Abstract
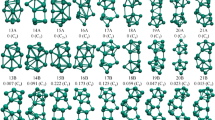
The present study reports the geometry, electronic structure and properties of neutral and anionic transition metal (TM = Ti, Zr and Hf)) doped germanium clusters containing 1 to 20 germanium atoms within the framework of linear combination of atomic orbitals density functional theory under spin polarized generalized gradient approximation. Different parameters, like, binding energy (BE), embedding energy (EE), energy gap between the highest occupied and lowest unoccupied molecular orbitals (HOMO-LUMO), ionization energy (IP), electron affinity (EA), chemical potential etc. of the energetically stable clusters (ground state cluster) in each size are calculated. From the variation of these parameters with the size of the clusters the most stable cluster within the range of calculation is identified. It is found that the clusters having 20 valence electrons turn out to be relatively more stable in both the neutral and the anionic series. The sharp drop in IP as the valence electron count increases from 20 to 21 in neutral cluster is in agreement with predictions of shell models. To study the vibrational nature of the clusters, IR and Raman spectrum of some selected TM@Gen (n = 15,16,17) clusters are also calculated and compared. In the end, relevance of calculated results to the design of Ge-based super-atoms is discussed.




TM@Ge(n) (TM = Ti, Zr and Hf; n = 1-20) clusters












Similar content being viewed by others
References
Ho KM, Shvartsberg AA, Pan B, Lu ZY, Wang CZ, Wacker JG, Fye JL, Jarrod MF (1998) Nature 392:582–585
Wang J, Chen X, Liu JH (2008) J Phys Chem A 112:8868–8876
Zhao WJ, Wang YX (2008) Chem Phys 352:291–296
Jarrold MF, Constant VA (1991) Phys Rev Lett 67:2994–2997
Benedict LX, Puzer A, Willimson AJ, Grossman JC, Galli G, Klepeis JE, Raty JY, Pankratov O (2003) Phys Rev B 68:85310–85317
Brown WL, Freeman RR, Raghavachari K, Schluter M (1987) Science 235:860–865
Hiura H, Miyazaki T, Kanayama T (2001) Phys Rev Lett 86:1733–1736
Hayashi S, Kanzaya Y, Kataoka M, Nagarede T, Yamamoto K (1993) Z Phys D Atom Mol Cl 26:144–146
Bandyopadhyay D, Kaur P, Sen P (2010) J Phys Chem A 114:12986–12991
Polman A (2002) Nat Matters 1:10–12
Bandyopadhyay D, Sen P (2010) J Phys Chem A 114:1835–1842
Jarrold MF, Bower JE (1992) J Chem Phys 96:9180–9190
Kumar V, Kawazoe Y (2001) Phys Rev Lett 87:045503–045506
Kumar V, Kawazoe Y (2002) Phys Rev Lett 88:235504–235507
Bandyopadhyay D (2008) J Appl Phys 104:084308–084314
Bandyopadhyay D (2009) Mol Simul 35:381–394
Kumar M, Bandyopadhyay D (2008) Chem Phys 353:170–176
Beck SM (1987) J Chem Phys 87:4233–4234
Beck SM (1989) J Chem Phys 90:6306–6312
Ohara M, Miyajima K, Pramann A, Nakajima A, Kaya K (2002) J Phys Chem A 106:3702–3705
Han JG (2000) Chem Phys Lett 324:143–148
Wang JL, Wang GH, Zhao JJ (2001) Phys Rev B 64:205411–305415
Hou XJ, Gopakumar G, Lievens P, Nguyen MT (2997) J Phys Chem A 111:13544–13553
Negishi Y, Kawamata H, Hayase T, Gomei T, Kishi R, Hayakawa F, Nakajima A, Kaya K (1997) Chem Phys Lett 269:199–207
Bandyopadhyay D (2009) Nanotechnology 20:275202–275213
Huheey JE, Keiter EA, Keiter RL (2000) Inorganic Chemistry: principles of structure and reactivity, 4th edn. Harper-Collins College Publisher, New York
Sen P, Mitas L (2003) Phys Rev B 68:155404–155407
Reveles JU, Khanna SN (2005) Phys Rev B 72:165413–165418
Guo LJ, Zhao G, Gu Y, Liu X, Zeng Z (2008) Phys Rev B 77:195417–195424
Wigner E, Witmer EE (1928) Z Phys 51:859–886
Koyasu K, Akutsu M, Mitsui M, Nakajima A (2005) J Am Chem Soc 127:4998–4999
Kumar V (2003) Eur Phys J D 24:227–232
Burke K, Perdew JP et al. (1998) In: Dobson JF, Vignale G, Das MP (eds) Electronic Density Functional Theory: Recent Progress and New Directions. Plenum
Perdew JP (1991) In: Ziesche P, Eschrig H (eds) Electronic Structure of solids ’91. Akademie, Berlin
Becke AD (1988) Phys Rev A 38:3098–3100
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Wang J, Han GJ (2005) J Chem Phys 123:064306–064321
Han JG, Hagelberg F (2001) J Mol Struct THEOCHEM 549:165–180
Guo P, Ren ZY, Wang F, Bian J, Han JG, Wang GH (2004) J Chem Phys 121:12265–12275
Guo LJ, Liu X, Zhaoa GF, Luo YH (2007) J Chem Phys 126:234704–234710
Nagendran S, Sen SS, Roesky HW, Koley D, Grubmüller H, Pal A, Herbst-Irmer R (2008) Organometallics 27:5459–5463
Khon W, Sham LJ (1965) Phys Rev 140:A1133–A1138
Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Baboul AG, Stefanov BB, Liu B, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (2004) Gaussian 03, Revision E.01. Gaussian, Inc, Wallingford, CT
Lu J, Nagase S (2003) Chem Phys Lett 372:394–398
Kumar V, Kawazoe Y (2002) Appl Phys Lett 80:859–861
Kumar V, Kawazoe Y (2007) Phys Rev B 75:155425–155435
de Heer WA (1993) Rev Mod Phys 65:611–676
Acknowledgments
Complete computations using Gaussian 03 were performed at the cluster computing facility, Harish-Chandra Research Institute, Allahabad, UP, India (http://cluster.hri.res.in).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, M., Bhattacharyya, N. & Bandyopadhyay, D. Architecture, electronic structure and stability of TM@Ge(n) (TM = Ti, Zr and Hf; n = 1-20) clusters: a density functional modeling. J Mol Model 18, 405–418 (2012). https://doi.org/10.1007/s00894-011-1122-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-011-1122-4