Abstract
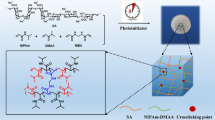
Developing a low-cost and well-recyclable adsorbent with high adsorption capacity is greatly desirable in dye wastewater treatment. Here, we demonstrate a kind of novel tough and reusable hydrogel beads with quite high capacity of dye adsorption via incorporating mussel-bioinspired poly(L-DOPA) (PDOPA) into alginate/poly(acrylamide) double network (DN) hydrogels. The synthesized PDOPA nanoaggregates were introduced into the DN hydrogels by simple one-pot mixing with the monomers prior to polymerization. The fabricated hydrogel beads exhibited high mechanical strength and good elastic recovery due to the interpenetrating Ca2+-alginate and poly(acrylamide) networks. It was shown that the beads exhibited relatively high dye adsorption capacity compared to other adsorbents reported in literature, and the introduction of PDOPA with an appropriate amount raised the adsorption capacity. It is believed that the addition of PDOPA and the matrix of double network architecture contributed synergistically to the high adsorption capacity of hydrogel beads. Moreover, the desorption of dyes could be easily realized via rinsing in acidic water and ethanol solution. The hydrogel beads remained the high adsorption capacity even after 5 times of adsorption and desorption cycles. This tough and stable hydrogel with high adsorption capacity may have potential in treatment of dye wastewater released by textile dyeing industry.
Similar content being viewed by others
References
Boyd, C. E., "Water quality: an introduction", Springer, 2015.
Campos, C. J. A.; Avant, J.; Gustar, N.; Lowther, J.; Powell, A.; Stockley, L.; Lees, D. N. Fate of human noroviruses in shellfish and water impacted by frequent sewage pollution events. Environ. Sci. Tech. 2015, 49, 8377–8385.
Wu, C.; Maurer, C.; Wang, Y.; Xue, S.; Davis, D. L. Water pollution and human health in China. Environ. Health Perspect. 1999, 107, 251–256.
Liu, T. Y.; Bian, L. X.; Yuan, H. G.; Pang, B.; Lin, Y. K.; Tong, Y.; Van der Bruggen, B.; Wang, X. L. Fabrication of a high-flux thin film composite hollow fiber nanofiltration membrane for wastewater treatment. J. Membr. Sci. 2015, 478, 25–36.
Liu, C.; Cheng, L.; Zhao, Y.; Zhu, L. Interfacially crosslinked composite porous membranes for ultrafast removal of anionic dyes from water through permeating adsorption. J. Hazard. Mater. 2017, 337, 217–225.
Fu, F.; Xie, L.; Tang, B.; Wang, Q.; Jiang, S. Application of a novel strategy-advanced Fenton-chemical precipitation to the treatment of strong stability chelated heavy metal containing wastewater. Chem. Eng. J. 2012, 189–190, 283–287.
Ali, I. New generation adsorbents for water treatment. Chem. Rev. 2012, 112, 5073–5091.
Wang, W.; Tade, M. O.; Shao, Z. Research progress of perovskite materials in photocatalysis- and photovoltaics-related energy conversion and environmental treatment. Chem. Soc. Rev. 2015, 44, 5371–5408.
Bansal, R. C.; Goyal, M., "Activated carbon adsorption", CRC press, 2005.
Alver, E.; Metin, A. Ü. Anionic dye removal from aqueous solutions using modified zeolite: Adsorption kinetics and isotherm studies. Chem. Eng. J. 2012, 200, 59–67.
Hua, M.; Zhang, S.; Pan, B.; Zhang, W.; Lv, L.; Zhang, Q. Heavy metal removal from water/wastewater by nanosized metal oxides: a review. J. Hazard. Mater. 2012, 211–212, 317–331.
Morin-Crini, N.; Crini, G. Environmental applications of waterinsoluble β-cyclodextrin-epichlorohydrin polymers. Prog. Polym. Sci. 2013, 38, 344–368.
Gong, J.; Lin, H.; Antonietti, M.; Yuan, J. Nitrogen-doped porous carbon nanosheets derived from poly(ionic liquid)s: hierarchical pore structures for efficient CO2 capture and dye removal. J. Mater. Chem. A 2016, 4, 7313–7321.
Zhu, H.; Yang, X.; Cranston, E. D.; Zhu, S. Flexible and porous nanocellulose aerogels with high loadings of metal-organicframework particles for separations applications. Adv. Mater. 2016, 28, 7652–7657.
Ersan, G.; Kaya, Y.; Apul, O. G.; Karanfil, T. Adsorption of organic contaminants by graphene nanosheets, carbon nanotubes and granular activated carbons under natural organic matter preloading conditions. Sci. Total Environ. 2016, 565, 811–817.
Liang, H. W.; Cao, X.; Zhang, W. J.; Lin, H. T.; Zhou, F.; Chen, L. F.; Yu, S. H. Robust and highly efficient free-standing carbonaceous nanofiber membranes for water purification. Adv. Funct. Mater. 2011, 21, 3851–3858.
Smith, S. C.; Rodrigues, D. F. Carbon-based nanomaterials for removal of chemical and biological contaminants from water: A review of mechanisms and applications. Carbon 2015, 91, 122–143.
