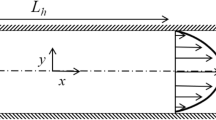
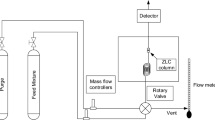
Abstract
Aqueous diffusivities and solubilities of NO were determined by using a chemiluminescence detector to measure time-dependent fluxes across stagnant liquid films confined between polydimethylsiloxane membranes. The NO diffusivities in pure water and PBS at 25∘C were found to be (2.21 ± 0.02) × 10−5 cm2 s−1 and (2.21 ± 0.04) × 10−5 cm2 s−1, respectively. Although lower than most previous values for NO at room temperature, these diffusivities are very similar to those for O2, as one would expect. Extrapolation to 37°C yielded a value of 3.0 × 10−5 cm2 s−1. The solubility of NO in water at 25°C was (1.94 ± 0.03) × 10−6 mol cm−3 atm−1, in agreement with the literature. This agreement, along with the excellent fits obtained to the transient flux data (<4% rms error in each experiment), supports the validity of the simultaneously measured diffusivity. The solubility of NO in PBS at 25°C was (1.75 ± 0.02) × 10−6 mol cm−3 atm−1. The modest (10%) reduction in solubility relative to that in pure water is consistent with the usual effects of salts on gas solubilities.
Similar content being viewed by others
References
Armor, J. N. Influence of pH and ionic-strength upon solubility of NO in aqueous solution. J. Chem. Eng. Data. 19:82–84, 1974.
Barrie, J. A., and D. Machin. The sorption and diffusion of water in silicone rubbers. Part II. Filled rubbers. J. Macromol. Sci. Phys. 4:673–692, 1969.
Butler, A. R., I. L. Megson, and P. G. Wright. Diffusion of nitric oxide and scavenging by blood in the vasculature. Biochim. Biophys. Acta. 1425:168–76, 1998.
Chen, B., M. Keshive, and W. M. Deen. Diffusion and reaction of nitric oxide in suspension cell cultures. Biophys. J. 75:745–754, 1998.
Chen, B., and W. M. Deen. Analysis of the effects of cell spacing and liquid depth on nitric oxide and its oxidation products in cell cultures. Chem. Res. Toxicol. 14:135–147, 2001.
Chen, B., and W. M. Deen. Effect of liquid depth on the synthesis and oxidation of nitric oxide in macrophage cultures. Chem. Res. Toxicol. 15:490–496, 2002.
Dean, J. A. (ed.) Lange’s Handbook of Chemistry, 15th ed. New York: McGraw-Hill, 1999, p. 5.7.
Denicola, A., J. M. Souza, R. Radi, and E. Lissi. Nitric oxide diffusion in membranes determined by fluorescence quenching. Arch. Biochem. Biophys. 328:208–12, 1996.
Goldstick, T. K., and I. Fatt. Diffusion of oxygen in solutions of blood proteins. Chem. Eng. Prog. Symp. Ser. 66:101–113, 1970.
Gros, G., W. Moll, H. Hoppe, and H. Gros. Proton transport by phosphate diffusion—a mechanism of facilitated CO2 transfer. J. Gen. Physiol. 67:773–790, 1976.
Hermann, C., I. Dewes, and A. Schumpe. The estimation of gas solubilities in salt solutions. Chem. Eng. Sci. 50:1673–1675, 1995.
Himmelblau, D. M. Diffusion of dissolved gases in liquids. Chem. Rev. 64:527–550, 1964.
Ho, C. S., L. K. Ju, R. F. Baddour, and D. I. C. Wang. Simultaneous measurement of oxygen diffusion coefficients and solubilities in electrolyte solutions with a polarographic oxygen electrode. Chem. Eng. Sci. 43:3093–3107, 1988.
Jordan, J., E. Ackerman, and R. L. Berger. Polarographic diffusion coefficients of oxygen defined by activity gradients in viscous media. J. Am. Chem. Soc. 78:2979–2983, 1956.
Jordan, J., and W. E. Bauer. Correlations between solvent structure, viscosity and polarographic diffusion coefficients of oxygen. J. Am. Chem. Soc. 81:3915–3919, 1959.
Lamers-Lemmers, J., L. J. C. Hoofd, and B. Oeseburg. Non-steady-state O2 diffusion in metmyoglobin solutions studied in a diffusion chamber. Biochem. Biophys. Res. Commun. 276:773–778, 2000.
Lancaster, J. R., Jr. Diffusion of free nitric oxide. Methods Enzymol. 268:31–50, 1996.
