Abstract
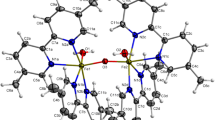
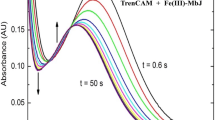
Pyochelin, its analog 3′′-nor-NH-pyochelin, and the related methyl hydroxamate, 2-(2′-hydroxyphenyl)-4,5-dihydrothiazol-4-carboxylic acid methoxymethyl amide, have been prepared together with their Fe(III) complexes. The solution stoichiometry and the coordination of the three Fe(III) complexes in methanol or buffered (pH∼2) 50:50 (v/v) methanol–water mixtures were determined using various spectroscopic methods: UV–vis absorption, X-ray absorption, extended X-ray absorption fine structure and electron paramagnetic resonance. All three systems showed both a 1:1 and 2:1 ligand–Fe(III) stoichiometry, but presented different coordination properties. Conditional formation constants (pH∼2) were determined for both the 1:1 and 2:1 complexes in all three systems. Computation of the coordination-conformational energies by semiempirical methods indicated that the coordination in the case of the 2:1 complexes of pyochelin–Fe(III) and 3′′-nor-NH-pyochelin–Fe(III) was asymmetrical, with one molecule of pyochelin (or 3′′-nor-NH-pyochelin) tetradentately coordinated (O1, N1, N2 and O3) to the Fe(III), and the second molecule bound bidentately (O1, N1 or N2, O3), to complete the octahedral geometry. In contrast, two molecules of the methyl hydroxamate each provided a set of tridentate ligand atoms in the formation of the 2:1 ligand–Fe(III) complex. These results are consistent with the role of pyochelin in the uptake of iron by the FptA receptor in the outer membrane of Pseudomonas aeruginosa and in several gram-negative bacteria.











Similar content being viewed by others
References
Liu PV, Shokrani F (1978) Infect Immun 22:878–890
Sokol PA (1986) J Clin Microbiol 23:560–562
Sokol PA, Woods DE (1983) Infect Immun 40:665–669
Cox CD, Rinehart KL, Moore ML, Cook JC (1981) Proc Natl Acad Sci USA 78: 4256–4260
Cox CD, Graham R (1979) J Bacteriol 137(1):357–364
Cox CD (1980) J Bacteriol 142:581–587
Beier RC, Stipanovic RD (1989) Biomed Environ Mass Spectrom 18:185–191
Namiranian S, Richardson DJ, Russell DA, Sodeau JR (1997) Photochem Photobiol 65: 777–782
Klumpp C, Burger A, Mislin GL, Abdallah MA (2005) Bioorg Med Chem Lett 15(6):1721–1724
Cobessi D, Celia H, Pattus F (2005) J Mol Biol 352: 893–904
Zamri A, Abdallah MA (2000) Tetrahedron 56:249–256; corrigendum: Zamri A, Abdallah MA (2000) Tetrahedron 56:9397
Zamri A, Schalk IJ, Pattus F, Abdallah MA (2003) Bioorg Med Chem Lett 13:1147–1150
Mislin GL, Burger A, Abdallah MA (2004) Tetrahedron 60(52):12139–12145
Schlegel K, Taraz K, Budzikiewicz H (2004) BioMetals 17:409–414
Ecker DJ, Lancaster JR Jr, Emery T (1982) J Biol Chem 257(15):8623–8626
Acknowledgements
We wish to thank Françoise Hoegy for her expert technical assistance. We acknowledge the French Institute of Taipei and the French Minister of Foreign Affairs for a grant and for travel expenses. We also thank Academia Sinica and the National Science Council of Taiwan (Republic of China) for travel expenses.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tseng, CF., Burger, A., Mislin, G.L.A. et al. Bacterial siderophores: the solution stoichiometry and coordination of the Fe(III) complexes of pyochelin and related compounds. J Biol Inorg Chem 11, 419–432 (2006). https://doi.org/10.1007/s00775-006-0088-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-006-0088-7