Abstract
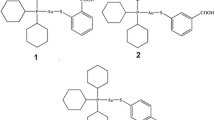
Based on the recent studies depicting the potential of heterometallic gold complexes as potent antiproliferative agents, herein we first reported the preliminary mechanistic data on the in-vitro antiproliferative activity of tricyclohexylphosphanegold(I) n-mercaptobenzoate, Cy3PAu(n-MBA) where n = 2 (1), 3 (2) and 4 (3), and MBA = mercaptobenzoic acid, treated using MCF-7 breast cancer and A2780 ovarian cancer cells, respectively. 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay was used to assess the cytotoxicity of both cancer cells treated with 1–3, respectively. The IC50 of 1–3 were applied to the subsequent assays including cell invasion and thioredoxin reductase (TrxR) as well as ubiquitin activities specifically on Lys48 and Lys63-linked polyubiquitin chains via flowcytometric analysis. The mechanistic effect of 1–3 towards both cells were evaluated on human p53 signaling gene expressions via RT2 profiler Polymerase Chain Reductase (PCR) array. 1–3 were found to be highly cytotoxic towards both MCF-7 and A2780 cancer cell lines with the compounds were more sensitive towards the latter cells. 1–3 also suppressed TrxR and cell invasion activities by modulating p53 related genes related with proliferation, invasion and TrxR activities i.e. CCNB1, TP53, CDK4 etc. 1–3 also regulated Lys48 and Lys63-linked polyubiquitination by reactivation of p53, suggesting the ability of this gene in regulating inhibition of cytoskeletal reorganization via epithelial–mesenchymal transition (EMT), required for tumor progression. Taken together, the overall findings denoted that 1–3 exerted potent antiproliferative activity in MCF-7 and A2780 cells via activation of the p53 signaling pathway.





Similar content being viewed by others
References
Almutlaq BA, Almuazzi RF, Almuhayfir AA, Alfouzan AM, Alshammari BT, AlAnzi HF, Ahmed HG (2017) Breast cancer in Saudi Arabia and its possible risk factors. J Cancer Policy 12:83–89
Berners-Price SJ, Girard GR, Hill DT, Sutton BM, Jarrett PS, Faucette LF, Johnson RK, Mirabelli CK, Sadler PJ (1990) Cytotoxicity and antitumor activity of some tetrahedral bis(diphosphino)gold(I) chelates. J Med Chem 33:1386–1392
Bhatia M, McGrath KL, Di Trapani G, Charoentong P, Shah F, King MM, Clarke FM, Tonissen KF (2016) The thioredoxin system in breast cancer cell invasion and migration. Redox Biol 8:68–78
Boulikas T, Vougiouka M (2003) Cisplatin and platinum drugs at themolecular level. Oncol Rep 10:1663–1682
Brezden CB, Phillips KA, Abdolell M, Bunston T, Tannock IF (2000) Cognitive function in breast cancer patients receiving adjuvant chemotherapy. J Clin Oncol 18:2695–2701
Brooks CL, Gu W (2011) p53 regulation by ubiquitin. FEBS Lett 585:2803–2809
Chaudière J, Tappel AL (1984) Interaction of gold(I) with the active site of selenium-glutathione peroxidase. J Inorg Biochem 20:313–325
Cookson PD, Tiekink ERT (1992) Syntheses and structural studies of triorganophosphinegold(I) mercaptobenzoate complexes. J Coord Chem 26:313–320
Daigle DJ, Pepperman AB Jr, Vail SL (1974) Synthesis of a monophosphorus analog of hexamethylenetetramine. J Heterocyclic Chem 11:407
Darensbourg DJ, Ortiz CG, Kamplain JW (2004) A new water-soluble phosphine derived from 1,3,5-triaza-7-phosphaadamantane (PTA),† 3,7-diacetyl-1,3,7-triaza-5-phosphabicyclo[3.3.1]nonane. Structural, bonding, and solubility properties. Organometallics 23:1747
de Vos D, Clements P, Pyke SM, Smyth DR, Tiekink ERT (1999) Characterisation and in vitro cytotoxicity of triorganophosphinegold(I) 2-mercaptobenzoate complexes. Met Based Drugs 8:303–306
de Vos D, Smyth DR, Tiekink ER (2002) Cytotoxicity of triorganophosphinegold(I) n-mercaptobenzoates, n = 2, 3 and 4. Met-Based Drugs 8:303–306
de Vos D, Ho SY, Tiekink ER (2004) Cytotoxicity profiles for a series of triorganophosphinegold(I) dithiocarbamates and triorganophosphinegold(I) xanthates. Bioinorg Chem Appl 2:141–154
Donzelli E, Carfi M, Miloso M, Strada A, Galbiati A, Bayssas M, Griffon-Etienne G, Caveletti G (2004) Neurotoxicity of platinum compounds: comparison of the effects of cisplatin and oxaliplatin on the human neuroblastoma cell line SH-SY5Y. J Neurooncol 67:65–73
