Abstract
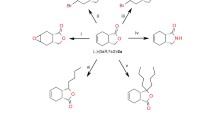
Both R- and S-phenylethyl alcohol of high enantiomeric purity (98%) and with a satisfactory yield (40–80%) were obtained by bioreduction of acetophenone, catalyzed by whole cells of baker’s yeast.
Similar content being viewed by others
References
C Barbieri L Bossi P D’Arrigo GP Fantoni S Servi (2001) ArticleTitleBioreduction of aromatic ketones: preparation of chiral benzyl alcohols in both enantiomeric forms J. Mol. Catal B: Enzymatic 11 415–421 Occurrence Handle1:CAS:528:DC%2BD3MXptlSrsQ%3D%3D
D Buisson X Baucherel E Levoirier S Juge (2000) ArticleTitleBaker’s yeast reduction of α-alkyl-α-hydroxy-β-keto esters Tetrahedron Lett. 41 1389–1392 Occurrence Handle10.1016/S0040-4039(99)02274-1 Occurrence Handle1:CAS:528:DC%2BD3cXhslSnu7g%3D
E Davoli A Forni I Moretti F Prati G Torre (1999) ArticleTitle(R)-(+) and (S)-(-) ethyl 4,4,4-trifluoro-3-hydroxy butanoate by enantioselective baker’s yeast reduction Enzyme Microb. Technol. 25 149–152 Occurrence Handle10.1016/S0141-0229(99)00025-3 Occurrence Handle1:CAS:528:DyaK1MXks1Wrurs%3D
EPS Filho JAR Rodriques PJS Moran (2001) ArticleTitleEffect of additives on the bioreduction of 2-chloro-1-phenyl-2-propen-1-one by baker’s yeast J. Mol. Catal B: Enzymatic 15 23–28 Occurrence Handle10.1016/S1381-1177(00)00249-6 Occurrence Handle1:CAS:528:DC%2BD3MXmt1Kksr4%3D
DR Griffin F Yang G Carta JL Gainer (1998) ArticleTitleAsymmetric reduction of acetophenone with calcium-alginate-entrapped baker’s yeast in organic solvents Biotechnol. Prog. 14 588–593 Occurrence Handle10.1021/bp9800586 Occurrence Handle1:CAS:528:DyaK1cXkvFKgtrc%3D Occurrence Handle9694680
Y Kodama M Imoto M Ohta A S Kitani Ito (2001) ArticleTitleSelective reduction of acetophenone to 1-phenylethanol in aqueous media J. Electroanal. Chem. 507 103–109 Occurrence Handle10.1016/S0022-0728(01)00436-3 Occurrence Handle1:CAS:528:DC%2BD3MXkvFyitLc%3D
K Nakamura S Kondo Y Kawai N Nakajima A Ohno (1994) ArticleTitlePurification and characterization of α-ketoester reductases from baker’s yeast Biosci. Biotechnol. Biochem. 58 2236–2240 Occurrence Handle1:CAS:528:DyaK2MXjtFWqsr0%3D
M Wills M Gamble M Palmer A Smith J Studley J Kenny (1999) ArticleTitleNovel catalysts for asymmetric reduction of carbonyl groups J. Mol. Catal A: Chemical 146 139–148 Occurrence Handle10.1016/S1381-1169(99)00062-X Occurrence Handle1:CAS:528:DyaK1MXns1SntLo%3D
E Żymańczyk-Duda M Brzezińska-Rodak B Lejczak (2004) ArticleTitleStereochemical control of asymmetric hydrogen transfer employing five different kinds of fungi in anhydrous hexane Enzyme Microb. Technol. 34 578–582
Author information
Authors and Affiliations
Corresponding author
Additional information
Revisions requested 29 November 2005; Revisions received 9 January 2006
Rights and permissions
About this article
Cite this article
Brzezińska-Rodak, M., Żymańczyk-Duda, E., Klimek-Ochab, M. et al. A Simple and Green Procedure for the Microbial Effective Synthesis of 1-phenylethyl Alcohol in Both Enantiomeric Forms. Biotechnol Lett 28, 511–513 (2006). https://doi.org/10.1007/s10529-006-0009-4
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10529-006-0009-4