Abstract
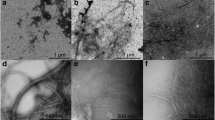
Cellulose nanofibrils (CNFs), unique and promising natural materials have gained significant attention recently for biomedical applications, due to their special biomechanical characteristics, surface chemistry, good biocompatibility and low toxicity. However, their long bio-persistence in the organism may provoke immune reactions and this aspect of CNFs has not been studied to date. Therefore, the aim of this work was to examine and compare the cytocompatibility and immunomodulatory properties of CNFs in vitro. CNFs (diameters of 10–70 nm; lengths of a few microns) were prepared from Norway spruce (Picea abies) by mechanical fibrillation and high pressure homogenisation. L929 cells, rat thymocytes or human peripheral blood mononuclear cells (PBMNCs) were cultivated with CNFs. None of the six concentrations of CNFs (31.25 µg/ml–1 mg/ml) induced cytotoxicity and oxidative stress in the L929 cells, nor induced necrosis and apoptosis of thymocytes and PBMNCs. Higher concentrations (250 µg/ml–1 mg/ml) slightly inhibited the metabolic activities of the L929 cells as a consequence of inhibited proliferation. The same concentrations of CNFs suppressed the proliferation of PBMNCs to phytohemaglutinine, a T-cell mitogen, and the process was followed by down-regulation of interleukin-2 (IL-2) and interferon-γ production. The highest concentration of CNFs inhibited IL-17A, but increased IL-10 and IL-6 production. The secretion of pro-inflammatory cytokines, IL-1β and tumor necrosis factor-α as well as Th2 cytokine (IL-4), remained unaltered. In conclusion, the results suggest that these CNFs are cytocompatible nanomaterial, according to current ISO criteria, with non-inflammatory and non-immunogenic properties. Higher concentrations seem to be tolerogenic to the immune system, a characteristic very desirable for implantable biomaterials.







Similar content being viewed by others
References
Adamson R (2009) Role of macrophages in normal wound healing: an overview. J Wound Care 18:349–351
Alexandrescu L, Syverud K, Gatti A, Chinga-Carrasco G (2013) Cytotoxicity tests of cellulose nanofibril-based structures. Cellulose 20:1765–1775
Backdahl H, Helenius G, Bodin A, Nannmark U, Johansson BR, Risberg B, Gatenholm P (2006) Mechanical properties of bacterial cellulose and interactions with smooth muscle cells. Biomaterials 27:2141–2149
Bhattacharya M, Malinen MM, Lauren P, Lou YR, Kuisma SW, Kanninen L, Lille M, Corlu A, GuGuen-Guillouzo C, Ikkala O, Laukkanen A, Urtti A, Yliperttula M (2012) Nanofibrillar cellulose hydrogel promotes three-dimensional liver cell culture. J Control Release 164:291–298
Boyman O, Sprent J (2012) The role of interleukin-2 during homeostasis and activation of the immune system. Nat Rev Immunol 12:180–190
Chen Y, Junger WG (2012) Measurement of oxidative burst in neutrophils. Methods Mol Biol 844:115–124
Clift MJ, Foster EJ, Vanhecke D, Studer D, Wick P, Gehr P, Rothen-Rutishauser B, Weder C (2011) Investigating the interaction of cellulose nanofibers derived from cotton with a sophisticated 3D human lung cell coculture. Biomacromolecules 12:3666–3673
Colic M, Gasic S, Vucevic D, Pavicic L, Popovic P, Jandric D, Medic-Mijacevic L, Rakic L (2000) Modulatory effect of 7-thia-8-oxoguanosine on proliferation of rat thymocytes in vitro stimulated with concanavalin A. Int J Immunopharmacol 22:203–212
Čolić M, Gazivoda D, Vučević D, Vasilijić S, Rudolf R, Lukić A (2009) Proinflammatory and immunoregulatory mechanisms in periapical lesions. Mol Immunol 47:101–113
Čolić M, Rudolf R, Stamenković D, Anžel I, Vučević D, Jenko M, Lazić V, Lojen G (2010) Relationship between microstructure, cytotoxicity and corrosion properties of a Cu–Al–Ni shape memory alloy. Acta Biomater 6:308–317
Deng DX, Cai L, Chakrabarti S, Cherian MG (1999) Increased radiation-induced apoptosis in mouse thymus in the absence of metallothionein. Toxicology 134:39–49
