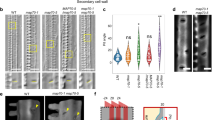
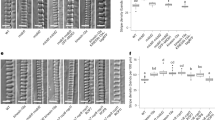
Abstract
The water and mineral conductive tube, the xylem vessel and tracheid, is a highly conspicuous tissue due to its elaborately patterned secondary-wall deposition. One constituent of the xylem vessel and tracheid, the tracheary element, is an empty dead cell that develops secondary walls in the elaborate patterns. The wall pattern is appropriately regulated according to the developmental stage of the plant. The cytoskeleton is an essential component of this regulation. In fact, the cortical microtubule is well known to participate in patterned secondary cell wall formation. The dynamic rearrangement of the microtubules and actin filaments have also been recognized in the cultured cells differentiating into tracheary elements in vitro. There has recently been considerable progress in our understanding of the dynamics and regulation of cortical microtubules, and several plant microtubule associated proteins have been identified and characterized. The microtubules have been observed during tracheary element differentiation in living Arabidopsis thaliana cells. Based on this recently acquired information on the plant cytoskeleton and tracheary element differentiation, this review discusses the role of the cytoskeleton in secondary cell wall formation.



Similar content being viewed by others
References
Baskin TI (2001) On the alignment of cellulose microfibrils by cortical microtubules: a review and a model. Protoplasma 215:150–171
Bichet A, Desnos T, Turner S, Grandjean O, Hofte H (2001) BOTERO1 is required for normal orientation of cortical microtubules and anisotropic cell expansion in Arabidopsis. Plant J 25:137–148
Bierhorst DW, Zamora PM (1965) Primary xylem elements and element associations of angiosperms. Am J Bot 52:657–710
Blancaflor BE, Hasenstein KH (2000) Methods for detection and identification of F-actin in fixed and permeabilized plant tissues. In: Staiger CJ, Baluska F, Volkmann D, Barlow PW (eds) Actin: a dynamic framework for multiple plant cell function. Kluwer, Dordrecht, pp 601–618
Bouquin T, Mattsson O, Naested H, Foster R, Mundy J (2003) The Arabidopsis lue1 mutant defines a katanin p60 ortholog involved in hormonal control of microtubule orientation during cell growth. J Cell Sci 116:791–801
Brower DJ, Hepler PK (1976) Microtubules and secondary wall deposition in xylem: the effects of isopropyl N-phenylcarbamate. Protoplasma 87:91–111
Burk DH, Ye ZH (2002) Alteration of oriented deposition of cellulose microfibrils by mutation of a katanin-like microtubule-severing protein. Plant Cell 14:2145–2160
Burk DH, Liu B, Zhong R, Morrison WH, Ye ZH (2001) A katanin-like protein regulates normal cell wall biosynthesis and cell elongation. Plant Cell 13:807–827
Chaffey, Barlow (2000) Actin in the secondary vascular system of woody plants. In: Staiger CJ, Baluska F, Volkmann D, Barlow PW (eds) Actin: a dynamic framework for multiple plant cell function. Kluwer, Dordrecht, pp 587–600
Chaffey N, Barlow P (2002) Myosin, microtubules, and microfilaments: co-operation between cytoskeletal components during cambial cell division and secondary vascular differentiation in trees. Planta 214:526–536
Chaffey NJ, Barnett JR, Barlow PW (1997) Cortical microtubule involvement in bordered pit formation in secondary xylem vessel elements of Aesculus hippocastanum L. (Hippocastanaceae): a correlative study using electron microscopy and indirect immunofluorescence microscopy. Protoplasma 197:64–75
Chaffey NJ, Barnett JR, Barlow PW (1999) A cytoskeletal basis for wood formation in angiosperm trees: the involvement of cortical microtubules. Planta 208:19–30
Chaffey N, Barlow P, Barnett J (2000) A cytoskeletal basis for wood formation in angiosperm trees: the involvement of microfilaments. Planta 210:890–896