Elie BT, Levine C, Ubarretxena-Belandia I, Varela-Ramírez A, Aguilera RJ, Ovalle R, Contel M (2009) Water soluble phosphane–gold(I) complexes. Applications as recyclable catalysts in a three-component coupling reaction and as antimicrobial and anticancer agents. Eur J Inorg Chem 23:3421–3430
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136:E359–E386
Fricker SP (2010) Cysteine proteases as targets for metal-based drugs. Metallomics 2:366–377
Gandin V, Fernandes AP, Rigobello MP, Dani B, Sorrentino F, Tisato F, Bjornstedt M, Bindoli A, Sturaro A, Rella R, Marzano C (2010) Cancer cell: death induced by phosphine gold(I) compounds targeting thioredoxin reductase. Biochem Pharmacol 15:90–101
Gunatilleke SS, Barrios AM (2006) Inhibition of lysosomal cysteine proteases by a series of Au(I) complexes: a detailed mechanistic investigation. J Med Chem 49:3933–3937
Hammond-Martel I, Yu H, Affar B (2012) Roles of ubiquitin signaling in transcription regulation. Cell Signal 24:410–421
Harris S, Levine AJ (2005) The p53 pathway: positive and negative feedback loops. Oncogene 24:2899–2908
Jain AK, Barton MC (2010) Making sense of ubiquitin ligases that regulate p53. Cancer Biol Ther 10:665–672
Jamaludin NS, Goh ZJ, Cheah YK, Ang KP, Sim JH, Khoo CH, Fairuz ZA et al (2013) Phosphanegold(I) dithiocarbamates, R3PAu[SC(=S)N((i)Pr)CH2CH2OH] for R = Ph, Cy and Et: role of phosphane-bound R substituents upon in vitro cytotoxicity against MCF-7R breast cancer cells and cell death pathways. Eur J Med Chem 67:127–141
Jin S, Levine AJ (2001) The p53 functional circuit. J Cell Sci 114:4139–4140
Karataş OF, Sezgin E, Aydin O, Culha M (2009) Interaction of gold nanoparticles with mitochondria. Colloids Surf B 71:315–318
Laine A, Topisirovic I, Zhai D, Reed JC, Borden KL, Ronai Z (2006) Regulation of p53 localization and activity by Ubc13. Mol Cell Biol 26:8901–8913
Le Cam L, Linares K, Paul C, Julien E, Lacroix M, Hatchi E, Triboulet R, Bossis G, Shmueli A, Rodriguez MS, Coux O, Sardet C (2006) E4F1 is an atypical ubiquitin ligase that modulates p53 effector functions independently of degradation. Cell 127:775–788
Lima JC, Rodriguez L (2011) Phosphine-gold(I) compounds as anticancer agents: general description and mechanisms of action. Anticancer Agents Med Chem 11:921–928
Liu B, Chen Y, St. Clair DK (2008) ROS and p53: versatile partnership. Free Radic Biol Med 44:1529–1535
Marzo T, Cirri D, Gabbiani C, Gamberi T, Magherini F, Messori L et al (2020) Auranofin, Et3PAuCl, and Et3PAuI are highly cytotoxic on colorectal cancer cells: a chemical and biological study. ACS Med Chem Lett 8:997–1001
Menendez D, Shatz M, Resnick MA (2013) Interactions between the tumor suppressor p53 and immune responses. Curr Opin Oncol 25:85–92
Meundaeng N, Rujiwatra A, Prior TJ (2016) Polymorphism in metal complexes of thiazole-4-carboxylic acid. Transit Met Chem 41:783–793
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Muniyappa H, Das KC (2008) Activation of c-Jun N-terminal kinase (JNK) by widely used specific p38 MAPK inhibitor SB202190 and SB203580: A MLK-3-MKK7-dependent mechanism. Cell Signal 20:675–683
Muñoz-Fontela C, Mandinova A, Aaronson SA, Lee SW (2016) Emerging roles of p53 and other tumour-suppressor genes in immune regulation. Nat Rev Immunol 16:741–750
Nathan JA, Kim HT, Ting L, Gygi SP, Goldberg AL (2013) Why do cellular proteins linked to K63-polyubiquitin chains not associate with proteasomes? EMBO J 32:552–565
Nishinaka Y, Nakamura H, Masutani H, Yodoi J (2001) Redox control of cellular function by thioredoxin: a new therapeutic direction in host defense. Arch Immunol Ther Exp 49:285–292
Qian Y, Chen X (2010) Tumor suppression by p53: making cells senescent. Histol Histopathol 25:515–526
Rufini A, Tucci P, Celardo I, Melino G (2013) Senescence and aging: the critical roles of p53. Oncogene 32:5129–5143
Sasada T, Sono H, Yodoi J (1996) Thioredoxin/adult T-cell leukemia-derived factor (ADF) and redox regulation. J Toxicol Sci 21:285–287
Schaeffer D, Somarelli JA, Hanna G, Palmer GM, Garcia-Blanco MA (2014) Cellular migration and invasion uncoupled: increased migration is not an inexorable consequence of epithelial-to-mesenchymal transition. Mol Cell Biol 34:3486–3499
