shells and their specific surface area measurement. Biomacromol 8:252–257. https://doi.org/10.1021/bm0603589
Gopi S, Balakrishnan P, Pius A, Thomas S (2017) Chitin nanowhisker (ChNW)-functionalized electrospun PVDF membrane for enhanced removal of Indigo carmine. Carbohydr Polym 165:115–122. https://doi.org/10.1016/j.carbpol.2017.02.046
Gopi S, Balakrishnan P, Divya C, Valic S, Govorcin Bajsic E, Pius A, Thomas S (2017b) Facile synthesis of chitin nanocrystals decorated on 3D cellulose aerogels as a new multi-functional material for waste water treatment with enhanced anti-bacterial and anti-oxidant properties. New J Chem 41(21):12746–12755. https://doi.org/10.1039/C7NJ02392H
Gopi S, Pius A, Kargl R, Kleinschek KS, Thomas S (2019) Fabrication of cellulose acetate/chitosan blend films as efficient adsorbent for anionic water pollutants. Polym Bull 76:1557–1571. https://doi.org/10.1007/s00289-018-2467-y
Gopi S, Pius A, Thomas S (2016) Enhanced adsorption of crystal violet by synthesized and characterized chitin nano whiskers from shrimp shell. J Water Process Eng 14:1–8. https://doi.org/10.1016/j.jwpe.2016.07.010
Herrera M, Thitiwutthisakul K, Yang X, Rujitanaroj PO, Rojas R, Berglund L (2018) Preparation and evaluation of high-lignin content cellulose nanofibrils from eucalyptus pulp. Cellulose 25(5):3121–3133. https://doi.org/10.1007/s10570-018-1764-9
Hoeng F, Denneulin A, Neuman C, Bras J (2015) Charge density modification of carboxylated cellulose nanocrystals for stable silver nanoparticles suspension preparation. J Nanopart Res 17(6):244. https://doi.org/10.1007/s11051-015-3044-z
Ifuku S, Tsukiyama Y, Yukawa T, Egusa M, Kaminaka H, Izawa H, Morimoto M, Saimoto H (2015) Facile preparation of silver nanoparticles immobilized on chitin nanofiber surfaces to endow antifungal activities. Carbohydr Polym 117:813–817. https://doi.org/10.1016/j.carbpol.2014.10.042
Istominaa AP, Bogdanova OI, Streltsov DR, Chvalun SN (2019) Stability of suspensions of α-chitin nanocrystals obtained by TEMPO oxidation. Polym Sci Ser A 61(5):589–597. https://doi.org/10.1134/s0965545X19050080
Ji H, Xiang Z, Qi H, Han T, Pranovich A, Song T (2019) Strategy towards one-step preparation of carboxylic cellulose nanocrystals and nanofibrils with high yield, carboxylation and highly stable dispersibility using innocuous citric acid. Green Chem 21(8):1956–1964. https://doi.org/10.1039/C8GC03493A
Jiang J, Ye W, Yu J, Fan Y, Ono Y, Saito T, Isogai A (2018) Chitin nano-crystals prepared by oxidation of apha-chitin using the O2/Laccase/TEMPO system. Carbohydr Polym 189:178–183. https://doi.org/10.1016/j.carbpol.2018.01.096
João CFC, Silva JC, Borges JP (2015) Chitin-based nanocomposites: biomedical applications. Eco-friendly Polym Nanocompos 74:439–457. https://doi.org/10.1007/978-81-322-2473-0$414
Kalman F, Borsal J, Kemeny S, Rusznak I (1988) Effect of the reaction conditions on the degree of substitution of carboxymethyl cellulose. Colloid Polym Sci 266:716–720. https://doi.org/10.1007/BF01410280
Kalaivani R, Maruthupandy M, Muneeswaran T, Hameedha Beevi A, Anand M, Ramakritinan CM, Kumaraguru AK (2018) Synthesis of chitosan mediated silver nanoparticles (Ag NPs) for potential antimicrobial applications. Frontiers Lab Med 2(1):30–35. https://doi.org/10.1016/j.flm.2018.04.002
