Abstract
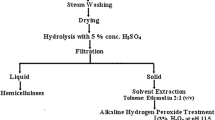
Sugarcane bagasse was selected as residual feedstock to investigate the effects of an acid/peroxide-alkali (APA) combined pretreatment on the extraction of cellulose and on its applicability for producing cellulose acetate. The pretreatment was effective in disrupting the recalcitrant lignocellulose structure and isolating cellulose (SBC), which reached 85.67% purity. High performance liquid chromatography, 1H and 13C nuclear magnetic resonance, Fourier transform infrared spectroscopy, thermogravimetry, calorimetry, X-ray diffractometry (XRD), scanning electron microscopy and elemental analysis were applied to characterize the materials. Crystallinity and accessibility were evaluated by Segal and deconvolution crystallinity indexes, full-width at half maximum, crystallite size (L), total crystalline index and lateral order index parameters. SBC cellulose presented lower crystallinity and was classified after XRD peak deconvolution as a mixture of cellulose II polymorph with considerable amorphicity and some features of remaining cellulose Iβ from sugarcane bagasse, while CC was labeled type Iβ, indicating that the sustainable material is more susceptible to chemical modifications. The degrees of substitution of acetylation products showed that both celluloses have satisfactory reactivity and reached mono, di and triacetylation in different reaction conditions. SBC cellulose is presented as an efficient and renewable alternative to commercial cellulose for producing chemicals, and it is proposed that the APA pretreatment had a significant influence on the low crystallinity and high accessibility of this material.
Graphical abstract










Similar content being viewed by others
References
Abdelwahab NA, Ammar NS, Ibrahim HS (2015) Graft copolymerization of cellulose acetate for removal and recovery of lead ions from wastewater. Int J Biol Macromol 79:913–922. https://doi.org/10.1016/j.ijbiomac.2015.05.022
Agarwal UP, Reiner RS, Ralph SA (2010) Cellulose I crystallinity determination using FT-Raman spectroscopy: univariate and multivariate methods. Cellulose 17:721–733. https://doi.org/10.1007/s10570-010-9420-z
Agarwal UP, Ralph SA, Reiner RS, Baez C (2016) Probing crystallinity of never-dried wood cellulose with Raman spectroscopy. Cellulose 23:125–144. https://doi.org/10.1007/s10570-015-0788-7
Agarwal UP, Ralph SA, Baez C, Reiner RS, Verrill SP (2017) Effect of sample moisture content on XRD-estimated cellulose crystallinity index and crystallite size. Cellulose 24:1971–1984. https://doi.org/10.1007/s10570-017-1259-0
Assumpção SMN, Pontes LAM, Carvalho LS, Campos LMA, Andrade JCF, Silva EG (2016) Pré-tratamento combinado H2SO4/H2O2/NaOH para obtenção das frações lignocelulósicas do bagaço da cana-de-açúcar. Revista Virtual de Química 8:803–822. https://doi.org/10.5935/1984-6835.20160059
Barud HS, Araújo AM Jr, Santos DB, Assunção RMN, Meireles CS, Cerqueira DA, Rodrigues Filho G, Ribeiro CA, Messaddeq Y, Ribeiro SJL (2008) Thermal behavior of cellulose acetate produced from homogeneous acetylation of bacterial cellulose. Thermochim Acta 471:61–69. https://doi.org/10.1016/j.tca.2008.02.009
Brodeur-Campbell M, Klinger J, Shonnard D (2012) Feedstock mixture effects on sugar monomer recovery following dilute acid pretreatment and enzymatic hydrolysis. Biores Tech 116:320–326. https://doi.org/10.1016/j.biortech.2012.03.090
Candido RG, Godoy GG, Gonçalves AR (2017) Characterization and application of cellulose acetate synthesized from sugarcane bagasse. Carbohyd Polym 167:280–289. https://doi.org/10.1016/j.carbpol.2017.03.057
Cardona CA, Quintero JA, Paz IC (2010) Production of bioethanol from sugarcane bagasse: status and perspectives. Biores Tech 101:4754–4766
Carrillo I, Mendonça RT, Ago M, Rojas OJ (2018) Comparative study of cellulosic components isolated from different Eucalyptus species. Cellulose 25:1011–1029. https://doi.org/10.1007/s10570-018-1653-2