Chan J, Jensen CG, Jensen LC, Bush M, Lloyd CW (1999) The 65-kDa carrot microtubule-associated protein forms regularly arranged filamentous cross-bridges between microtubules. Proc Natl Acad Sci USA 96:14931–14936
Chan J, Mao G, Smertenko A, Hussey PJ, Naldrett M, Bottrill A, Lloyd CW (2003a) Identification of a MAP65 isoform involved in directional expansion of plant cells. FEBS Lett 534:161–163
Chan J, Calder GM, Doonan JH, Lloyd CW (2003b) EB1 reveals mobile microtubule nucleation sites in Arabidopsis. Nat Cell Biol 5:967–971
Chan J, Calder G, Fox S, Lloyd C (2005) Localization of the microtubule end binding protein EB1 reveals alternative pathways of spindle development in Arabidopsis suspension cells. Plant Cell 17:1737–1748
Chang HY, Smertenko AP, Igarashi H, Dixon DP, Hussey PJ (2005) Dynamic interaction of NtMAP65-1a with microtubules in vivo. J Cell Sci 118:3195–3201
Demura T, Tashiro G, Horiguchi G, Kishimoto N, Kubo M, Matsuoka N, Minami A, Nagata-Hiwatashi M, Nakamura K, Okamura Y, Sassa N, Suzuki S, Yazaki J, Kikuchi S, Fukuda H (2002) Visualization by comprehensive microarray analysis of gene expression programs during transdifferentiation of mesophyll cells into xylem cells. Proc Natl Acad Sci USA 99:15794–15799
Dhonukshe P, Gadella TW Jr (2003) Alteration of microtubule dynamic instability during preprophase band formation revealed by yellow fluorescent protein-CLIP170 microtubule plus-end labeling. Plant Cell 15:597–611
Dhonukshe P, Mathur J, Hulskamp M, Gadella TW Jr (2005) Microtubule plus-ends reveal essential links between intracellular polarization and localized modulation of endocytosis during division-plane establishment in plant cells. BMC Biol 3:11. DOI 10.1186/1741-7007-3-11
Dixit R, Cyr R (2004) Encounters between dynamic cortical microtubules promote ordering of the cortical array through angle-dependent modifications of microtubule behavior. Plant Cell 16:3274–3284
Esau K (1977) Anatomy of seed plants, 2nd edn. Wiley, New York
Esau K, Charvat I (1978) On vessel member differentiation in the bean (Phaseolus vulgaris L.). Ann Bot 42:665–677
Fackenthal JD, Turner FR, Raff EC (1993) Tissue-specific microtubule functions in Drosophila spermatogenesis require the β2-tubulin isotype-specific carboxy terminus. Dev Biol 158:213–227
Falconer MM, Seagull RW (1985a) Immunofluorescent and calcofluor white staining of developing tracheary elements in Zinnia elegans L. suspension cultures. Protoplasma 125:190–198
Falconer MM, Seagull RW (1985b) Xylogenesis in tissue culture: taxol effects on microtubule reorientation and lateral association in differentiating cells. Protoplasma 128:157–166
Falconer MM, Seagull RW (1986) Xylogenesis in tissue culture II: Microtubules, cell-shape and secondary wall patterns. Protoplasma 133:140–148
Falconer MM, Seagull RW (1988) Xylogenesis in tissue culture III: continuing wall deposition during tracheary element differentiation. Protoplasma 144:10–16
Fukuda H (1987) A change in tubulin synthesis in the process of tracheary element differentiation and cell-division of isolated Zinnia mesophyll cells. Plant Cell Physiol 28:517–528
Fukuda H (1989) Regulation of tubulin degradation in isolated Zinnia mesophyll cells in culture. Plant Cell Physiol 30:243–252
Fukuda H (1992) Tracheary element formation as a model system of cell differentiation. Int Rev Cytol 136:289–332
Fukuda H (1996) Xylogenesis: initiation, progression, and cell death. Ann Rev Plant Physiol Plant Mol Biol 47:299–325
Fukuda H (2000) Programmed cell death of tracheary elements as a paradigm in plants. Plant Mol Biol 44:245–253
Fukuda H (2004) Signals that control plant vascular cell differentiation. Nat Rev Mol Cell Biol 5:379–391