Kargarzadeh H, Ahmad I, Abdullah I, Dufresne A, Zainudin SY, Sheltami RM (2012) Effects of hydrolysis conditions on the morphology, crystallinity, and thermal stability of cellulose nanocrystals extracted from kenaf bast fibers. Cellulose 19:855–866. https://doi.org/10.1007/s10570-012-9684-6
Larbi F, Garcia A, Del Valle LJ, Hamou A, Puiggali J, Belgacem N, Bras J (2018) Comparison of nanocrystals and nanofibers produced from shrimp shell alpha-chitin: From energy production to material cytotoxicity and Pickering emulsion properties. Carbohydr Polym 196:385–397. https://doi.org/10.1016/j.carbpol.2018.04.094
Lee CL, Syu CM, Huang CH, Chiou HP, Chao YJ, Yang CC (2013) Cornered silver and silverplatinum nanodisks: preparation and promising activity for alkaline oxygen reduction catalysis. Appl Catal B 132–133:229–236. https://doi.org/10.1016/j.apcatb.2012.11.038
Li WW, Cai GX, Zhang PD (2019) A simple and rapid fourier transform infrared method for the determination of the degree of acetyl substitution of cellulose nanocrystals. J Mater Sci 54:8047–8056. https://doi.org/10.1007/s10853-019-03471-2
Lin X, Guo X, Xu C, Wu M (2019) Carboxymethyl cellulose assisted mechanical preparation of cellulose nanocrystals with high yield. Cellulose 26:5227–5236. https://doi.org/10.1007/s10570-019-02452-7
Liu LQ, An XY, Zhang H, Lu ZH, Nie SX, Cao HB, Xu QL, Liu HB (2020) Ball milling pretreatment facilitating α-amylase hydrolysis for production of starch-based bio-latex with high performance. Carbohydr Polym 242:116384. https://doi.org/10.1016/j.carbpol.2020.116384
Matsuda H (1987) Preparation and utilization of esterified woods bearing carboxyl groups. Wood Sci Technol 21:75–88. https://doi.org/10.1007/BF00349719
Millet MA, Moore WE, Saeman JE (1954) Preparation and properties of hydrocelluloses. Ind Eng Chem Res 46(7):1493–1497. https://doi.org/10.1021/ie50535a051
Mohaiyiddin MS, Lin OH, Owi WT, Chan CH, Chia CH, Zakaria S, Villagracia AR, Akil HM (2016) Characterization of nanocellulose recovery from Elaeis guineensis frond for sustainable development. Clean Technol Environ Policy 18(8):2503–2512. https://doi.org/10.1007/s10098-016-1191-2
Naduparambath S, Jinitha TV, Shaniba V, Sreejith MP, Balan AK, Purushothaman E (2017) Isolation and characterisation of cellulose nanocrystals from sago seed shells. Carbohydr Polym 180:13–20. https://doi.org/10.1016/j.carbpol.2017.09.088
Nair KG, Dufresne A (2003) Crab shell chitin whisker reinforced natural rubber nanocomposites. 1. processing and swelling behavior. Biomacromol 4:657–665. https://doi.org/10.1021/bm020127b
Oun AA, Rhim JW (2017) Effect of oxidized chitin nanocrystals isolated by ammonium persulfate method on the properties of carboxymethyl cellulose-based films. Carbohydr Polym 175:712–720. https://doi.org/10.1016/j.carbpol.2017.08.052
Oun AA, Rhim JW (2018) Effect of isolation methods of chitin nanocrystals on the properties of chitin-silver hybrid nanoparticles. Carbohydr Polym 197:349–358. https://doi.org/10.1016/j.carbpol.2018.06.033
Paillet M, Dufresne A (2001) Chitin whisker reinforced thermoplastic nanocomposites. Macromolecules 34(19):6527–6530. https://doi.org/10.1021/ma002049v
