Abstract
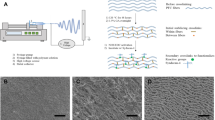
There are recent reports of hybrid tissue–fabric materials with good performance—high biocompatibility and high mechanical strength. In this study, we demonstrate the capability of a hybrid material as a long-term filter for blood proteins. Polyester fabrics were implanted into rats to fabricate hybrid tissue–fabric material sheets. The hybrid materials comprised biological tissue grown on the fabric. The materials were extracted from the rat’s body, approximately 100 days post-implantation. The tissues were decellularized to prevent immunological rejection. An antithrombogenicity test was performed by dropping blood onto the hybrid material surface. The hybrid material showed lesser blood coagulation than polysulfone and cellulose. Blood plasma was filtered using the hybrid material to evaluate the protein removal percentage and the lifetime of the hybrid material in vitro. The hybrid material showed a comparable performance to conventional filters for protein removal. Moreover, the hybrid material could work as a protein filter for 1 month, which is six times the lifetime of polysulfone.






Similar content being viewed by others
References
Chandran, K. B., D. Gao, G. Han, H. Baraniewski, and J. D. Corson. Finite-element analysis of arterial anastomoses with vein, Dacron and PTFE grafts. Med. Biol. Eng. Comput. 30:413–418, 1992.
Dungel, P., N. Long, B. Yu, Y. Moussy, and F. Moussy. Study of the effects of tissue reactions on the function of implanted glucose sensors. J. Biomed. Mater. Res. A 85(3):699–706, 2008.
Funamoto, S., K. Nam, T. Kimura, A. Murakoshi, Y. Hashimoto, K. Niwaya, and A. Kishida. The use of high-hydrostatic pressure treatment to decellularize blood vessels. Biomaterials 31:3590–3595, 2010.
Fung, Y. C. Biomechanics: Mechanical Properties of Living Tissues (2nd ed.). New York: Springer, 1993.
Gough, D. A., L. S. Kumosa, T. L. Routh, J. T. Lin, and J. Y. Lucisano. Function of an implanted tissue glucose sensor for more than 1 year in animals. Sci. Transl. Med. 2:42–53, 2010.
Honge JL, Funder J, Hansen E, Dohmen PM, Konertz W, Hasenkam JM. Recellularization of aortic valves in pigs. Eur. J. Cardiothorac. Surg. 39:829–834, 2011.
Iatridis, J. C., J. Wu, J. A. Yandow, and H. M. Langevin. Subcutaneous tissue mechanical behavior is linear and viscoelastic under uniaxial tension. Connect. Tissue Res. 44:208–217, 2003.
Ishihara, K., H. Fujita, T. Yoneyama, and Y. Iwasaki. Antithrombogenic polymer alloy composed of 2-methacryloyloxyethyl phosphorylcholine polymer and segmented polyurethane. J. Biomater. Sci. Polym. Ed. 11:1183–1195, 2000.
Jia, W. Z., K. Wang, and X. H. Xia. Elimination of electrochemical interferences in glucose biosensors. TrAC Trends Anal. Chem. 29:306–318, 1994.
Ju, Y. M., B. Yu, L. West, Y. Moussy, and F. Moussy. A novel porous collagen scaffold around an implantable biosensor for improving biocompatibility. II. Long term in vitro/in vivo sensitivity characteristics of sensors with NDGA or GA crosslinked collagen scaffolds. J. Biomed. Mater. Res. A 92:650–658, 2010.
Kaltenbrunner, M., T. Sekitani, J. Reeder, T. Yokota, K. Kuribara, T. Tokuhara, and T. Someya. An ultra-lightweight design for imperceptible plastic electronics. Nature 499:458–463, 2013.
Kawase, Y., Y. Inoue, T. Isoyama, I. Saito, H. Nakagawa, T. Ono, and H. Kumagai. Development of hybrid blood removal cannula use of titan rigid for ventricular assist device (VAD). Seitai Ikougaku 51:R-229, 2013.
Kim, W. G., J. K. Park, and W. Y. Lee. Tissue-engineered heart valve leaflets: an effective method of obtaining acellularized valve xenografts. Int. J. Artif. Organs 25:791–797, 2002.
Kingshott, P., H. Thissen, and H. J. Griesser. Effects of cloud-point grafting, chain length, and density of PEG layers on competitive adsorption of ocular proteins. Biomaterials 23:2043–2056, 2002.
