Abstract
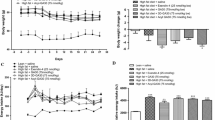
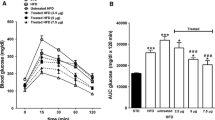
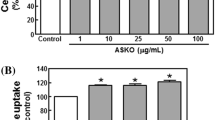
The in vivo effects of gavage administration of the synthetic, functional biomimetic cation [Cr3O(O2CCH2CH3)6(H2O)3]+ to healthy and type 2 diabetic model rats are described. After 24 weeks of treatment (0–1,000 μg Cr/kg body mass) of healthy Sprague Dawley rats, the cation results in a lowering (P<0.05) of fasting blood plasma low-density lipoprotein (LDL) cholesterol, total cholesterol, triglycerides, and insulin levels and of 2-h plasma insulin and glucose concentrations after a glucose challenge. Zucker obese rats (a model of the early stages of type 2 diabetes) and Zucker diabetic fatty rats (a model for type 2 diabetes) after supplementation (1,000 μg Cr/kg) have lower fasting plasma total, high-density lipoprotein, and LDL cholesterol, triglycerides, glycated hemoglobin, and insulin levels and lower 2-h plasma insulin levels in glucose tolerance tests. The lowering of plasma insulin concentrations with little effect on glucose concentrations suggests that the supplement increases insulin sensitivity. The cation after 12 and 22 or 24 weeks of administration lowers (P<0.05) fasting plasma glycated hemoglobin levels in the Zucker diabetic and Zucker obese rats, respectively, and thus can improve the glucose status of the diabetic models. The effects cannot be attributed to the propionate ligand.



Similar content being viewed by others
Abbreviations
- HDL:
-
High-density lipoprotein
- LDL:
-
Low-density lipoprotein
- PTP 1B:
-
Protein tyrosine phosphatase 1B
- SD:
-
Standard deviation
- VLDL:
-
Very low density lipoprotein
- ZDF:
-
Zucker diabetic fatty
- ZKO:
-
Zucker obese
References
Vincent JB (2001) Polyhedron 20:1–26
Vincent JB (2000) Acc Chem Res 33:503–510
Lukaski HC (1999) Ann Rev Nutr 19:279–301
Striffler JS, Law JS, Polansky MM, Bhathena SJ, Anderson RA (1995) Metabolism 44:1314–1320
Striffler JS, Polansky MM, Anderson RA (1999) Metabolism 48: 1063–1068
Trumbo P, Yates AA, Schlicker S, Poos M (2001) J Am Diet Assoc 101:294–301
Anderson RA, Kozlovsky AS (1985) Am J Clin Nutr 41:1177–1183
Bunker VW, Lawson MS, Delues HT, Clayton BE (1984) Am J Clin Nutr 39:797–802
Offenbacher EG, Spencer H, Dowling HJ, Pi-Sunyer FX (1986) Am J Clin Nutr 44:77–82
Pittler MH, Stevinson C, Ernst E (2003) Int J Obesity 27:522–529
Nissen SL, Sharp RL (2003) J Appl Physiol 94:651–659
Vincent JB (2003) Sports Med 33:213–230
Morris BW, MacNeil S, Hardisty CA, Heller S, Burgin C, Gray TA (1999) J Trace Elem Med Biol 13:57–61
Althius MD, Jordan NE, Ludington EA, Wittes JT (2002) Am J Clin Nutr 76:148–155
Anderson RA, Cheng NC, Bryden NA, Polansky MM, Cheng N, Chi J, Feng J (1997) Diabetes 46:1786–1791
Hellerstein MK (1998) Nutr Rev 56:302–306
Anderson RA (1998) J Am Coll Nutr 17:548–555
Sun Y, Mallya K, Ramirez J, Vincent JB (1999) J Biol Inorg Chem 4:838–845
Sun Y, Clodfelder BJ, Shute AA, Irvin T, Vincent JB (2002) J Biol Inorg Chem 7:852–862
Cefalu WT, Wang ZQ, Zhang XH, Baldor LC, Russell JC (2002) J Nutr 132:1107–1114
Anderson RA, Bryden NA, Polansky MM (1997) J Am Coll Nutr 16:273–279
Davis CM, Royer AC, Vincent JB (1997) Inorg Chem 36:5316–5320
Johnson MK, Powell DB, Cannon RD (1981) Spectrochim Acta 37A: 995–1006
Clodfelder BJ, Chang C, Vincent JB (2004) Trace Elem Biol Res 98:159–170
Earnshaw A, Figgis BN, Lewis J (1966) J Chem Soc A 1656–1663
Schwarz K, Mertz W (1959) Arch Biochim Biophys 85:292–295
Fish WW (1988) Methods Enzymol 158:357–364
Miller-Ihli NJ (1996) J Food Comp Anal 9:290–300
Hasten DL, Hegsted M, Keenan MJ, Morris GS (1997) Nutr Res 17:283–294
Hasten DL, Hegsted M, Keenan MJ, Morris GS (1997) Nutr Res 17:1175–1186