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK (2010) Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels 3:10. https://doi.org/10.1186/1754-6834-3-10
Pereira AGB, Muniz EC, Hsieh YL (2014) Chitosan-sheath and chitin-core nanowhiskers. Carbohydr Polym 107:158–166. https://doi.org/10.1016/j.carbpol.2014.02.046
Phanthong P, Guan G, Ma Y, Hao X, Abudula A (2016) Effect of ball milling on the production of nanocellulose using mild acid hydrolysis method. J Taiwan Inst Chem Eng 60:617–622. https://doi.org/10.1016/j.jtice.2015.11.001
Romano P, Fabritius H, Raabe D (2007) The exoskeleton of the lobster Homarus americanus as an example of a smart anisotropic biological material. Acta Biomater 3:301–309. https://doi.org/10.1016/j.actbio.2006.10.003
Sadeghifar H, Filpponen I, Clarke SP, Brougham DF, Argyropoulos DS (2011) Production of cellulose nanocrystals using hydrobromic acid and click reactions on their surface. Journal of Material Science 46(22):7344–7355. https://doi.org/10.1007/s10853-011-5696-0
Sadeghifar H, Filpponen I, Clarke SP, Brougham DF, Argyropoulos DS (2011) Production of cellulose nanocrystals using hydrobromic acid and click reactions on their surface. J Mater Sci 46(22):7344–7355. https://doi.org/10.1007/s10853-011-5696-0
Salaberria AM, Fernandes SCM, Diaz RH, Labidi J (2015) Processing of α-chitin nanofibers by dynamic high pressure homogenization: Characterization and antifungal activity against A. niger. Carbohydr Polym 116:286–191. https://doi.org/10.1016/j.carbpol.2014.04.047
Seta FT, An XY, Liu LQ, Zhang H, Yang J, Zhang W, Nie SX, Yao SQ, Cao HB, Xu QL, Bu YF, Liu HB (2020) Preparation and characterization of high yield cellulose nanocrystals (CNC) derived from ball mill pretreatment and maleic acid hydrolysis. Carbohydr Polym 234:115942. https://doi.org/10.1016/j.carbpol.2020.115942
Song Y, Elsayed-Ali HE (2010) Aqueous phase Ag nanoparticles with controlled shapes fabricated by a modified nanosphere lithography and their optical properties. Appl Surf Sci 256(20):5961–5967. https://doi.org/10.1016/j.apsusc.2010.03.102
Tahir M, Tahir B, Saidina Amin NA (2016) Synergistic effect in plasmonic Au/Ag alloy NPs co-coated TiO2 NWs toward visible-light enhanced CO2 photoreduction to fuels. Appl Catal B 204:548–560. https://doi.org/10.1016/j.apcatb.2016.11.062
Torlopovv MA, Martakov IS, Mikhaylov VI, Tsvetkov NV, Krivoshapkin PV (2017) Regulation of structure, rheological and surface properties of chitin nanocrystal dispersions. Carbohydr Polym 174:1164–1171. https://doi.org/10.1016/j.carbpol.2017.07.036
Uzun I, Celik O (2015) Physicochemical characterization and the comparison of chitin and chitin modified with maleic anhydride. Orient J Chem 31(2):619–627. https://doi.org/10.13005/ojc/310202
Wang QX, Yan Y, Chang L, Zhou J (2018) Fabrication and characterization of chitin nanofibers through esterification and ultrasound treatment. Carbohydr Polym 180:81–87. https://doi.org/10.1016/j.carbpol.2017.09.010
Wang R, Chen L, Zhu JY, Yang R (2017) Tailored and integrated production of carboxylated cellulose nanocrystals (CNC) with nanofibrils (CNF) through maleic acid hydrolysis. ChemNanoMat 3(5):328–335. https://doi.org/10.1002/cnma.201700015
Xia Y, Gilroy KD, Peng HC, Xia X (2017) Seed-mediated growth of colloidal metal nanocrystals. Angew Chem Int Ed 56(1):60–95. https://doi.org/10.1002/anie.201604731