Selenius M, Hedman M, Brodin D, Gandin V, Rigobello MP, Flygare J, Fernandes AP et al (2012) Effects of redox modulation by inhibition of thioredoxin reductase on radiosensitivity and gene expression. J Cell Mol Med 16:1593–1605
Sharma HK, Chhangte L, Dolui AK (2001) Traditional medicinal plants in Mizoram, India. Fitoterapia 72:146–161
Smyth DR, Vincent BR, Tiekink ERT (2001) Polymorphism in (tricyclohexylphosphine)gold(I) 2-mercaptobenzoate. a tale of two structural motifs. Crystal Growth Design 1:113–117
Stegh AH (2012) Targeting the p53 signaling pathway in cancer therapy - the promises, challenges and perils. Exp Opin Ther Targets 16:67–83. https://doi.org/10.1517/14728222.2011.643299
Sui X, Zhu J, Tang H, Wang C, Zhou J, Han W, Wang X, Fang Y, Xu Y, Li D, Chen R, Ma J, Jing Z, Gu X, Pan H, He C (2015) p53 controls colorectal cancer cell invasion by inhibiting the NF-κB-mediated activation of Fascin. Oncotarget 6:22869–22879
Swatek KN, Komander D (2016) Ubiquitin modifications. Cell Res 26:399–422
Taguchi T, Nazneen A, Abid MR, Razzaque MS (2005) Cisplatin associated nephrotoxicity and pathological events. Contrib Nephrol 14:107–121
Thiery JP (2002) Epithelial-mesenchymal transitions in tumour progression. Nat Rev Cancer 2:442–454
Tiekink ERT (2002) Gold derivatives for the treatment of cancer. Crit Rev Oncol Hematol 42:225–248
Tiekink ERT (2003) Gold compounds in medicine: potential anti-tumour agents. Gold Bull 36:117–124
Topisirovic I, Siddiqui N, Orolicki S, Skrabanek LA, Tremblay M, Hoang T, Borden KLB (2009) Stability of eukaryotic translation initiation factor 4E mRNA is regulated by HuR, and this activity is dysregulated in cancer. Mol Cell Biol 29:1152–1162
Trudu F, Amato F, Vaňhara P, Pivetta T, Peña-Méndez EM, Havel J (2015) Coordination compounds in cancer: past, present and perspectives. J Appl Biomed 13:79–103
Varughese S, Kiran KSRN, Solanko KA, Bond AD, Desiraju GR (2011) Interaction anisotropy and shear instability of aspirin polymorphs established by nanoindentation. Chem Sci 2:2236–2242
Vogelstein B, Lane D, Levine AJ (2000) Surfing the p53 network. Nature 408:307–310
Walther W, Althagafi D, Curran D, O’Beirne C, Mc Carthy C, Ott I, Tacke M et al (2020) In-vitro and in-vivo investigations into the carbene-gold anticancer drug candidates NHC*-Au-SCSNMe2 and NHC*-Au-S-GLUC against advanced prostate cancer PC3. Anticancer Drugs 31:672–683
Wetzel C, Kunz PC, Kassack MU, Hamacher A, Böhler P, Watjen W, Ott I, Rubbiani R, Spingler B (2011) Gold(I) complexes of water-soluble diphos-type ligands: synthesis, anticancer activity, apoptosis and thioredoxin reductase inhibition. Dalton Trans 40:9212–9220
Yokomizo A, Ono M, Nanri H, Makino Y, Ohga T, Wada M (1995) Cellular levels of thioredoxin associated with drug sensitivity to cisplatin, mitomycin C, doxorubicin and etoposide. Cancer Res 55:4293–4296
Zitvogel L, Galluzzi L, Smyth MJ, Kroemer G (2013) Mechanism of action of conventional and targeted anticancer therapies: reinstating immunosurveillance. Immunity 39:74–88
Acknowledgements
This research was funded by Ministry of Higher Education, Malaysia, Grant Numbers UM.C/HIR-MOHE/SC/03 and UM.C/HIR-MOHE/SC/12. We thanked Universiti Malaya for providing the grants under High Impact Research Schemes and collaborated with us by providing the compounds and the funding. We also thanked Prof Dr Noor Saadah Abd Rahman, the Deputy Vice Chancellor (Research and Innovation) at the University of Malaya for giving us the consent to publish the data
Author information
Authors and Affiliations
Contributions
Conceptualization, RAH; methodology, RAH; validation, AKP, CPF and RAH; formal analysis, AKP and CPF; investigation, AKP and CPF; data curation, AKP; writing—original draft preparation, AKP; writing—review and editing, RAH; visualization, AKP and CPF; supervision, RAH; project administration, RAH. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ang, K.P., Chan, P.F. & Hamid, R.A. Antiproliferative activity exerted by tricyclohexylphosphanegold(I) n-mercaptobenzoate against MCF-7 and A2780 cell lines: the role of p53 signaling pathways. Biometals 34, 141–160 (2021). https://doi.org/10.1007/s10534-020-00269-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-020-00269-7