Fukuda H, Kobayashi H (1989) Dynamic organization of the cytoskeleton during tracheary-element differentiation. Dev Growth Differ 31:9–16
Fukuda H, Komamine A (1980) Establishment of an experimental system for the study of tracheary element differentiation from single cells isolated from the mesophyll of Zinnia elegans. Plant Physiol 65:57–60
Funada R, Abe H, Furusawa O, Imaizumi H, Fukazawa K, Ohtani J (1997) The orientation and localization of cortical microtubules in differentiating conifer tracheids during cell expansion. Plant Cell Physiol 38:210–212
Funada R, Miura H, Shibagaki M, Furusawa O, Miura T, Fukatsu E, Kitin P (2001) Involvement of localized cortical microtubules in the formation of a modified structure of wood. J Plant Res 114:491–497
Gardiner JC, Taylor NG, Turner SR (2003) Control of cellulose synthase complex localization in developing xylem. Plant Cell 15:1740–1748
Granger CL, Cyr RJ (2000) Microtubule reorganization in tobacco BY-2 cells stably expressing GFP-MBD. Planta 210:502–509
Gunning BES, Hardham AR (1982) Microtubules. Annu Rev Plant Biol 33:651–698
Haigler CH, Brown RM (1986) Transport of rosettes from the Golgi apparatus to the plasma membrane in isolated mesophyll cells of Zinnia elegans during differentiation to tracheary elements in suspension culture. Protoplasma 134:111–120
Hardham AR, Gunning BES (1978) Structure of cortical microtubule arrays in plant cells. J Cell Biol 77:14–34
Hardham AR, Gunning BES (1979) Interpolation of microtubules into cortical arrays during cell elongation and differentiation in roots of Azolla pinnata. J Cell Sci 37:411–442
Hardham AR, Gunning BES (1980) Some effects of colchicine on microtubules and cell division in roots of Azolla pinnata. Protoplasma 102:31–51
Hasezawa S, Ueda K, Kumagai F (2000) Time-sequence observations of microtubule dynamics throughout mitosis in living cell suspensions of stable transgenic Arabidopsis: direct evidence for the origin of cortical microtubules at M/G1 interface. Plant Cell Physiol 41:244–250
Hashimoto (2003) Dynamics and regulation of plant interphase microtubules: a comparative view. Curr Opin Plant Biol 6:568–576
Hepler PK (1981) Morphogenesis of tracheary elements and guard cells. Cell Biol Monogr 8:327–347
Hepler PK, Fosket DE (1971) The role of microtubules in vessel member differentiation in Coleus. Protoplasma 72:213–236
Hepler PK, Newcomb EH (1964) Microtubules and fibrils in the cytoplasm of Coleus cells undergoing secondary wall deposition. J Cell Biol 20:529–533
Hogetsu T (1991) Mechanism for formation of the secondary wall thickening in tracheary elements: microtubules and microfibrils of tracheary elements of Pisum sativum L. and Commelina communis L. and the effects of amiprophosmethyl. Planta 185:190–200
Hosokawa M, Suzuki S, Umezawa T, Sato Y (2001) Progress of lignification mediated by intercellular transportation of monolignols during tracheary element differentiation of isolated Zinnia mesophyll cells. Plant Cell Physiol 42:959–968
Hoyle HD, Raff EC (1990) Two Drosophila beta tubulin isoforms are not functionally equivalent. J Cell Biol 111:1009–1026
Ito J, Fukuda H (2002) ZEN1 is a key enzyme in the degradation of nuclear DNA during programmed cell death of tracheary elements. Plant Cell 14:3201–3211
Janson ME, de Dood ME, Dogterom M (2003) Dynamic instability of microtubules is regulated by force. J Cell Biol 161:1029–1034
Ketelaar T, Anthony RG, Hussey PJ (2004) Green fluorescent protein-mTalin causes defects in actin organization and cell expansion in Arabidopsis and inhibits actin depolymerizing factor’s actin depolymerizing activity in vitro. Plant Physiol 136:3990–3998
Kobayashi H, Fukuda H, Shibaoka H (1987) Reorganization of actin-filaments associated with the differentiation of tracheary elements in zinnia mesophyll-cells. Protoplasma 138:69–71