Kishi, A., T. Isoyama, I. Saito, H. Miura, H. Nakagawa, A. Kouno, and M. Noshiro. Use of in vivo insert molding to form a jellyfish valve leaflet. Artif. Organs 34(12):1125–1131, 2010.
Le Tissier, P., J. P. Stoye, Y. Takeuchi, C. Patience, and R. A. Weiss. Two sets of human-tropic pig retrovirus. Nature 389:681–682, 1997.
Leypoldt, J. K. Fouling of ultrafiltration and hemodialysis membranes by plasma proteins. Blood Purif. 12:285–291, 1994.
Liu, G. F., Z. J. He, D. P. Yang, X. F. Han, T. F. Guo, C. G. Hao, and C. L. Nie. Decellularized aorta of fetal pigs as a potential scaffold for small diameter tissue engineered vascular graft. Chin. Med. J. (Engl.) 121:1398–1406, 2008.
Morais, J. M., F. Papadimitrakopoulos, and D. J. Burgess. Biomaterials/tissue interactions: possible solutions to overcome foreign body response. AAPS J. 12:188–196, 2010.
Murase, Y., Y. Narita, H. Kagami, K. Miyamoto, Y. Ueda, M. Ueda, and T. Murohara. Evaluation of compliance and stiffness of decellularized tissues as scaffolds for tissue-engineered small caliber vascular grafts using intravascular ultrasound. ASAIO J. 52:450–455, 2006.
Nakayama, Y., and T. Tsujinaka. Acceleration of robust “Biotube” vascular graft fabrication by in-body tissue architecture technology using a novel eosin Y-releasing mold. J. Biomed. Mater. Res. B Appl. Biomater. 102(2):231–238, 2014.
Nakayama, Y., S. Yamaoka, M. Yamanami, M. Fujiwara, M. Uechi, K. Takamizawa, and H. Yaku. Water-soluble argatroban for antithrombogenic surface coating of tissue-engineered cardiovascular tissues. J. Biomed. Mater. Res. B Appl. Biomater. 99:420–430, 2011.
Nauli, A. M., Y. Sun, J. D. Whittimore, S. Atyia, G. Krishnaswamy, and S. M. Nauli. Chylomicrons produced by Caco-2 cells contained ApoB-48 with diameter of 80–200 nm. Physiol. Rep. 2:e12018, 2014.
Nishida, K., M. Sakakida, K. Ichinose, T. Uemura, M. Uehara, K. Kajiwara, and N. Nakabayashi. Development of a ferrocene-mediated needle-type glucose sensor covered with newly designed biocompatible membrane, 2-methacryloyloxyethyl phosphorylcholine-co-n-butyl methacrylate. Med. Prog. Technol. 21:91–103, 1995.
Novak, M. T., F. Yuan, and W. M. Reichert. Modeling the relative impact of capsular tissue effects on implanted glucose sensor time lag and signal attenuation. Anal. Bioanal. Chem. 398:1695–1705, 2010.
Onuki, Y., U. Bhardwaj, F. Papadimitrakopoulos, and D. J. Burgess. A review of the biocompatibility of implantable devices: current challenges to overcome foreign body response. J. Diabetes Sci. Technol. 2:1003–1015, 2008.
Patience, C., Y. Takeuchi, and R. A. Weiss. Infection of human cells by an endogenous retrovirus of pigs. Nat. Med. 3:282–286, 1997.
Patil, S., N. F. Martinez, J. R. Lozano, and R. Garcia. Force microscopy imaging of individual protein molecules with sub-pico Newton force sensitivity. J. Mol. Recognit. 20:516–523, 2007.
Quinn, C. P., C. P. Pathak, A. Heller, and J. A. Hubbell. Photo-crosslinked copolymers of 2-hydroxyethyl methacrylate, poly(ethylene glycol) tetra-acrylate and ethylene dimethacrylate for improving biocompatibility of biosensors. Biomaterials 16:389–396, 1995.
Ranieri, J. P., D. L. Hern-Anderson, A. M. J. Gonin, and B. K. McIlroy Processed ratite carotid arteries as xenogeneic small bore vascular grafts. U.S. Patent No. 20,020,077,697, 2002.
Reach, G., and G. S. Wilson. Can continuous glucose monitoring be used for the treatment of diabetes? Anal. Chem. 64:381A–386A, 1992.
Reddy, S. M., and P. M. Vagama. Surfactant-modified poly(vinyl chloride) membranes as biocompatible interfaces for amperometric enzyme electrodes. Anal. Chim. Acta 350:77–89, 1997.