Cerqueira DA, Filho GR, Meireles CS (2007) Optimization of sugarcane bagasse cellulose acetylation. Carbohydr Polym 69:579–582. https://doi.org/10.1016/j.carbpol.2007.01.010
Chandel AK, Antunes FAF, Anjos V, Bell MJV, Rodrigues LN, Polikarpov I, Azevedo ER, Bernardinelli OD, Rosa CA, Pagnocca FC, Silva SS (2014) Multi-scale structural and chemical analysis of sugarcane bagasse in the process of sequential acid– base pretreatment and ethanol production by Scheffersomyces shehatae and Saccharomyces cerevisiae. Biotechnol Biofuels 7:1–17. https://doi.org/10.1186/1754-6834-7-63
Cheng G, Varanasi P, Li C, Liu H, Melnichenko YB, Simmons BA, Singh S (2011) Transition of cellulose crystalline structure and surface morphology of biomass as a function of ionic liquid pretreatment and its relation to enzymatic hydrolysis. Biomacromology 12:933–941. https://doi.org/10.1021/bm101240zf
Ciolacu D, Popa VI (2010) Cellulose allomorphs—overview and perspective. In: Lejeune A, Deprez T (eds) Cellulose: structure and properties, derivatives and industrial uses. Nova Science Publishers, New York, pp 1–38
Ciolacu D, Pitol-Filho L, Ciolacu F (2012) Studies concerning the accessibility of different allomorphic forms of cellulose. Cellulose 19:55–68. https://doi.org/10.1007/s10570-011-9620-1
Corrales RCNR, Mendes FMT, Perrone CC, Sant’Anna C, Souza W, Abud Y, Bom SEP, Ferreira-Leitão V (2012) Structural evaluation of sugar cane bagasse steam pretreated in the presence of CO2 and SO2. Biotechnol Biofuels 5:1–8. https://doi.org/10.1186/1754-6834-5-36
Daud WWR, Djuned FM (2015) Cellulose acetate from oil palm empty fruit bunch via a one-step heterogeneous acetylation. Carbohyd Polym 132:252–260. https://doi.org/10.1016/j.carbpol.2015.06.011
Diep NQ, Sakanishi K, Nakagoshi N, Fujimoto S, Minowa T, Tran XD (2012) Biorefinery: concepts, current status, and development trends. Int J Biomass Renew 1:1–8
El Nemr A, Ragab S, El Sikaily A, Khaled A (2015) Synthesis of cellulose triacetate from cotton cellulose by using NIS as a catalyst under mild reaction conditions. Carbohyd Polym 130:41–48. https://doi.org/10.1016/j.carbpol.2015.04.065
El Oudiani A, Chaabouni Y, Msahli S, Sakli F (2011) Crystal transition from cellulose I to cellulose II in NaOH treated Agave americana L. fibre. Carbohyd Polym 86:1221–1229. https://doi.org/10.1016/j.carbpol.2011.06.037
Endo T, Aung EM, Fujii S, Hosomi S, Kimizu M, Ninomiya K, Takahashi K (2017) Investigation of accessibility and reactivity of cellulose pretreated by ionic liquid at high loading. Carbohydr Polym 176:365–373. https://doi.org/10.1016/j.carbpol.2017.08.105
Fan G, Wang M, Liao C, Fang T, Li J, Zhou R (2013) Isolation of cellulose from rice straw and its conversion into cellulose acetate catalyzed by phosphotungstic acid. Carbohyd Polym 94:71–76. https://doi.org/10.1016/j.carbpol.2013.01.073
Fan G, Liao C, Fang T, Luo S, Song G (2014) Amberlyst 15 as a new and reusable catalyst for the conversion of cellulose into cellulose acetate. Carbohyd Polym 112:203–209. https://doi.org/10.1016/j.carbpol.2014.05.082
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21:885–896. https://doi.org/10.1007/s10570-013-0030-4
French AD, Santiago Cintrón M (2013) Cellulose polymorphy, crystallite size, and the Segal Crystallinity Index. Cellulose 20:583–588. https://doi.org/10.1007/s10570-012-9833-y
Goodlett VW, Dougherty JT, Patton HW (1971) Characterization of cellulose acetates by nuclear magnetic resonance. J Polym Sci A 9:155–161. https://doi.org/10.1002/pol.1971.150090114
Gouveia ER, Nascimento RT, Souto-Maior AM, Rocha GJM (2009) Validation of methodology for the chemical characterization of sugar cane bagasse. Química Nova 32:1500–1503. https://doi.org/10.1590/S0100-40422009000600026
He J, Zhang M, Cui S, Wang S (2009) High-quality cellulose triacetate prepared from bamboo dissolving pulp. J Appl Polym Sci 113:456–465. https://doi.org/10.1002/app.30008
