Abstract
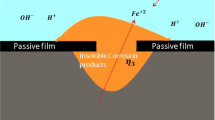
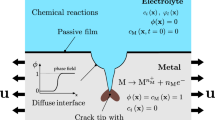
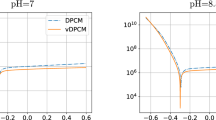
In this paper, a model is established to simulate the time-dependent deposition of corrosion product on the metal surface by considering mass transfer, electrochemical reactions and precipitation reaction. The model is also capable of tacking the movement of metal corrosion interface and the growing interface of the corrosion product deposits via arbitrary Lagrangian–Eulerian finite element method. The current model not only can be used to predict the time-dependent metal corrosion but also for investigating the influences of the deposits’ nature on metal corrosion. The numerical results of current density and corrosion rate are in good agreement with experiments. The presented model predicts that an exponential relationship exists between the maximum corrosion depth and the porosity of corrosion product deposits, and it is also predicted that the growth of the corrosion product layer is linear relative with the root of time, which is consistent with the existing theories.













Similar content being viewed by others
Abbreviations
- a:
-
Anode
- c:
-
Cathode
- c i :
-
Concentration of species i, mol/m3
- c i,bluk :
-
Concentration of species i in bulk solution, mol/m3
- D i :
-
Diffusion coefficient of species i, m2/s
- D ie :
-
Effective diffusion coefficient of species i, m2/s
- F :
-
Faraday’s constant, C/mol
- f(ϕ):
-
Piecewise linear interpolation of polarization data
- J C :
-
Current density of deposits model, A/m2
- J N :
-
Current density of non-deposit model, A/m2
- k :
-
Reaction rate constant for the precipitation reaction of Mg(OH)2, m6/(mol2 s)
- k sp :
-
Apparent solution product constant of Mg(OH)2, mol3/m9
- L c :
-
The length of cathode, m
- m :
-
Cementation exponent
- M i :
-
Molecular weight of species i, kg/mol
- n :
-
Saturation coefficient
- n :
-
Normal vector
- n i :
-
Electrons number of species i
- N i :
-
Molar flux of species i, mol/(m2 s)
- N i,e :
-
Effective molar flux of species i, mol/(m2 s)
- N M,PE :
-
MacMullin number
- R :
-
Universal gas constant, J/(K mol)
- R i :
-
Reaction rate of species i, mol/(m3 s)
- s L :
-
Fluid saturation
- t :
-
Time, s
- T :
-
Temperature, K
- u mi :
-
Mobility of species i, mol m2/(J s)
- u mi,e :
-
Effective mobility of species i, mol m2/(J s)
- v :
-
Velocity vector, m/s
- \( \overline{x} \) :
-
Distance along the metal surface, m
- x :
-
x co-ordinate in spatial frame, m
- y :
-
y co-ordinate in spatial frame, m
- X :
-
X co-ordinate in reference frame, m
- Y :
-
Y co-ordinate in reference frame, m
- z i :
-
Charge of species i
- ϕ :
-
Potential, V vs SCE
- σ :
-
Conductivity of the electrolyte, S/m
- σ e :
-
Effective conductivity of the deposits, S/m
- Γ:
-
Boundary
- ε :
-
Porosity of the deposits
- τ :
-
Tortuosity of the deposits
- v i :
-
Stoichiometric coefficient for species i
- υ :
-
Relative permittivity
- ρ i :
-
Density of species i, kg/m3
- ζ :
-
The dependent variable in step function H
- ξ :
-
Factor in step function H
References
Revie RW, Uhlig HH (2008) Metallic coatings, in corrosion and corrosion control: An introduction to corrosion science and engineering, 4th edn, John Wiley & Sons, Inc., Hoboken, NJ, USA. doi:10.1002/9780470277270.ch1
Wang L, Liu G, Xue D (2010) Electrochim Acta 55:6796–6801
Wang L, Liu G, Xue D (2011) Electrochim Acta 56:6277–6283
Wang L, Liu G, Zou L, Xue D (2011) Appl Surf Sci 257:5519–5523
Wang L, Liu G, Xue D (2010) Mater Lett 64:2475–2478
Gojkovic SL, Zecevic SK, Drazic DM (1994) Electrochim Acta 39:975–982
