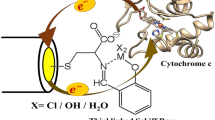
Abstract
Naturally occurring hemin cofactor has been functionalized to introduce two terminal alkyne groups. This modified hemin has been successfully covalently attached to mixed self-assembled monolayers of alkanethiols and azide-terminated alkanethiols on gold electrodes using a CuI-catalyzed 1,3-cycloaddition reaction. However these hemin-modified electrodes could not be used to reconstitute apomyoglobin on gold electrodes owing to the hydrophobicity of the alkane thiol self-assembled monolayer. Modification of existing techniques allowed covalent attachment of alkyne-terminated electroactive species onto mixed monolayers of azidothiols and carboxylatoalkanethiols on electrodes using the same CuI-catalyzed 1,3-cycloaddition reaction. Apomyoglobin could be reconstituted using the hemin covalently attached to these hydrophilic electrodes. The electrochemical data, UV–vis absorption data, surface-enhanced resonance Raman spectroscopy data, and atomic force microscopy data indicate the presence of these modified myoglobin proteins on these electrodes. The direct attachment of the heme cofactor of these modified myoglobin proteins to the electrode allows fast electron transfer to the heme center from the electrode and affords efficient O2-reducing bioelectrodes under physiological conditions.
Graphical Abstract



















Similar content being viewed by others
Notes
We were unable to obtain good-quality SERRS data on the reduced form in the absence of imidazole. We think this is due to high O2 binding affinity of Mb, which reacts with any O2 that is left behind in the buffer after repeated freeze–pump–thaw cycles.
References
Shikama K (1998) Chem Rev 98:1357–1373
Mozzarelli A, Rivetti C, Rossi GL, Henry ER, Eaton WA (1991) Nature 351:416–419
Collman JP, Boulatov R, Sunderland CJ, Fu L (2004) Chem Rev 104:561–588
Jones RD, Summerville DA, Basolo F (1979) Chem Rev 79:139–179
Williams RJP (1956) Chem Rev 56:299–328
Kaila VRI, Verkhovsky MI, Wikstrom M (2010) Chem Rev 110:7062–7081
Ferguson-Miller S, Babcock GT (1996) Chem Rev 96:2889–2908
Bertini I, Cavallaro G, Rosato A (2006) Chem Rev 106:90–115
Rouzer CA, Marnett LJ (2003) Chem Rev 103:2239–2304
Weissbuch I, Leiserowitz L (2008) Chem Rev 108:4899–4914
Trijbels JMF, Sengers RCA, Ruitenbeek W, Fischer JC, Bakkeren JAJM, Janssen AJM (1988) Eur J Pediatr 148:92–97
Liu G, Gooding JJ (2006) Langmuir 22:7421–7430
Zhang HM, Li NQ (2000) Bioelectrochemistry 53:97–101
Dronov R, Kurth DG, Möhwald H, Spricigo R, Leimkühler S, Wollenberger U, Rajagopalan KV, Scheller FW, Lisdat F (2008) J Am Chem Soc 130:1122–1123
Bonk SM, Lisdat F (2009) Biosens Bioelectron 25:739–744
Kumar SA, Chen SM (2007) Biosens Bioelectron 22:3042–3050
Cracknell JA, Vincent KA, Armstrong FA (2008) Chem Rev 108:2439–2461
Feng JJ, Zhao G, Xu JJ, Chen HY (2005) Anal Biochem 342:80–286
Chung YH, Lee T, Min J, Choi JW (2010) Mol Cryst Liq Cryst 519:19–26
Murgida DH, Hildebrandt P, Wei J, He YF, Haiying L, Waldeck DH (2004) J Phys Chem B 108:2261–2269
Wei JJ, Liu HY, Khoshtariya DE, Yamamoto H, Dick A, Waldeck DH (2002) Angew Chem Int Ed 41:4700–4703
Vojtechovsky J, Chu K, Berendzen J, Sweet RM, Schlichting I (1999) Biophys J 77:2153–2174
Carlsson GH, Nicholls P, Svistunenko D, Berglund GI, Hajdu J (2005) Biochemistry 44:635
Matsui T, Iwasaki M, Sugiyama R, Unno M, Ikeda-Saito M (2010) Inorg Chem 49:3602–3609
Wang L, Cowley AB, Terzyan S, Zhang X, Benson DR (2007) Proteins 67:293–304
Hayashi T, Hisaeda Y (2002) Acc Chem Res 35:35–43
Kolb HC, Finn MG, Sharpless KB (2001) Angew Chem Int Ed 40:2004–2021
Hong V, Presolski SI, Ma C, Finn MG (2009) Angew Chem Int Ed 48:9879–9883
Moses JE, Moorhouse AD (2007) Chem Soc Rev 36:1249–1262
Lu C, Zhao X, Lu Y, Rousseau DL, Yeh SR (2010) J Am Chem Soc 132:1598–1605
Yeung N, Lin YW, Gao YG, Zhao X, Russell BS, Lei L, Miner KD, Robinson H, Lu Y (2009) Nature 462:1079–1084
Lin YW, Yeung N, Gao YG, Miner KD, Tian S, Robinson H, Lu Y (2010) Proc Natl Acad Sci USA 107:8581–8586
Collman JP, Devaraj NK, Chidsey CED (2004) Langmuir 20:1051–1053
Zhang Y, Wilson GS (1993) J Electroanal Chem 345:253–271
Teale FWJ (1959) Biochim Biophys Acta 35:543
Yonetani T, Asakura T (1969) J Biol Chem 244:4580–4588
Bulovas A, Talaikytė Z, Niaura G, Kažemėkaitė M, Marcinkevičienė L, Bachmatova I, Meškys R, Razumas V (2007) Chemija 18:9–15
David DG, Orleron DJ (1966) Anal Chem 38:179–183
Yan L, Marzolin C, Terfort A, Whitesides GM (1997) Langmuir 13:6704–6712
Arnold R, Azzam W, Terfort A, Wöll C (2002) Langmuir 18:3980–3992
Winter N, Vieceli J, Benjamin I (2008) J Phys Chem B 112:227–231
Guo LH, McLendon G, Razafitrimo H, Gao Y (1996) J Mater Chem 6:369–374
Weidinger IM, Murgida DH, Dong WF, Mohwald H, Hildebrandt P (2006) J Phys Chem B 110:522–529
Wegerich F, Turano P, Allegrozzi M, Mohwald H, Lisdat F (2009) Anal Chem 81:2976–2984
Bowen WJ (1949) J Biol Chem 179:235–245
Hu S, Smith KM, Spiro TG (1996) J Am Chem Soc 118:12638
Kent MS, Yim H, Sasaki DY (2004) Langmuir 20(7):2819–2829
Bard AJ, Faulkner LR (1980) Electrochemical methods. Wiley, New York, p 300
Springer BA, Egeberg KD, Sligar SG (1989) J Biol Chem 264:3057–3060
Carver TE Jr, Brantley RE, Singleton EW, Arduini RM, Quillin ML, Phillips GN, Olson JS (1992) J Biol Chem 267:14443–14450
Brantley RE Jr, Smerdon SJ, Wilkinson AJ, Singleton EW, Olson JS (1993) J Biol Chem 268:6995–7010
Acknowledgment
This research was sponsored by DST grant SR/S1/IC-35/2009.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mukherjee, S., Sengupta, K., Das, M.R. et al. Site-specific covalent attachment of heme proteins on self-assembled monolayers. J Biol Inorg Chem 17, 1009–1023 (2012). https://doi.org/10.1007/s00775-012-0915-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-012-0915-y