Kobayashi H, Fukuda H, Shibaoka H (1988) Interrelation between the spatial disposition of actin-filaments and microtubules during the differentiation of tracheary elements in cultured zinnia cells. Protoplasma 143:29–37
Korolev AV, Chan J, Naldrett MJ, Doonan JH, Lloyd CW (2005) Identification of a novel family of 70 kDa microtubule-associated proteins in Arabidopsis cells. Plant J 42:547–555
Kost B, Spielhofer P, Chua NH (1998) A GFP-mouse talin fusion protein labels plant actin filaments in vivo and visualizes the actin cytoskeleton in growing pollen tubes. Plant J 16:393–401
Kubo M, Udagawa M, Nishikubo N, Horiguchi G, Yamaguchi M, Ito J, Mimura T, Fukuda H, Demura T (2005) Transcription switches for protoxylem and metaxylem vessel formation. Genes Dev 19:1855–1860
Kumagai F, Yoneda A, Tomida T, Sano T, Nagata T, Hasezawa S (2001) Fate of nascent microtubules organized at the M/G1 interface, as visualized by synchronized tobacco BY-2 cells stably expressing GFP-tubulin: time-sequence observations of the reorganization of cortical microtubules in living plant cells. Plant Cell Physiol 42:723–732
Ledbetter, Porter (1963) A “microtubule” in plant cell fine structure. J Cell Biol 19:239–250
Littauer UZ, Giveon D, Thierauf M, Ginzburg I, Ponstingl H (1986) Common and distinct tubulin binding sites for microtubule-associated proteins. Proc Natl Acad Sci USA 83:7162–7166
Maitra SC, De DN (1971) Role of microtubules in secondary thickening of differentiating xylem element. J Ultra Res 34:15–22
Mao T, Jin L, Li H, Liu B, Yuan M (2005) Two microtubule-associated proteins of the Arabidopsis MAP65 family function differently on microtubules. Plant Physiol 138:654–662
Marc J, Granger CL, Brincat J, Fisher DD, Kao Th, McCubbin AG, Cyr RJ (1998) GFP–MAP4 reporter gene for visualizing cortical microtubule rearrangements in living epidermal cells. Plant Cell 10:1927–1939
Mathur J, Mathur N, Kernebeck B, Srinivas BP, Hulskamp M (2003) A novel localization pattern for an EB1-like protein links microtubule dynamics to endomembrane organization. Curr Biol 13:1991–1997
Milioni D, Sado PE, Stacey NJ, Domingo C, Roberts K, McCann MC (2001) Differential expression of cell-wall-related genes during the formation of tracheary elements in the Zinnia mesophyll cell system. Plant Mol Biol 47:221–238
Milioni D, Sado PE, Stacey NJ, Roberts K, McCann MC (2002) Early gene expression associated with the commitment and differentiation of a plant tracheary element is revealed by cDNA-amplified fragment length polymorphism analysis. Plant Cell 14:2813–2824
Muller S, Smertenko A, Wagner V, Heinrich M, Hussey PJ, Hauser MT (2004) The plant microtubule-associated protein AtMAP65-3/PLE is essential for cytokinetic phragmoplast function. Curr Biol 14:412–417
Murata T, Sonobe S, Baskin TI, Hyodo S, Hasezawa S, Nagata T, Horio T, Hasebe M (2005) Microtubule-dependent microtubule nucleation based on recruitment of γ-tubulin in higher plants. Nat Cell Biol 7:961–968
Nakajima K, Furutani I, Tachimoto H, Matsubara H, Hashimoto T (2004) SPIRAL1 encodes a plant-specific microtubule-localized protein required for directional control of rapidly expanding Arabidopsis cells. Plant Cell 16:1178–1190
Nakamura M, Naoi K, Shoji T, Hashimoto T (2004) Low concentrations of propyzamide and oryzalin alter microtubule dynamics in Arabidopsis epidermal cells. Plant Cell Physiol 45:1330–1334
Nakashima J, Mizuno T, Takabe K, Fujita M, Saiki H (1997) Direct visualization of lignifying secondary wall thickenings in Zinnia elegans cells in culture. Plant Cell Physiol 38:818–827
Nakashima J, Takebe K, Fujita M, Fukuda H (2000) Autolysis during in vitro tracheary element differentiation: formation and location of the perforation. Plant Cell Physiol 41:1267–1271