Jing, G.; Wang, L.; Yu, H.; Amer, W. A.; Zhang, L. Recent progress on study of hybrid hydrogels for water treatment. Colloids Surf., A 2013, 416, 86–94.
Ju, K. Y.; Lee, Y.; Lee, S.; Park, S. B.; Lee, J. K. Bioinspired polymerization of dopamine to generate melanin-like nanoparticles having an excellent free-radical-scavenging property. Biomacromolecules 2011, 12, 625–632.
Pinnen, F.; Cacciatore, I.; Cornacchia, C.; Sozio, P.; Iannitelli, A.; Costa, M.; Pecci, L.; Nasuti, C.; Cantalamessa, F.; Di Stefano, A. Synthesis and study of l-DOPA-glutathione codrugs as new anti-parkinson agents with free radical scavenging properties. J. Med. Chem. 2007, 50, 2506–2515.
Han, L.; Lu, X.; Liu, K.; Wang, K.; Fang, L.; Weng, L.T.; Zhang, H.; Tang, Y.; Ren, F.; Zhao, C. Mussel-inspired adhesive and tough hydrogel based on nanoclay confined dopamine polymerization. ACS nano 2017, 11, 2561–2574.
Muya, F. N.; Sunday, C. E.; Baker, P.; Iwuoha, E. Environmental remediation of heavy metal ions from aqueous solution through hydrogel adsorption: a critical review. Water Sci. Tech. 2016, 73, 983–992.
Gong, J. P.; Katsuyama, Y.; Kurokawa, T.; Osada, Y. Doublenetwork hydrogels with extremely high mechanical strength. Adv. Mater. 2003, 15, 1155–1158.
Tanaka, Y.; Gong, J. P.; Osada, Y. Novel hydrogels with excellent mechanical performance. Prog. Polym. Sci. 2005, 30, 1–9.
Chen, Q.; Chen, H.; Zhu, L.; Zheng, J. Fundamentals of double network hydrogels. J. Mater. Chem. B 2015, 3, 3654–3676.
Pourjavadi, A.; Nazari, M.; Kabiri, B.; Hosseini, S. H.; Bennett, C. Preparation of porous graphene oxide/hydrogel nanocomposites and their ability for efficient adsorption of methylene blue. RSC Adv. 2016, 6, 10430–10437.
Zhuang, Y.; Yu, F.; Chen, J.; Ma, J. Batch and column adsorption of methylene blue by graphene/alginate nanocomposite: Comparison of single-network and doublenetwork hydrogels. J. Environ. Chem. Eng. 2016, 4, 147–156.
Zhuang, Y.; Yu, F.; Chen, H.; Zheng, J.; Ma, J.; Chen, J. Alginate/graphene double-network nanocomposite hydrogel beads with low-swelling, enhanced mechanical properties, and enhanced adsorption capacity. J. Mater. Chem. A 2016, 4, 10885–10892.
Fan, J.; Shi, Z.; Lian, M.; Li, H.; Yin, J. Mechanically strong graphene oxide/sodium alginate/polyacrylamide nanocomposite hydrogel with improved dye adsorption capacity. J. Mater. Chem. A 2013, 1, 7433.
Deng, S.; Xu, H.; Jiang, X.; Yin, J. Poly(vinyl alcohol) (PVA)-enhanced hybrid hydrogels of hyperbranched poly(ether amine) (hPEA) for selective adsorption and separation of dyes. Macromolecules 2013, 46, 2399–2406.
Xie, Y.; Yan, B.; Xu, H.; Chen, J.; Liu, Q.; Deng, Y.; Zeng, H. Highly regenerable mussel-inspired Fe3O4@polydopamine-Ag core-shell microspheres as catalyst and adsorbent for methylene blue removal. ACS Appl. Mater. Interfaces 2014, 6, 8845–8852.
Yu, L.; Liu, X.; Yuan, W.; Brown, L. J.; Wang, D. Confined flocculation of ionic pollutants by poly(L-DOPA)-based polyelectrolyte complexes in hydrogel beads for threedimensional, quantitative, efficient water decontamination. Langmuir 2015, 31, 6351–6366.
Sun, J. Y.; Zhao, X.; Illeperuma, W. R. K.; Chaudhuri, O.; Oh, K. H.; Mooney, D. J.; Vlassak, J. J.; Suo, Z. Highly stretchable and tough hydrogels. Nature 2012, 489, 133.
Zheng, S.; Wang, T.; Liu, D.; Liu, X.; Wang, C.; Tong, Z. Fast deswelling and highly extensible poly(N-isopropylacrylamide)-hectorite clay nanocomposite cryogels prepared by freezing polymerization. Polymer 2013, 54, 1846–1852.
Jiang, J. H.; Zhu, L. P.; Zhang, H. T.; Zhu, B. K.; Xu, Y. Y. Improved hydrodynamic permeability and antifouling properties of poly (vinylidene fluoride) membranes using polydopamine nanoparticles as additives. J. Membr. Sci. 2014, 457, 73–81.
Zhang, W.; Ying, Y.; Ma, J.; Guo, X.; Huang, H.; Liu, D.; Zhong, C. Mixed matrix membranes incorporated with polydopamine-coated metal-organic framework for dehydration of ethylene glycol by pervaporation. J. Membr. Sci. 2017, 527, 8–17.
Wang, Z.; Wang, D.; Zhang, S.; Hu, L.; Jin, J. Interfacial design of mixed matrix membranes for improved gas separation performance. Adv. Mater 2016, 28, 3399–3405.
Cao, L.; Lv, F.; Liu, Y.; Wang, W.; Huo, Y.; Fu, X.; Sun, R.; Lu, Z. A high performance O2 selective membrane based on CAU-1-NH2@polydopamine and the PMMA polymer for Liair batteries. Chem. Commun. 2015, 51, 4364–4367.
Dong, Z.; Wang, D.; Liu, X.; Pei, X.; Chen, L.; Jin, J. Bioinspired surface-functionalization of graphene oxide for the adsorption of organic dyes and heavy metal ions with a superhigh capacity. J. Mater. Chem. A 2014, 2, 5034–5040.
Pinnen, F.; Cacciatore, I.; Cornacchia, C.; Sozio, P.; Iannitelli, A.; Costa, M.; Pecci, L.; Nasuti, C.; Cantalamessa, F.; Di Stefano, A. Synthesis and study of L-DOPA-glutathione codrugs as new anti-parkinson agents with free radical scavenging properties. J. Med. Chem. 2007, 50, 2506–2515.
Han, L.; Lu, X.; Liu, K.; Wang, K.; Fang, L.; Weng, L. T.; Zhang, H.; Tang, Y.; Ren, F.; Zhao, C.; Sun, G.; Liang, R.; Li, Z. Mussel-inspired adhesive and tough hydrogel based on nanoclay confined dopamine polymerization. ACS Nano 2017, 11, 2561–2574.
Zhao, F. Y.; Ji, Y. L.; Weng, X. D.; Mi, Y. F.; Ye, C. C.; An, Q. F.; Gao, C. J. High-flux positively charged nanocomposite nanofiltration membranes filled with poly(dopamine) modified multiwall carbon nanotubes. ACS Appl. Mater. Interfaces 2016, 8, 6693.
Wang, Z.; Wang, D.; Zhang, S.; Hu, L.; Jin, J. Interfacial design of mixed matrix membranes for improved gas separation performance. Adv. Mater. 2016, 28, 3399–3405.
Wang, W.; Zong, L.; Wang, A. A nanoporous hydrogel based on vinyl-functionalized alginate for efficient absorption and removal of Pb2+ ions. Int. J. Biol. Macromol. 2013, 62, 225–231.
Yang, C. H.; Wang, M. X.; Haider, H.; Yang, J. H.; Sun, J. Y.; Chen, Y. M.; Zhou, J.; Suo, Z. Strengthening alginate/ polyacrylamide hydrogels using various multivalent cations. ACS Appl. Mater. Interfaces 2013, 5, 10418–10422.
Gong, J. P. Why are double network hydrogels so tough? Soft Matter 2010, 6, 2583–2590.
Ng, J. C. Y. Kinetics of pollutant sorption by biosorbents: review. Sep. Purif. Rev. 2000, 29, 189–232.
Zhou, J.; Hao, B.; Wang, L.; Ma, J.; Cheng, W. Preparation and characterization of nano-TiO2/chitosan/poly(N-isopropylacrylamide) composite hydrogel and its application for removal of ionic dyes. Sep. Purif. Technol. 2017, 176, 193–199.
Hassan, A. F.; Abdel-Mohsen, A. M.; Fouda, M. M. G. Comparative study of calcium alginate, activated carbon, and their composite beads on methylene blue adsorption. Carbohydr. Polym. 2014, 102, 192–198.
Ma, T.; Chang, P. R.; Zheng, P.; Zhao, F.; Ma, X. Fabrication of ultra-light graphene-based gels and their adsorption of methylene blue. Chem. Eng. J. 2014, 240, 595–600.
Dragan, E. S.; Apopei Loghin, D. F. Enhanced sorption of methylene blue from aqueous solutions by semi-IPN composite cryogels with anionically modified potato starch entrapped in PAAm matrix. Chem. Eng. J. 2013, 234, 211–222.
Saber-Samandari, S.; Saber-Samandari, S.; Nezafati, N.; Yahya, K. Efficient removal of lead (II) ions and methylene blue from aqueous solution using chitosan/Fe-hydroxyapatite nanocomposite beads. J. Environ. Manage. 2014, 146, 481–490.
Acknowledgments
This work was financialy supported by the National Natural Science Foundation of China (Nos. 51573159 and 51273176) and the Fundamental Research Funds for the Central Universities (No. 2016QNA4032).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Zhang, PB., Tang, AQ., Wang, ZH. et al. Tough poly(L-DOPA)-containing Double Network Hydrogel Beads with High Capacity of Dye Adsorption. Chin J Polym Sci 36, 1251–1261 (2018). https://doi.org/10.1007/s10118-018-2163-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-018-2163-2