Morris GS, Guidry KA, Hegsted M, Hasten DL (1995) Nutr Res 15: 1045–1052
Sparks JD, Shaw WN, Corsetti JP, Bolognino M, Pesek JF, Sparks CE (2000) Metabolism 49:1424–1430
Brunzell JD, Chait A (1990) Lipoprotein pathophysiology and treatment. In: Rifkin H, Porte D Jr (eds) Ellenberg and Rifkin’s diabetes mellitus: theory and practice. Elsevier, New York, pp 756–767
Lukaski HC, Bolonchuk W, Siders WA, Milne DB (1996) Am J Clin Nutr 63:954–965
Anderson RA, Polansky MM (1995) Biol Trace Elem Res 50:97–108
Shute AA, Chakov NE, Vincent JB (2001) Polyhedron 20:2241–2252
Striffler JS, Polansky MM, Anderson RA (1998) Metabolism 47: 396–400
Olin KL, Stearns DM, Armstrong WH, Keen CL (1994) Trace Elem Electrolytes 11:182–186
Anderson RA, Bryden NA, Polansky MM, Gautschi K (1996) J Trace Elem Exp Med 9:11–25
Ginsberg HN (2000) J Clin Invest 106:453–458
Shute AA, Vincent JB (2002) J Inorg Biochem 89:272–278
Vincent JB (2004) Biol Trace Elem Res 99:1–16
Vincent JB (2003) Sports Med 33:213–230
Stearns DM, Wise JP Sr, Patierno SR, Wetterhahn KE (1995) FASEB J9: 1643–1648
Bagchi D, Bagchi M, Balmoori J, Ye X, Stohs SJ (1997) Res Commun Mol Pathol Pharmacol 97:335–346
Bagchi D, Stohs SJ, Downs BW, Bagchi M, Preuss HG (2002) Toxicology 180:5–22
Manygoats KR, Yazzie M, Stearns DM (2002) J Biol Inorg Chem 7:791–798
Stearns DM, Silveira SM, Wolf KK, Luke AM (2002) Mutat Res 513: 135–142
Speetjens JK, Collins RA, Vincent JB, Woski SA (1999) Chem Res Toxicol 12:483–487
Mahboob L, McNeil L, Toliver T, Odgen L (2000) Toxicol Sci 66(1-S): 32
Hepburn DDD, Xiao J, Bindom S, Vincent JB, O’Donnell (2003) Proc Natl Acad Sci USA 100:3766–3771
Hepburn DDD, Burney JM, Woski SA, Vincent JB (2003) Polyhedron 22: 455–463
http://www.foodstandards.gov.uk/news. newsarchive/safetyhighdosesvitsandmins accessed on 08 May 2003
Kato I, Vogelman JH, Dilman V, Karkoszka J, Frenkel K, Durr NP, Orentreich N, Toniolo P (1998) Eur J Epidemiol 14:621–626
Speetjens JK, Parand A, Crowder MW, Vincent JB, Woski SA (1999) Polyhedron 18:2617-2624
Stearns DM, Belbruno JJ, Wetterhahn KE (1995) FASEB J 9:1650–1657
Ross SA, Gulve EA, Wang M (2004) Chem Rev 104:1255–1282
Zhang B, Salituro G, Szalkowski D, Li Z, Zhang Y, Royo I, Vilella D, Diez MT, Pelaez F, Ruby C, Kendall RL, Mao X, Griffin P, Calaycay J, Zierath JR, Heck JV, Smith RG, Moller DE (1999) Science 284:974–977
Li M, Youngren JF, Manchem VP, Kozlowski M, Zhang BB, Maddux BA, Goldfine ID (2001) Diabetes 50:2323–2328
Pender C, Goldfine ID, Manchem VP, Evans JL, Spevak WR, Shi S, Rao S, Bajjalieh S, Maddux BA, Youngren JF (2002) J Biol Chem 277: 43565–43571
Peters KG, Davis MG, Howard BW, Pokross M, Rastogi V, Diven C, Greis KD, Eby- Wilkens E, Maier M, Evdokimov A, Soper S, Genbauffe F (2003) J Inorg Biochem 96:321–330
Goldfine AB, Patti ME, Zuberi L, Goldstein BJ, LeBlanc R, Landaker EJ, Jiang ZY, Willsky GR, Kahn CR (2000) Metabolism 49:400–410
Goldstein BJ, Zhu L, Hager R, Zilering A, Sun Y, Vincent JB (2001) J Trace Elem Exp Med 14:393–404
Acknowledgements
The authors wish to thank Christine Chang, Jelena Hamilton, Allison Pickering, and James A. Neville and the staff of The University of Alabama Animal Care Facility for assistance with the rat studies. Funding was provided by the National Institutes of Health (DK62094–01) (J.B.V.).
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s00775-005-0651-7
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Clodfelder, B.J., Gullick, B.M., Lukaski, H.C. et al. Oral administration of the biomimetic [Cr3O(O2CCH2CH3)6(H2O)3]+ increases insulin sensitivity and improves blood plasma variables in healthy and type 2 diabetic rats. J Biol Inorg Chem 10, 119–130 (2005). https://doi.org/10.1007/s00775-004-0618-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-004-0618-0