Nielsen MG, Turner FR, Hutchens JA (2001) Axoneme-specific β-tubulin specialization: a conserved C-terminal motif specifies the central pair. Curr Biol 11:529–533
Obara K, Kuriyama H, Fukuda H (2001) Direct evidence of active and rapid nuclear degradation triggered by vacuole rupture during programmed cell death in Zinnia. Plant Physiol 125:615–626
Oda Y, Mimura T, Hasezawa S (2005) Regulation of secondary cell wall development by cortical microtubules during tracheary element differentiation in Arabidopsis cell suspensions. Plant Physiol 137:1027–1036
Olyslaegers G, Verbelen JP (1998) Improved staining of F-actin and co-localization of mitochondria in plant cells. J Microsc 192:73–77
Pickett-Heaps JD (1966) Incorporation of radioactivity into wheat xylem walls. Planta 71:1–14
Pickett-Heaps JD (1967) The effects of colchicine on the ultrastructure of dividing plant cell, xylem wall differentiation and distribution of cytoplasmic microtubules. Dev Biol 15:206–236
Pickett-Heaps JD, Northcote DH (1966) Relationship of cellular organelles to the formation and development of the plant cell wall. J Exp Bot 17:20–26
Roberts AW, Haigler CH (1994) Cell Expansion and tracheary element differentiation are regulated by extracellular pH in mesophyll cultures of Zinnia elegans L. Plant Physiol 105:699–706
Roberts AW, Uhnak KS (1998) Tip growth in xylogenic suspension cultures of Zinnia elegans L.: implications for the relationship between cell shape and secondary-cell-wall pattern in tracheary elements. Protoplasma 204:103–113
Roberts AW, Frost AO, Roberts EM, Haigler CH (2004) Roles of microtubules and cellulose microfibril assembly in the localization of secondary-cell-wall deposition in developing tracheary elements. Protoplasma 224:217–229
Salnikov VV, Grimson MJ, Delmer DP, Haigler CH (2001) Sucrose synthase localizes to cellulose synthesis sites in tracheary elements. Phytochemistry 57:823–833
Sano T, Higaki T, Oda Y, Hayashi T, Hasezawa S (2005) Appearance of actin microfilament “twin peaks” in mitosis and their function in cell plate formation, as visualized in tobacco BY-2 cells expressing GFP-fimbrin. Plant J 44(4):595–605
Sawa S, Demura T, Horiguchi G, Kubo M, Fukuda H (2005) The ATE genes are responsible for repression of transdifferentiation into xylem cells in Arabidopsis. Plant Physiol 137:141–148
Sedbrook JC (2004) MAPs in plant cells: delineating microtubule growth dynamics and organization. Curr Opin Plant Biol 7:632–640
Sedbrook JC, Ehrhardt DW, Fisher SE, Scheible WR, Somerville CR (2004) The Arabidopsis SKU6/SPIRAL1 gene encodes a plus end-localized microtubule-interacting protein involved in directional cell expansion. Plant Cell 16:1506–1520
Shaw SL, Kamyar R, Ehrhardt DW (2003) Sustained microtubule tread-milling in Arabidopsis cortical arrays. Science 300:1715–1718
Sheahan MB, Staiger CJ, Rose RJ, McCurdy DW (2004) A green fluorescent protein fusion to actin-binding domain 2 of Arabidopsis fimbrin highlights new features of a dynamic actin cytoskeleton in live plant cells. Plant Physiol 136:3968–3978
Smertenko AP, Chang HY, Wagner V, Kaloriti D, Fenyk S, Sonobe S, Lloyd C, Hauser MT, Hussey PJ (2004) The Arabidopsis microtubule-associated protein AtMAP65-1: molecular analysis of its microtubule bundling activity. Plant Cell 16:2035–2047
Takahashi H, Hirota K, Kawahara A, Hayakawa E, Inoue Y (2003) Randomization of cortical microtubules in root epidermal cells induces root hair initiation in lettuce (Lactuca sativa L.) seedlings. Plant Cell Physiol 44:350–359
Taylor JG, Haigler CH (1993) Patterned secondary cell-wall assembly in tracheary elements occurs in a self-perpetuating cascade. Acata Bot Neer 42:153–163
Taylor JG, Owen TP, Koonce LT, Haigler CH (1992) Dispersed lignin in tracheary elements treated with cellulose synthesis inhibitors provides evidence that molecules of the secondary cell-wall mediate wall patterning. Plant J 2:959–970
