Abstract
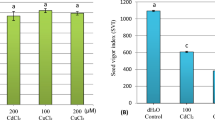
Although negative effects on the offspring fitness can be triggered by the mother-plant exposure to environmental stresses, some plants are able to “remember” past incidents and enhance the progeny tolerance. Here, the mineral profile, cytogenetic modifications, and physiological potential of seeds from two tomato cultivars, with contrasting tolerance degrees to cadmium (Cd) toxicity, were evaluated after plant exposure to this metal. Both cultivars exhibited high Cd translocation to the seeds; however, the tolerant tomato accumulated more Cd than did the sensitive one. As a consequence of the Cd accumulation, reductions in the Mn concentration in Cd-challenged plants were detected. Surprisingly, seed germination and vigor were increased in the tolerant tomato cultivar after Cd exposure, despite increases in the chromosomal abnormalities. By contrast, seeds from the sensitive cultivar exhibited no changes in their physiological potential after Cd exposure, despite Cd-induced reductions in the mitotic index. Moreover, bunch position exerted effects on the vigor and type of chromosomal abnormality. The results show that maternal plant exposure to Cd can affect tomato offspring by changing the seed physiological potential, and such effect can be partially explained by alterations in the seed-derived elements (essential and non-essential) and genotype-dependent tolerance mechanisms.



Similar content being viewed by others
References
Alves LA, Monteiro CC, Carvalho RF, Ribeiro PC, Tezotto T, Azevedo RA, Gratão PL (2017) Cadmium stress related to root-to-shoot communication depends on ethylene and auxin in tomato plants. Environ Exp Bot 134:102–115. https://doi.org/10.1016/j.envexpbot.2016.11.008
Azevedo RA, Gratão PL, Monteiro CC, Carvalho RF (2012) What is new in the research on cadmium-induced stress in plants? Food Energy Secur 1(2):133–140. https://doi.org/10.1002/fes3.10
Bayçu G, Gevrek-Kürüm N, Moustaka J, Csatári I, Rognes SE, Moustakas M (2017a) Cadmium-zinc accumulation and photosystem II responses of Noccaea caerulescens to Cd and Zn exposure. Environ Sci Pollut Res 24(3):2840–2850. https://doi.org/10.1007/s11356-016-8048-4
Bayçu G, Rognes SE, Özden H, Gören-Saglam N, Csatári I, Szabó S (2017b) Abiotic stress effects on the antioxidative response profile of Albizia julibrissin Durazz. (Fabaceae). Braz J Bot 40(1):21–32. https://doi.org/10.1007/s40415-016-0318-3
Benavides MP, Gallego SM, Tomaro ML (2005) Cadmium toxicity in plants. Braz J Plant Physiol 17(1):21–34. https://doi.org/10.1590/S1677-04202005000100003
Bergougnoux V (2014) The history of tomato: from domestication to biopharming. Biotechnol Adv 32(1):170–189. https://doi.org/10.1016/j.biotechadv.2013.11.003
Borges KLR, Salvato F, Alcântara BK, Nalin RS, Piotto FA, Azevedo RA (2018) Temporal dynamic responses of roots in contrasting tomato genotypes to cadmium tolerance. Ecotoxicology, in press. https://doi.org/10.1007/s10646-017-1889-x
Branco-Neves S, Soares C, Sousa A, Martins V, Azenha M, Gerós H, Fidalgo F (2017) An efficient antioxidant system and heavy metal exclusion from leaves make Solanum cheesmaniae more tolerant to Cu than its cultivated counterpart. Food Energy Secur 6(3):123–133. https://doi.org/10.1002/fes3.114
Brasil (2009) Ministério da Agricultura, Pecuária e Abastecimento. Regras para análise de sementes. Ministério da Agricultura, Pecuária e Abastecimento. Secretaria de Defesa Agropecuária. Brasília: MAPA/ACS. 395p. Disponible at :http://www.agricultura.gov.br/arq_editor/file/laborat%c3%b3rio/sementes/regras%20para%20analise%20de%20sementes.pdf
Cuypers AC, