Abstract
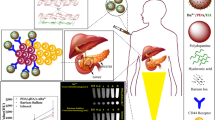
Hyperbranched polysulfonamine (HPSA) is a promising biomaterial due to its highly branched spherical architecture and efficient intracellular translocation. To realize the functionalization of HPSA, both N-succinimidyl 3-(2-pyridyldithio) propionate (SPDP) for tethering the human-mouse chimeric monoclonal antibody CH12 and N-hydroxy succinimidyl S-acetylmercaptoacetyltriglycinate (NHS-MAG3) for labeling 188Re were sequentially grafted onto the primary amine terminals of HPSA via covalent linkages, attaining the SPDP-HPSA-MAG3 intermediate. In order to reserve the structural integrity of CH12, the fragment crystallizable (Fc) region was also processed by oxidation of oligosaccharide moieties with sodium periodate and then reacted with N-(κ-maleimidoundecanoic acid) hydrazide (KMUH). After chelating 188Re with MAG3 group, the SPDP was reduced to PDP and connected onto the maleinimide group at the Fc region. As a result, both the epidermal growth factor receptor vIII (EGFRvIII) targeted monoclonal antibody CH12 and the radionuclide 188Re were conjugated to the HPSA-based vehicles, forming the 188Re-labeled and CH12-tethered HPSA (CH12-HPSA-188Re). The molecular weight and in vitro stability of CH12-HPSA-188Re were evaluated by gel electrophoresis and paper chromatography. On one hand, the CH12-HPSA-188Re could specifically bind to the EGFRvIII-positive human hepatocarcinoma cells in vitro. On the other hand, it could also target at the tumor tissue of nude mice in vivo. Hence, the CH12-HPSA-188Re could effectively target at the human hepatocarcinoma and facilitate the tumor detection and targeted radioimmunotherapy.
Similar content being viewed by others
References
Hamoudeh, M., Kamleh, M.A., Diab, R. and Fessi, H., Adv. Drug Deliv. Rev., 2008, 60: 1329
Janib, S.M., Moses, A.S. and MacKay, J.A., Adv. Drug Deliv. Rev., 2010, 62: 1052
Kim, J.H., Park, K., Nam, H.Y., Lee, S., Kim, K. and Kwon, I.C., Prog. Polym. Sci., 2007, 32: 1031
Srinivas, M., Aarntzen, E.H.J.G., Bulte, J.W.M., Oyen, W.J., Heerschap, A., de Vries, I.J.M. and Figdor, C.G., Adv. Drug Deliv. Rev., 2010, 62: 1080
Boschi, A., Uccelli, L., Duatti, A., Bolzati, C., Refosco, F., Tisato, F., Romagnoli, R., Baraldi, P.G., Varani, K. and Borea, P.A., Bioconjugate Chem., 2003, 14: 1279
Kim, C., Favazza, C. and Wang, L.V., Chem. Rev., 2010, 110: 2756
Schottelius, M., Wester, H.J., Reubi, J.C., Senekowitsch-Schmidtke, R. and Schwaiger, M., Bioconjugate Chem., 2002, 13: 1021
Schweitzer, D., Shearer, J., Rittenberg, D.K., Shoner, S.C., Ellison, J.J., Loloee, R., Lovell, S., Barnhart, D. and Kovacs, J.A., Inorg. Chem., 2002, 41: 3128
Walther, M., Jung, C.M., Bergmann, R., Pietzsch, J., Rode, K., Fahmy, K., Mirtschink, P., Stehr, S., Heintz, A., Wunderlich, G., Kraus, W., Pietzsch, H.J., Kropp, J., Deussen, A. and Spies, H., Bioconjugate Chem., 2006, 18: 216
Schroeder, A., Heller, D.A., Winslow, M.M., Dahlman, J.E., Pratt, G.W., Langer, R., Jacks, T. and Anderson, D.G., Nat. Rev. Cancer, 2012, 12: 39
Fox, M.E., Szoka, F.C. and Fréchet, J.M.J., Acc. Chem. Res., 2009, 42: 1141
Liu, J.Y., Huang, W., Pang, Y., Huang, P., Zhu, X.Y., Zhou, Y.F. and Yan, D.Y., Angew. Chem. Int. Ed., 2011, 50: 9162
Delcea, M., Möhwald, H. and Skirtach, A.G., Adv. Drug Deliv. Rev., 2011, 63: 730
Zhou, Y.F., Huang, W., Liu, J.Y., Zhu, X.Y. and Yan, D.Y., Adv. Mater., 2010, 22: 4567
Calderón, M., Quadir, M.A., Sharma, S.K. and Haag, R., Adv. Mater., 2010, 22: 190
Chen, Y., Zhou, L.Z., Pang, Y., Huang, W., Qiu, F., Jiang, X.L., Zhu, X.Y., Yan, D.Y. and Chen, Q., Bioconjugate Chem., 2011, 22: 1162
Chen, S.Y., Tan, Z.H., Li, N., Wang, R.B., He, L., Shi, Y.F., Jiang, L., Li, P.Y. and Zhu, X.Y., Macromol. Biosci., 2011, 11: 828
Chen, S.Y., Sun, G.M., Jiang, L., Li, N., Li, P.Y. and Zhu, X.Y., Chem. J. Chin. Univer., 2009, 30: 825
Chen, L., Zhu, X.Y., Yan, D.Y., Chen, Y., Chen, Q. and Yao, Y.F., Angew. Chem. Int. Ed., 2006, 118: 93
Yan, D.Y. and Gao, C., Macromolecules, 2000, 33: 7693
Noh, T., Kook, Y.H., Park, C., Youn, H., Kim, H., Oh, E.T., Choi, E.K., Park, H.J. and Kim, C., J. Polym. Sci. Part A: Polym. Chem., 2008, 46: 7321
Boswell, C.A., Tesar, D.B., Mukhyala, K., Theil, F.P., Fielder, P.J. and Khawli, L.A., Bioconjugate Chem., 2010, 21: 2153
Guin, S., Yao, H.P. and Wang, M.H., Mol. Pharm., 2009, 7: 386
Shi, J., Zhi, J., Tong, B., Zhao, W., Shen, J. and Dong, Y., Prog. Chem., 2008, 20: 558
Huang, X., El-Sayed, I.H., Qian, W. and El-Sayed, M.A., J. Am. Chem. Soc., 2006, 128: 2115
Huang, X., El-Sayed, I.H., Qian, W. and El-Sayed, M.A., Nano Lett., 2007, 7: 1591
Hussain, A.F., Kampmeier, F., von Felbert, V., Merk, H.F., Tur, M.K. and Barth, S., Bioconjugate Chem., 2011, 22: 2487
Lee, J., Choi, Y., Kim, K., Hong, S., Park, H.Y., Lee, T., Cheon, G.J. and Song, R., Bioconjugate Chem., 2010, 21: 940
Quiles, S., Raisch, K.P., Sanford, L.L., Bonner, J.A. and Safavy, A., J. Med. Chem., 2009, 53: 586
Wängler, C., Moldenhauer, G., Eisenhut, M., Haberkorn, U. and Mier, W., Bioconjugate Chem., 2008, 19: 813
Yu, J., Javier, D., Yaseen, M.A., Nitin, N., Richards-Kortum, R., Anvari, B. and Wong, M.S., J. Am. Chem. Soc., 2010, 132: 1929
Sano, D., Berlin, J.M., Pham, T.T., Marcano, D.C., Valdecanas, D.R., Zhou, G., Milas, L., Myers, J.N. and Tour, J.M., ACS Nano, 2012, 6: 2497
Cha, M.Y., Lee, K.O., Kang, S.J., Jung, Y.H., Song, J.Y., Choi, K.J., Byun, J.Y., Lee, H.J., Lee, G.S., Park, S.B. and Kim, M.S., J. Med. Chem., 2012, 55: 2846
Sharpe, M.A., Marcano, D.C., Berlin, J.M., Widmayer, M.A., Baskin, D.S. and Tour, J.M., ACS Nano, 2012, 6: 3114
Ongarora, B.G., Fontenot, K.R., Hu, X., Sehgal, I., Satyanarayana-Jois, S.D. and Vicente, M.G.H., J. Med. Chem., 2012, 55: 3725
Zhang, Y., Hong, H., Engle, J.W., Yang, Y., Theuer, C.P., Barnhart, T.E. and Cai, W., Mol. Pharm., 2012, 9: 645
Jiang, H., Wang, H., Tan, Z., Hu, S., Wang, H., Shi, B., Yang, L., Li, P., Gu, J., Wang, H. and Li, Z., J. Biol. Chem., 2011, 286: 5913
Wang, H., Shi, B., Zhang, Q., Jiang, H., Hu, S., Kong, J., Yao, M., Yang, S. and Li, Z., FASEB J., 2012, 26: 73
Wang, H., Zhou, M., Shi, B., Zhang, Q., Jiang, H., Sun, Y., Liu, J., Zhou, K., Yao, M., Gu, J., Yang, S., Mao, Y. and Li, Z., Neoplasia, 2011, 13: 461
Iyer, A.K., Khaled, G., Fang, J. and Maeda, H., Drug Discovery Today, 2006, 11: 812
Maeda, H., Bharate, G.Y. and Daruwalla, J., Eur. J. Pharm. Biopharm., 2009, 71: 409
Maeda, H., Wu, J., Sawa, T., Matsumura, Y. and Hori, K., J. Controlled Release, 2000, 65: 271
Iznaga-Escobar, N., Appl. Radiation Isotopes, 2001, 54: 399
Das, S., Bosley, A.D., Ye, X., Chan, K.C., Chu, I., Green, J.E., Issaq, H.J., Veenstra, T.D. and Andresson, T., J. Proteome Res., 2010, 9: 6696
Meier, R.J., Steiner, M.S., Duerkop, A. and Wolfbeis, O.S., Anal. Chem., 2008, 80: 6274
Zilberstein, G., Korol, L., Righetti, P.G. and Bukshpan, S., Anal. Chem., 2007, 79: 8624
Dadachova, E. and Mirzadeh, S., Nucl. Med. Biol., 1997, 24: 605
Leschied, J.R., Wong, K.K. and Gross, M.D., Clin. Nucl. Med., 2012, 37: 382
Szabo, Z., Alachkar, N., Xia, J., Mathews, W.B. and Rabb, H., Semin. Nucl. Med., 2011, 41: 20
Author information
Authors and Affiliations
Corresponding authors
Additional information
The work was financially supported by the National Natural Science Foundation of China (Nos. 20974062, 30700175), National Basic Research Program (Nos. 2009CB930400, 2012CB821500), and China National Funds for Distinguished Young Scientists (No. 21025417).
Joint first authors
Rights and permissions
About this article
Cite this article
Li, N., Jin, Y., Xue, Lz. et al. 188Re-labeled hyperbranched polysulfonamine as a robust tool for targeted cancer diagnosis and radioimmunotherapy. Chin J Polym Sci 31, 530–540 (2013). https://doi.org/10.1007/s10118-013-1242-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-013-1242-7