Volovitch P, Allely C, Ogle K (2009) Corros Sci 51:1251–1262
Sancy M, Gourbeyre Y, Sutter EMM, Tribollet B (2010) Corros Sci 52:1222–1227
Liu W, Cao F, Jia B, Zheng L, Zhang J, Cao C, Li X (2010) Corros Sci 52:639–650
Shinozaki J, Muto I, Ogawa H, Hara N (2009) J Jpn Inst Met 73:533–541
Yuan Y, Ji Y, Jiang J (2009) Mater Struct 42:1443–1450
Yuan Y, Ji Y (2010) Int J Struct Eng 1:199–206
North RF, Pryor MJ (1970) Corros Sci 10:297–311
Yamashita M, Shimizu T, Konishi H, Mizuki J, Uchida H (2003) Corros Sci 45:381–394
Jönsson M, Persson D, Thierry D (2007) Corros Sci 49:1540–1558
Chang J, Guo X, Fu P, Peng L, Ding W (2007) Electrochim Acta 52:3160–3167
García KE, Morales AL, Barrero CA, Greneche JM (2006) Corros Sci 48:2813–2830
Zhang GA, Cheng YF (2011) Electrochim Acta 56:1676–1685
Jeannin M, Calonnec D, Sabot R, Refait P (2011) Electrochim Acta 56:1466–1475
Hamdy AS, Sa’eh AG, Shoeib MA, Barakat Y (2007) Electrochim Acta 52:7068–7074
Keresztes Z, Felhősi I, Kálmán E (2001) Electrochim Acta 46:3841–3849
Witte F, Fischer J, Nellesen J, Crostack H, Kaese V, Pisch A, Beckmann F, Windhagen H (2006) Biomaterials 27:1013–1018
Chen J, Wang J, Han E, Dong J, Ke W (2007) Electrochim Acta 52:3299–3309
Li Y, Zhang T, Wang F (2006) Electrochim Acta 51:2845–2850
Gong Y, Cao J, Meng XH, Yang ZG (2009) Mater Corros 60:899–908
Yang W, Ni R, Hua H (1984) Corros Sci 24:691–707
Sharland SM (1987) Corros Sci 27:289–323
Lin B, Hu R, Ye C, Li Y, Lin C (2010) Electrochim Acta 55:6542–6545
Lv G, Xu C, Lv Y, Cheng H, He Z (2008) Chin J Chem Eng 16:646–649
Perrin M, Gailleta L, Tessiera C, Idrissib H (2010) Corros Sci 52:1915–1926
Lia L, Lia X, Donga C, Huang Y (2009) Electrochim Acta 54:6389–6395
Królikowski A, Kuziak J (2011) Electrochim Acta 56:7845–7853
van Hunnik EWJ, Pots BFM, Hendriksen ELJA (1996) The formation of protective FeCO3 corrosion product layers in CO2 corrosion. CORROSION/96, paper no. 6. NACE International, Houston
Melchers RE (2003) Corros Sci 45:923–940
Melchers RE, Jeffrey R (2005) Corros Sci 47:1678–1693
Melchers RE, Chernov BB (2010) Corros Sci 52:449–454
Melchers RE (2006) Corros Eng Sci Technol 41:38–44
Melchers RE, Jeffrey R (2008) Electrochim Acta 54:80–85
Melchers RE (2006) Corros Sci 48:4174–4201
di Caprioa D, Stafiej J (2011) Electrochim Acta 56:3963–3968
Aarão Reis FDA, Stafiej J (2007) Crossover of interface growth dynamics during corrosion and passivation. J Phys Condens Matter. doi:10.1088/0953-8984/19/6/065125
Saunier J, Dymitrowska M, Chaussé A, Stafiej J, Badiali JP (2005) J Electroanal Chem 582:267–273
di Caprioa D, Stafiej J (2010) Electrochim Acta 55:3884–3890
Taleb A, Chaussé A, Dymitrowska M, Stafiej J, Badiali JP (2004) J Phys Chem B 108:952–958
Aarão Reis FDA, Stafiej J (2007) Scaling behavior in corrosion and growth of a passive film. Phys Rev E. doi:10.1103/PhysRevE.76.011512
Deshpande KB (2010) Corros Sci 52:3514–3522
Deshpande KB (2010) Corros Sci 52:2819–2826
Archie GE (1942) Trans Am Inst Min Metall Pet Eng 146:54–62
Yan JF, Nguyen TV, White RE, Griffin RB (1993) J Electrochem Soc 140:733–742
MacMullin RB, Muccini GA (1956) AIChE J 2:393–403
Deslouis C, Festy D, Gil O, Maillot V, Touzain S, Tribollet B (2000) Electrochim Acta 45:1837–1845
Sun W, Liu G, Wang L, Li Y (2012) Electrochim Acta 78:597–608
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, W., Liu, G., Wang, L. et al. An arbitrary Lagrangian–Eulerian model for studying the influences of corrosion product deposition on bimetallic corrosion. J Solid State Electrochem 17, 829–840 (2013). https://doi.org/10.1007/s10008-012-1935-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-012-1935-9