Dong S, Hirani AA, Colacino KR, Lee YW, Roman M (2012) Cytotoxicity and cellular uptake of cellulose nanocrystals. Nano Life 02:1241006
Dugan JM, Gough JE, Eichhorn SJ (2013) Bacterial cellulose scaffolds and cellulose nanowhiskers for tissue engineering. Nanomedicine 8:287–298
Ferraz N, Carlsson DO, Hong J, Larsson R, Fellstrom B, Nyholm L, Stromme M, Mihranyan A (2012) Haemocompatibility and ion exchange capability of nanocellulose polypyrrole membranes intended for blood purification. J R Soc Interface R Soc 9:1943–1955
Ferraz N, Leschinskaya A, Toomadj F, Fellström B, Strømme M, Mihranyan A (2013) Membrane characterization and solute diffusion in porous composite nanocellulose membranes for hemodialysis. Cellulose 20:2959–2970
Habibi Y (2014) Key advances in the chemical modification of nanocelluloses. Chem Soc Rev 43:1519–1542
Hua K, Carlsson DO, Ålander E, Lindström T, Strømme M, Mihranyan A, Ferraz N (2014) Translational study between structure and biological response of nanocellulose from wood and green algae. RSC Adv 4:2892
ISO10993-5 (2009) Biological evaluation of medical devices. Part 5. Test for in vitro cytotoxicity
Jia B, Li Y, Yang B, Xiao D, Zhang S, Rajulu AV, Kondo T, Zhang L, Zhou J (2013) Effect of microcrystal cellulose and cellulose whisker on biocompatibility of cellulose-based electrospun scaffolds. Cellulose 20:1911–1923
Jonoobi M, Aitomäki Y, Mathew AP, Oksman K (2014) Thermoplastic polymer impregnation of cellulose nanofibre networks: morphology, mechanical and optical properties. Compos A 58:30–35
Kalia S, Boufi S, Celli A, Kango S (2013) Nanofibrillated cellulose: surface modification and potential applications. Colloid Polym Sci 292:5–31
Kolakovic R, Peltonen L, Laukkanen A, Hirvonen J, Laaksonen T (2012) Nanofibrillar cellulose films for controlled drug delivery. Eur J Pharm Biopharm 82:308–315
Kollar P, Zavalova V, Hosek J, Havelka P, Sopuch T, Karpisek M, Tretinova D, Suchy P Jr (2011) Cytotoxicity and effects on inflammatory response of modified types of cellulose in macrophage-like THP-1 cells. Int Immunopharmacol 11:997–1001
Lin N, Dufresne A (2014) Nanocellulose in biomedicine: current status and future prospect. Eur Polym J 59:302–325
Liu K, Lin X, Chen L, Huang L, Cao S (2014) Dual-functional chitosan–methylisothiazolinone/microfibrillated cellulose biocomposites for enhancing antibacterial and mechanical properties of agar films. Cellulose 21:519–528
Long SA, Buckner JH (2011) CD4+FOXP3+ T regulatory cells in human autoimmunity: more than a numbers game. J Immunol 187:2061–2066
Lou YR, Kanninen L, Kuisma T, Niklander J, Noon LA, Burks D, Urtti A, Yliperttula M (2014) The use of nanofibrillar cellulose hydrogel as a flexible three-dimensional model to culture human pluripotent stem cells. Stem Cells Dev 23:380–392
Martins NCT, Freire CSR, Pinto RJB, Fernandes SCM, Neto CP, Silvestre AJD, Causio J, Baldi G, Sadocco P, Trindade T (2012) Electrostatic assembly of Ag nanoparticles onto nanofibrillated cellulose for antibacterial paper products. Cellulose 19:1425–1436
Mathew AP, Oksman K, Pierron D, Harmad M-F (2011) Crosslinked fibrous composites based on cellulose nanofibers and collagen with in situ pH induced fibrillation. Cellulose 19:139–150
Mathew AP, Oksman K, Pierron D, Harmand M-F (2012) Fibrous cellulose nanocomposite scaffolds prepared by partial dissolution for potential use as ligament or tendon substitutes. Carbohydr Polym 87:2291–2298
Mathew AP, Oksman K, Pierron D, Harmand MF (2013) Biocompatible fibrous networks of cellulose nanofibres and collagen crosslinked using genipin: potential as artificial ligament/tendons. Macromol Biosci 13:289–298
Mihajlovic D, Vucevic D, Chinou I, Colic M (2014) Royal jelly fatty acids modulate proliferation and cytokine production by human peripheral blood mononuclear cells. Eur Food Res Technol 238:881–887
Miyamoto T, Takahashi S, Ito H, Inagaki H, Noishiki Y (1989) Tissue biocompatibility of cellulose and its derivatives. J Biomed Mater Res 23:125–133