Horikawa Y (2017) Assessment of cellulose structural variety from different origins using near infrared spectroscopy. Cellulose 24:5313–5325. https://doi.org/10.1007/s10570-017-1518-0
Jahan MS, Sarkar M, Rahman MM (2015) Sodium carbonate pre-extraction of bamboo prior to soda-anthraquinone pulping. Biomass Convers Biorefin 5:417–423. https://doi.org/10.1007/s13399-015-0160-z
Kalogiannis KG, Stefanidis S, Marianou A, Michailof C, Kalogiann A, Lappas A (2015) Lignocellulosic biomass fractionation as a pretreatment step for production of fuels and green chemicals. Waste Biomass Valoriz 6:781–790. https://doi.org/10.1007/s12649-015-9387-y
Kautto J, Realff MJ, Ragauskas AJ (2013) Design and simulation of an organosolv process for bioethanol production. Biomass Convers Biorefin 3:199–212. https://doi.org/10.1007/s13399-013-0074-6
Kono H, Numata Y, Nagai N, Erata T, Takai M (1999) CPMAS 13C NMR and X-ray studies of cellooligosaccharide acetates as a model for cellulose triacetate. J Polym Sci A Polym Chem 37:4100–4107. https://doi.org/10.1002/(SICI)1099-0518(19991115)37:22%3c4100:AID-POLA8%3e3.0.CO;2-D
Kono H, Hashimoto H, Shimizu Y (2015) NMR characterization of cellulose acetate: chemical shift assignments, substituent effects, and chemical shift additivity. Carbohyd Polym 118:91–100. https://doi.org/10.1016/j.carbpol.2014.11.004
Kumar A, Negi YS, Choudhary V, Bhardwaj NK (2014) Characterization of cellulose nanocrystals produced by acid-hydrolysis from sugarcane bagasse as agro-waste. J Mater Phys Chem 2:1–8
Lavanya D, Kulkarni PK, Dixit M, Raavi PK, Krishna LNV (2011) Sources of cellulose and their applications—A review. Int J Drug Form Res 2:19–38
Nam S, French AD, Condon BD, Concha M (2016) Segal crystallinity index revisited by the simulation of X-ray diffraction patterns of cotton cellulose Iβ and cellulose II. Carbohyd Polym 135:1–9. https://doi.org/10.1016/j.carbpol.2015.08.035
Nelson ML, O’Connor RT (1964) Relation of certain infrared bands to cellulose crystallinity and crystal lattice type. Part I. Spectra of types I, II, III and of amorphous cellulose. J Appl Polym Sci 8:1311–1324. https://doi.org/10.1002/app.1964.070080322
O’Connor RT, DuPré EF, Mitcham D (1958) Applications of infrared absorption spectroscopy to investigations of cotton and modified cottons: Part I—Physical and crystalline modifications and oxidations. Text Res J 28:382–392. https://doi.org/10.1177/004051755802800503
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK (2010) Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels 3:1–10
Popescu CM, Larsson PT, Olaru N, Vasile C (2012) Spectroscopic study of acetylated kraft pulp fibers. Carbohyd Polym 88:530–536. https://doi.org/10.1016/j.carbpol.2011.12.046
Puleo AC, Paul DR, Kelley SS (1989) The effect of degree of acetylation on gas sorption and transport behavior in cellulose acetate. J Memb Sci 47:301–332. https://doi.org/10.1016/S0376-7388(00)83083-5
Rabelo SC, Andrade RR, Maciel Filho R, Costa AC (2014) Alkaline hydrogen peroxide pretreatment, enzymatic hydrolysis and fermentation of sugarcane bagasse to ethanol. Fuel 136:349–357. https://doi.org/10.1016/j.fuel.2014.07.033
Ramadoss G, Muthukumar K (2015) Influence of dual salt on the pretreatment of sugarcane bagasse with hydrogen peroxide for bioethanol production. Chem Eng J 260:178–187. https://doi.org/10.1016/j.fuel.2014.07.033
Rezende CA, Lima MA, Maziero P, Azevedo ER, Garcia W, Polikarpov I (2011) Chemical and morphological characterization of sugarcane bagasse submitted to delignification process for enhanced enzymatic digestibility. Biotechnol Biofuels 4:1–18. https://doi.org/10.1186/1754-6834-4-54
Rocha GJM, Nascimento VM, Rossell CEV (2014) Caracterização físico-química do bagaço da cana-de-açúcar. Technical Memorandum, CTBE. http://8k5sc3kntvi25pnsk2f69jf1.wpengine.netdna-cdn.com/wp-content/uploads/2016/08/MeT-102014-port.pdf