Lewis, R. S., W. M. Deen, S. R. Tannenbaum, and J. S. Wishnok. Membrane mass spectrometer inlet for quantitation of nitric oxide. Biol. Mass. Spectrom. 22:45–52, 1993.
Lewis, R. S., and W. M. Deen. Kinetics of the reaction of nitric oxide with oxygen in aqueous solutions. Chem. Res. Toxicol. 7:568–574, 1994.
Lide, D. R. (ed.) Handbook of Chemistry and Physics, 71st edn., Boca Raton: CRC Press, 1990–1991, p. 6–151.
Massey, L. K. Permeability Properties of Plastics and Elastomers—A Guide to Packaging and Barrier Materials, 2nd edn. Norwich: William Andrew Publishing/Plastics Design Library, 2003, Ch. 87.
Meldon, J. H. Reaction-Enhanced Mass Transfer in Thin Liquid Films, Sc. D. thesis, Mass. Inst. Of Tech., Cambridge, 1973.
Meldon, J. H., K. A. Smith, and C. K. Colton. Effect of weak acids upon transport of carbon dioxide in alkaline solutions. Chem. Eng. Sci. 32:939–950, 1977.
Nalwaya, N., and W. M. Deen. Analysis of cellular exposure to peroxynitrite in suspension cultures. Chem. Res. Toxicol. 16:920–32, 2003.
Nguyen, Q. T., Z. Bendjama, R. Clement, and Z. H. Ping. Poly(dimethylsiloxane) crosslinked in different conditions—Part II. Pervaporation of water-ethyl acetate mixtures. Phys. Chem. Chem. Phys. 2:395–400, 2000.
Reid, R. C., J. M. Prausnitz, and B. E. Poling. The Properties of Gases and Liquids, 4th ed. New York: McGraw-Hill, 1987, pp. 598–604.
Robb, W. L. Thin silicone membranes–Their permeation properties and some applications. Ann. N. Y. Acad. Sci. 146:138–147, 1968.
Sendroy, J., R. T. Dillon, and D. D. Van Slyke. Studies of gas and electrolyte equilibria in blood. XIX. The solubility and physical state of uncombined oxygen in blood. J. Biol. Chem. 105:597–632, 1934.
Shaw, A. W., and A. J. Vosper. Solubility of nitric oxide in aqueous and non-aqueous solvents. J. Chem. Soc. Faraday Trans. I. 73:1239–1244, 1977.
Stamler, J. S., D. I. Simon, J. A. Osborne, M. E. Mullins, O. Jaraki, T. Michel, D. J. Singel, and J. Loscalzo. S-Nitrosylation of proteins with nitric oxide—synthesis and characterization of biologically-active compounds. Proc. Natl. Acad. Sci. U.S.A. 89:444–448, 1992.
Stroeve, P. Diffusion with Reversible Chemical Reaction in Heterogeneous Media, Sc.D. thesis, Mass. Inst. of Tech., Cambridge, 1973.
Stroeve, P., C. K. Colton, and K. A. Smith. Steady-state diffusion of oxygen in red blood cell and model suspensions. AIChE J. 22:1133–1142, 1976.
Stroeve, P., and E. Ziegler. The transport of carbon dioxide in high molecular-weight buffer solutions. Chem. Eng. Commun. 6:81–103, 1980.
Vanderkooi, J. M., W. W. Wright, and M. Erecinska. Nitric oxide diffusion coefficients in solutions, proteins and membranes determined by phosphorescence. Biochim. Biophys. Acta. 1207:249–54, 1994.
Vaughn, M. W., L. Kuo, and J. C. Liao. Estimation of nitric oxide production and reaction rates in tissue by use of a mathematical model. Am. J. Physiol. 274:H2163–76, 1998.
Wang, C., and W. M. Deen. Nitric oxide delivery system for cell culture studies. Ann. Biomed. Eng. 31:65–79, 2003.
Ward, W. J. Analytical and experimental studies of facilitated transport. AIChE J. 16:405–410, 1970.
Wise, D. L., and G. Houghton. Diffusion coefficients of neon, krypton, xenon, carbon monoxide and nitric oxide in water at 10–60∘C. Chem. Eng. Sci. 23:1211–1216, 1968.
Wood, J., and J. Garthwaite. Models of the diffusional spread of nitric oxide: Implications for neural nitric oxide signaling and its pharmacological properties. Neuropharmacology. 33:1235–1244, 1994.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zacharia, I.G., Deen, W.M. Diffusivity and Solubility of Nitric Oxide in Water and Saline. Ann Biomed Eng 33, 214–222 (2005). https://doi.org/10.1007/s10439-005-8980-9
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10439-005-8980-9