Monopoli MP, Walczyk D, Campbell A, Elia G, Lynch I, Bombelli FB, Dawson KA (2011) Physical–chemical aspects of protein corona: relevance to in vitro and in vivo biological impacts of nanoparticles. J Am Chem Soc 133:2525–2534
Moreira S, Silva NB, Almeida-Lima J, Rocha HAO, Medeiros SRB, Alves C, Gama FM (2009) BC nanofibres: in vitro study of genotoxicity and cell proliferation. Toxicol Lett 189:235–241
Muhle H, Ernst H, Bellmann B (1997) Investigation of the durability of cellulose fibres in rat lungs. Ann Occup Hyg 41(Supplement 1):184–188
O’Flynn K, Krensky AM, Beverley PC, Burakoff SJ, Linch DC (1985) Phytohaemagglutinin activation of T cells through the sheep red blood cell receptor. Nature 313:686–687
Ouyang W, Rutz S, Crellin NK, Valdez PA, Hymowitz SG (2011) Regulation and functions of the IL-10 family of cytokines in inflammation and disease. Annu Rev Immunol 29:71–109
Pereira MM, Raposo NR, Brayner R, Teixeira EM, Oliveira V, Quintao CC, Camargo LS, Mattoso LH, Brandao HM (2013) Cytotoxicity and expression of genes involved in the cellular stress response and apoptosis in mammalian fibroblast exposed to cotton cellulose nanofibers. Nanotechnology 24:075103
Pitkänen M, Kangas H, Laitinen O, Sneck A, Lahtinen P, Peresin MS, Niinimäki J (2014) Characteristics and safety of nano-sized cellulose fibrils. Cellulose 21:3871–3886
Rahman I, Kode A, Biswas SK (2007) Assay for quantitative determination of glutathione and glutathione disulfide levels using enzymatic recycling method. Nat Protoc 1:3159–3165
Ryšavá J, Dyr JE, Homola J, Dostálek J, Křı́žová P, Mášová L, Suttnar J, Briestenský J, Santar I, Myška K, Pecka M (2003) Surface interactions of oxidized cellulose with fibrin(ogen) and blood platelets. Sens Actuators B 90:243–249
Tada H, Shiho O, Kuroshima K-I, Koyama M, Tsukamoto K (1986) An improved colorimetric assay for interleukin 2. J Immunol Methods 93:157–165
Tatrai E, Brozik M, Adamis Z, Meretey K, Ungvary G (1996) In vivo pulmonary toxicity of cellulose in rats. J Appl Toxicol 16:129–135
Tomic S, Dokic J, Vasilijic S, Ogrinc N, Rudolf R, Pelicon P, Vucevic D, Milosavljevic P, Jankovic S, Anzel I, Rajkovic J, Rupnik MS, Friedrich B, Colic M (2014) Size-dependent effects of gold nanoparticles uptake on maturation and antitumor functions of human dendritic cells in vitro. PLoS One 9:e96584
Valo H, Kovalainen M, Laaksonen P, Hakkinen M, Auriola S, Peltonen L, Linder M, Jarvinen K, Hirvonen J, Laaksonen T (2011) Immobilization of protein-coated drug nanoparticles in nanofibrillar cellulose matrices—enhanced stability and release. J Control Release 156:390–397
Vartiainen J, Pöhler T, Sirola K, Pylkkänen L, Alenius H, Hokkinen J, Tapper U, Lahtinen P, Kapanen A, Putkisto K, Hiekkataipale P, Eronen P, Ruokolainen J, Laukkanen A (2011) Health and environmental safety aspects of friction grinding and spray drying of microfibrillated cellulose. Cellulose 18:775–786
Wolf J, Rose-John S, Garbers C (2014) Interleukin-6 and its receptors: a highly regulated and dynamic system. Cytokine 70:11–20
Yanamala N, Farcas MT, Hatfield MK, Kisin ER, Kagan VE, Geraci CL, Shvedova AA (2014) In vivo evaluation of the pulmonary toxicity of cellulose nanocrystals: a renewable and sustainable nanomaterial of the future. ACS Sustain Chem Eng 2:1691–1698
Zhu J, Yamane H, Paul WE (2010) Differentiation of effector CD4 T cell populations (*). Annu Rev Immunol 28:445–489
Acknowledgments
This work was supported by the project MNT-Era-Net n-POSSCOG (Slovenian Ministry of Education, Science, Culture and Sport), and the project ON175102 (Ministry of Education, Science and Technological Development of R. Serbia). The authors thank Sergej Tomic, Jelena Djokic and Milan Markovic for technical support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Čolić, M., Mihajlović, D., Mathew, A. et al. Cytocompatibility and immunomodulatory properties of wood based nanofibrillated cellulose. Cellulose 22, 763–778 (2015). https://doi.org/10.1007/s10570-014-0524-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-014-0524-8