Taylor NG, Scheible WR, Cutler S, Somerville CR, Turner SR (1999) The irregular xylem3 locus of Arabidopsis encodes a cellulose synthase required for secondary cell wall synthesis. Plant Cell 11:769–779
Taylor NG, Laurie S, Turner SR (2000) Multiple cellulose synthase catalytic subunits are required for cellulose synthesis in Arabidopsis. Plant Cell 12:2529–2539
Taylor NG, Howells RM, Huttly AK, Vickers K, Turner SR (2003) Interactions among three distinct CesA proteins essential for cellulose synthesis. Proc Natl Acad Sci USA 100:1450–1455
Twell D, Park SK, Hawkins TJ, Schubert D, Schmidt R, Smertenko A, Hussey PJ (2002) MOR1/GEM1 has an essential role in the plant-specific cytokinetic phragmoplast. Nat Cell Biol 4:711–714
Tokunaga N, Sakakibara N, Umezawa T, Ito Y, Fukuda H, Sato Y (2005) Involvement of extracellular dilignols in lignification during tracheary element differentiation of isolated Zinnia mesophyll cells. Plant Cell Physiol 46:224–232
Ueda K, Matsuyama T, Hashimoto T (1999) Visualization of microtubules in living cells of transgenic Arabidopsis thaliana. Protoplasma 206:201–206
Uhnak KS, Roberts AW (1995) Microtubule rearrangements accompanying dedifferentiation in mesophyll cultures of Zinnia elegans L. Protoplasma 189:81–87
Van Bruaene N, Joss G, Van Oostveldt P (2004) Reorganization and in vivo dynamics of microtubules during Arabidopsis root hair development. Plant Physiol 136:3905–3919
Van Gestel K, Le J, Verbelen JP (2001) A comparison of F-actin labeling methods for light microscopy in different plant specimens: multiple techniques supplement each other. Micron 32:571–578
Van Damme D, Van Poucke K, Boutant E, Ritzenthaler C, Inze’ D, Geelen D (2004a) In vivo dynamics and differential microtubule-binding activities of MAP65 proteins. Plant Physiol 136:3956–3967
Van Damme D, Bouget FY, Van Poucke K, Inze D, Geelen D (2004b) Molecular dissection of plant cytokinesis and phragmoplast structure: a survey of GFP-tagged proteins. Plant J 40:386–398
Vos JW, Dogterom M, Emons AM (2004) Microtubules become more dynamic but not shorter during preprophase band formation: a possible “search-and-capture” mechanism for microtubule translocation. Cell Motil Cytoskeleton 57:246–258
Wang YS, Motes CM, Mohamalawari DR, Blancaflor EB (2004) Green fluorescent protein fusions to Arabidopsis fimbrin 1 for spatio-temporal imaging of F-actin dynamics in roots. Cell Motil Cytoskeleton 59:79–93
Whittington AT, Vugrek O, Wei KJ, Hasenbein NG, Sugimoto K, Rashbrooke MC, Wasteneys GO (2001) MOR1 is essential for organizing cortical microtubules in plants. Nature 411:610–613
Wicker-Planquart C, Stoppin-Mellet V, Blanchoin L, Vantard M (2004) Interactions of tobacco microtubule-associated protein MAP65-1b with microtubules. Plant J 39:126–134
Wooding FBP, Northcote DH (1964) The development of the secondary wall of the xylem in Acer pseudoplantanus. J Cell Biol 23:327–337
Yasuhara H, Muraoka M, Shogaki H, Mori H, Sonobe S (2002) TMBP200, a microtubule bundling polypeptide isolated from telophase tobacco BY-2 cells is a MOR1 homologue. Plant Cell Physiol 43:595–603
Ye ZH (2002) Vascular tissue differentiation and pattern formation in plants. Annu Rev Plant Biol 53:183–202
Yoshimura T, Demura T, Igarashi M, Fukuda H (1996) Differential expression of three genes for different beta-tubulin isotypes during the initial culture of Zinnia mesophyll cells that divide and differentiate into tracheary elements. Plant Cell Physiol 37:1167–1176
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oda, Y., Hasezawa, S. Cytoskeletal organization during xylem cell differentiation. J Plant Res 119, 167–177 (2006). https://doi.org/10.1007/s10265-006-0260-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-006-0260-8