Abstract
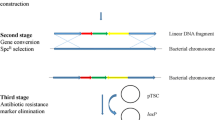
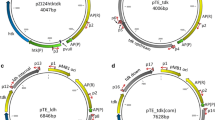
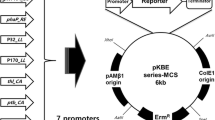
The shuttle vector, pUL6erm, was constructed by using a replicon from pL2, a multiple cloning site, colE1 ori, the ori of Gram-negative bacteria from vector pUC19, and the erythromycin resistance gene from pVA838 as a selection marker. pUL6erm could be transformed easily and maintained stably in Lactococcus lactis, Streptococcus thermophilus, Lactobacillus plantarum and Lactobacillus casei. Transformation assays of pUL6erm indicated that it had a narrow host range. β-Glucuronidase was induced in the presence of 0.3 M NaCl and 50 mM glutamate and expressed at 2.4 U mg−1 with the expression vector (pUL6erm–gadR–GUS) constructed based on pUL6erm carrying β-glucuronidase gene wuth a chloride-inducible (gadR) expression cassette using Pgad as promoter. Therefore, pUL6erm and pUL6erm–gadR–GUS might be a safe and useful genetic tool for the improvement of lactic acid bacteria.





Similar content being viewed by others
References
Alegre MT, Rodriguez MC, Mesas JM (2005) Nucleotide sequence, structural organization and host range of pRS4, a small cryptic Pediococcus pentosaceus plasmid that contains two cassettes commonly found in other lactic acid bacteria. FEMS Microbiol Lett 250:151–156
An HY, Miyamoto T (2006) Cloning and sequence of plasmid pLC494 isolated from human intestinal Lactobacillus casei: construction of an Escherichia coli–Lactobacillus shuttle vector. Plasmid 55:128–134
Anderson DG, McKay LL (1983) Simple and rapid method for isolating large plasmid DNA from lactic streptococci. Appl Environ Microbiol 46:549–552
Beltramo C, Oraby M, Burel G et al (2004) A new vector, pGID052, for genetic transfer in Oenococcus oeni. FEMS Microbiol Lett 236:53–60
Chang SM, Yan TR (2007) DNA sequence analysis of a cryptic plasmid pL2 from Lactococcus lactis subsp. lactis. Biotechnol Lett 29:1519–1527
Chang SM, Tsai CL, Wee WC, Yan TR (2013) Isolation and functional study of potentially probiotic Lactobacilli from Taiwan traditional paocai. Afr J Microbiol Res 7:683–691
de Vos WM (1999) Gene expression systems for lactic acid bacteria. Curr Opin Microbiol 2:289–295
Gasson MJ (1983) Plasmid complements of Streptococcus lactis NCDO 712 and other lactic streptococci after protoplast-induced curing. J Bacteriol 154:1–9
Gosalbes MJ, Esteban CD, Galan JL, Perez-Martinez G (2000) Integrative food-grade expression system based on the lactose regulon of Lactobacillus casei. Appl Environ Microbiol 66:4822–4828
Gravesen A, Josephsen J, von Wright A, Vogensen FK (1995) Characterization of the replicon from the lactococcal theta-replicating plasmid pJW563. Plasmid 34:105–118
Holo H, Nes IF (1989) High-frequency transformation, by electroporation, of Lactococcus lactis subsp. cremoris grown with glycine in osmotically stabilized media. Appl Environ Microbiol 55:3119–3123
Inoue H, Nojima H, Okayama H (1990) High efficiency transformation of Escherichia coli with plasmids. Gene 96:23–28
Jefferson RA, Burgess SM, Hirsh D (1986) β-Glucuronidase from Escherichia coli as a gene-fusion marker. Proc Natl Acad Sci USA 83:8447–8451
Klijn A, Moine D, Delley M et al (2006) Construction of a reporter vector for analysis of Bifidobacterium longum promoters. Appl Environ Microbiol 72:6401–7405
Marugg JD, Meijer W, Van Kranenburg R et al (1995) Medium-dependent regulation of proteinase gene expression in Lactocccus lactis: control of transcription initiation by specific dipeptides. J Bacteriol 177:2982–2989
Park WJ, Lee KH, Lee JM et al (2004) Characterization of pC7 from Lactobacillus paraplantarum C7 derived from Kimchi and development of lactic acid bacteria–Escherichia coli shuttle vector. Plasmid 52:84–88
Perez-Arellano I, Zuniga M, Perez-Martinez G (2001) Construction of compatible wide-host-range shuttle vector for lactic acid bacteria and Escherichia coli. Plasmid 46:106–116
Platteeuw C, Simons G, de Vos WM (1994) Use of the Escherichia coli β-glucuronidase (gusA) gene as a reporter gene for analyzing promoters in lactic acid bacteria. Appl Environ Microbiol 60:587–593
Pouwels PH, Leer RJ (1993) Genetics of lactobacilli: plasmids and gene expression. Antonie Van Leeuwenhoek 64:85–107
Projan SJ, Carleton S, Novick RP (1983) Determination of plasmid copy number by fluorescence densitometry. Plasmid 9:182–190
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, New York
Sander JW, Venema G, Kok J (1997) A chloride-inducible gene expression cassette and its use in induced lysis of Lactococcus lactis. Appl Environ Microbiol 63:4877–4882
Takala TM, Saris PEJ (2002) A food-grade cloning vector for lactic acid bacteria based on the nisin immunity gene nisI. Appl Microbiol Biotechnol 59:467–471
Takamatsu D, Osaki M, Sekizaki T (2001) Construction and characterization of Streptococcus suis–Escherichia coli shuttle cloning vector. Plasmid 45:101–113
Wright A, Raty K (1993) The nucleotide sequence for the replication region of pSV40, a lactococcal food grade cloning vector. Lett Appl Microbiol 17:25–28
Xiong AS, Peng RH, Zhuang J et al (2007) Directed evolution of beta-galactosidase from Escherichia coli into beta-glucuronidase. J Biochem Mol Biol 40:419–425
Yan TR, Wang MR, Chen CH (1998) A facile PCR method for detecting replication mode of lactococcial plasmids. Biotechnol Tech 12:85–89
Acknowledgments
This work was supported by Grants NSC 90-2214-E-036-005 and NSC 91-2214-E-036-001 from the National Science Council, Republic of China.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chang, SM., Yan, TR. Genetic engineering techniques for lactic acid bacteria: construction of a stable shuttle vector and expression vector for β-glucuronidase. Biotechnol Lett 36, 327–335 (2014). https://doi.org/10.1007/s10529-013-1363-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-013-1363-7