Rodrigues Filho G, Cruz SF, Pasquini D, Cerqueira DA, Prado VDS, Assunção RMN (2000) Water flux through cellulose triacetate films produced from heterogeneous acetylation of sugar cane bagasse. J Memb Sci 177:225–231. https://doi.org/10.1016/S0376-7388(00)00469-5
Rodrigues Filho G, Monteiro DS, Meireles CS, Assunção RMN, Cerqueira DA, Barud HS, Ribeiro SJL, Messadeq Y (2008) Synthesis and characterization of cellulose acetate produced from recycled newspaper. Carbohyd Polym 73:74–82. https://doi.org/10.1016/j.carbpol.2007.11.010
Samios E, Dart RK, Dawkins JV (1997) Preparation, characterization and biodegradation studies on cellulose acetates with varying degrees of substitution. Polymer 38:3045–3054. https://doi.org/10.1016/S0032-3861(96)00868-3
Scherrer P (1918) Bestimmung der Größe und der inneren Struktur von Kolloidteilchen mittels Röntgenstrahlen. Nach Gesel Wis Göt 26:98–100. https://doi.org/10.1007/978-3-662-33915-2_7
Segal L, Creely JJ, Martin AE Jr, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text Res J 29:786–794. https://doi.org/10.1177/004051755902901003
Shaikh HM, Pandare KV, Nair G, Varma AJ (2009) Utilization of sugarcane bagasse cellulose for producing cellulose acetates: Novel use of residual hemicellulose as plasticizer. Carbohyd Polym 76:23–29. https://doi.org/10.1016/j.carbpol.2008.09.014
Silverstein RM, Webster FX, Kiemle DJ (2005) Spectrometric identification of organic compounds, 8th edn. Wiley, New York
Singh RK, Singh AK (2013) Optimization of reaction conditions for preparing carboxymethyl cellulose from corn cobic agricultural waste. Waste Biomass Valoriz 4:129–137. https://doi.org/10.1007/s12649-012-9123-9
Sluiter JB, Chum H, Gomes AC, Tavares RPA, Azevedo V, Mello LAM, Pimenta MTB, Rabelo SC, Marabezi K, Curvelo AAS, Scolfaro M, Alves AR, Garcia WT, Carvalho W, Esteves PJ, Mendonça S, Oliveira PA, Ribeiro JAA, Mendes TD, Vicentin MP, Duarte CL, Mori MN (2016) Evaluation of Brazilian sugarcane bagasse characterization: an interlaboratory comparison study. J AOAC Int 99:579–585. https://doi.org/10.5740/jaoacint.15-0063
Sun JX, Sun XF, Sun RC, Su YQ (2004) Fractional extraction and structural characterization of sugarcane bagasse hemicelluloses. Carbohyd Polym 56:195–204. https://doi.org/10.1016/j.carbpol.2004.02.002
Testova L, Nieminena K, Penttilä PA, Serimaa R, Potthast A, Sixta H (2014) Cellulose degradation in alkaline media upon acidic pretreatment and stabilisation. Carbohydr Polym 100:185–194. https://doi.org/10.1016/j.carbpol.2013.01.093
Thakur VK, Thakur MK, Kessler (2017) Handbook of composites from renewable materials: polymeric composites, vol 1, 6th edn. Wiley, Hoboken. https://doi.org/10.1002/9781119441632
Vochozka M, Marousková A, Váchal J, Straková J (2016) Economic and environmental aspects of steam-explosion pretreatment. Waste Biomass Valoriz 7:1549–1554. https://doi.org/10.1007/s12649-016-9555-8
Wei WQ, Wu SB (2017) Conversion of eucalyptus cellulose into 5-hydroxymethylfurfural using Lewis acid catalyst in biphasic solvent system. Waste Biomass Valoriz 8:1303–1311. https://doi.org/10.1007/s12649-016-9676-0
Wright L, Boundy B, Perlack B, Davis S, Saulsbury B (2006) Biomass Energy Data Book, 1st edn. Oak Ridge National Laboratory, Tennessee
Acknowledgments
The authors would like to thank CAPES for the financial support, as well as the Northeastern Center of NMR Application and Use (CENAUREMN) for providing NMR analysis, the Chemistry Post-Graduate Program of the UFRN and the Green Chemistry Nucleus (NQV) of Salvador University (Bahia, Brazil).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Moura, H.O.M.A., Campos, L.M.A., da Silva, V.L. et al. Investigating acid/peroxide-alkali pretreatment of sugarcane bagasse to isolate high accessibility cellulose applied in acetylation reactions. Cellulose 25, 5669–5685 (2018). https://doi.org/10.1007/s10570-018-1991-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-018-1991-0