Reichert, W. M., and S. S. Saavedra. Materials consideration in the selection, performance, and adhesion of polymeric encapsulants for implantable sensors. Mater. Sci. Technol. 64(6):A381–A386, 1992.
Reichert, W. M. and A. A. Sharkawy. Biosensors. In: Handbook of Biomaterials Evaluation: Scientific, Technical, and Clinical Testing of Implant Materials. Philadelphia: Taylor & Francis, pp. 439–459, 1999.
Sakai, O., K. Kanda, K. Takamizawa, T. Sato, H. Yaku, and Y. Nakayama. Faster and stronger vascular “Biotube” graft fabrication in vivo using a novel nicotine-containing mold. J. Biomed. Mater. Res. B Appl. Biomater. 90:412–420, 2009.
Shaw, G. W., D. J. Claremont, and J. C. Pickup. In vitro testing of a simply constructed, highly stable glucose sensor suitable for implantation in diabetic patients. Biosens. Bioelectron. 6:401–406, 1991.
Sheller, N. B., S. Petrash, M. D. Foster, and V. V. Tsukruk. Atomic force microscopy and X-ray reflectivity studies of albumin adsorbed onto self-assembled monolayers of hexadecyltrichlorosilane. Langmuir 14:4535–4544, 1998.
Tanaka, M., T. Hayashi, and S. Morita. The roles of water molecules at the biointerface of medical polymers. Polym. J. 45:701–710, 2013.
Tanaka, M., T. Motomura, M. Kawada, T. Anzai, Y. Kasori, T. Shiroya, K. Shimura, and A. Mochizuki. Blood compatible aspects of poly(2-methoxyethylacrylate)(PMEA)—relationship between protein adsorption and platelet adhesion on PMEA surface. Biomaterials 21(14):1471–1481, 2000.
Tinkilic, N., O. Cubuk, and I. Isildak. Glucose and urea biosensors based on all solid-state PVC–NH2 membrane electrodes. Anal. Chim. Acta 452:29–34, 2002.
Toscano, A., and M. M. Santore. Fibrinogen adsorption on three silica-based surfaces: conformation and kinetics. Langmuir 22:2588–2597, 2006.
Vadgama, P. M., and G. Reach. Advances in Biosensors: Chemical Sensors for In Vivo Monitoring. London: JAI Press, 1993.
Valdes, T. I., and F. Moussy. A ferric chloride pre-treatment to prevent calcification of Nafion membrane used for implantable biosensors. Biosens. Bioelectron. 14(6):579–585, 1999.
Wisniewski, N., F. Moussy, and W. M. Reichert. Characterization of implantable biosensor membrane biofouling. Fresenius J. Anal. Chem. 366:611–621, 2000.
Xu, Y., G. Zhang, Y. Chang, Y. X. Qiu, and C. Wang. The preparation of acellular dermal matrices by freeze-thawing and ultrasonication process and the evaluation of its antigenicity. Cell Biochem. Biophys. 73(1):27–33, 2015.
Yokota, T., K. Kuribara, T. Tokuhara, U. Zschieschang, H. Klauk, K. Takimiya, and T. Someya. Flexible low-voltage organic transistors with high thermal stability at 250°C. Adv. Mater. 25:3639–3644, 2013.
Yokota, T., T. Sekitani, T. Tokuhara, N. Take, U. Zschieschang, H. Klauk, and T. Someya. Sheet-type flexible organic active matrix amplifier system using pseudo-CMOS circuits with floating-gate structure. Electron. Devices 59:3434–3441, 2012.
Zhang, S., Y. Benmakroha, P. Rolfe, T. Shinobu, and I. Kazuhiko. Development of a haemocompatible pO2 sensor with phospholipid-based copolymer membrane. Biosens. Bioelectron. 11:1019–1029, 1996.
Acknowledgments
The authors gratefully acknowledge Dr. Yoshinori Mitamura and Hidemoto Nakagawa for their helpful in-depth discussions on this study. This study was supported by JST ERATO. The strength test of hybrid material was supported by the cooperation program of research institutes in Tohoku University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Thurmon E. Lockhart oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Inoue, Y., Yokota, T., Sekitani, T. et al. Antithrombotic Protein Filter Composed of Hybrid Tissue-Fabric Material has a Long Lifetime. Ann Biomed Eng 45, 1352–1364 (2017). https://doi.org/10.1007/s10439-016-1781-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-016-1781-5