Yang J, An XY, Liu LQ, Seta FT, Zhang H, Nie SX, Yao SQ, Cao HB, Xu QL, Liu HB, Ni YH (2020) Chitin nano-crystals/sodium lignosulfonate/Ag NPs nanocomposites: a potent and green catalyst for efficient removal of organic contaminants. Cellulose 27:5071–5087. https://doi.org/10.1007/s10570-020-03161-2
Yeganeh F, Behrooz R, Rahimi M (2017) The effect of sulfuric acid and maleic acid on characteristics of nano-cellulose produced from waste office paper. Int J Nano Dimens 8(3):206–215
Yu Z, Dang Q, Liu C, Cha D, Zhang H, Zhu W, Zhang Q, Fan B (2017) Preparation and characterization of poly(maleic acid)-grafted cross-linked chitosan microspheres for Cd(II) adsorption. Carbohydr Polym 172:28–39. https://doi.org/10.1016/j.carbpol.2017.05.039
Yuan Y, Hong S, Lian H, Zhang K, Liimatainen H (2020) Comparison of acidic deep eutectic solvents in production of chitin nanocrystals. Carbohydr Polym 236:116095. https://doi.org/10.1016/j.carbpol.2020.116095
Zeng JB, He YS, Li SL, Wang YZ (2012) Chitin whiskers: an overview. Biomacromol 13(1):1–11. https://doi.org/10.1021/bm201564a
Zhang Y, Niu H, Zhang X, Pan J, Dong Y, Wang H, Gao Y (2017) Magnetic N-containing carbon spheres derived from sustainable chitin for the selective oxidation of C–H bonds. RSC Adv 7:51831–51837. https://doi.org/10.1039/C7RA10226G
Zhang Y, Xue C, Xue Y, Gao R, Zhang X (2005) Determination of the degree of deacetylation of chitin and chitosan by X-ray powder diffraction. Carbyhydr Res 340:1914–1917. https://doi.org/10.1016/j.carres.2005.05.005
Zhong TH, Wolcott MP, Liu H, Wang JW (2019) Developing chitin nanocrystals for flexible packaging coatings. Carbohydr Polym 226:115276. https://doi.org/10.1016/j.carbpol.2019.115276
Acknowledgments
The authors would like to acknowledge the financial support from the National Key Research and Development Program (Grant: 2019YFC1905904), the Natural Science Foundation of Tianjin (Grant: 19JCQNJC05400), the State Scholarship Fund for Post-Doctoral Fellow (PDF) from China Scholarship Council (CSC) (Grant: 201908120087), the research fund of Zhejiang Jing Xing Paper Joint Stock Co., Ltd. (Grant: 21843), and the Research Fund of Jiangsu Provincial Key Laboratory of Pulp and Paper Science and Technology, Nanjing Forestry University (Grant: KL201914).
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. Dr. LL is responsible for Software, Methodology, Experiments, Data curation, Validation, Writing - review & editing; Mr. FTS is responsible for Data curation, Methodology, Software, Visualization, Experiments, Writing - review & editing; Dr. LL and Mr. FTS contribute equally to this work as co-first authors. Dr. XA and Dr. HL as the corresponding authors; Mr. JY, Mr. WZ, Dr. HD, Mr. HC and Mr. QX for part contribution of software, data curation, project supporting, and analysis. All authors have given approval to the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, L., Seta, F.T., An, X. et al. Facile isolation of colloidal stable chitin nano-crystals from Metapenaeus ensis shell via solid maleic acid hydrolysis and their application for synthesis of silver nanoparticles. Cellulose 27, 9853–9875 (2020). https://doi.org/10.1007/s10570-020-03499-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-020-03499-7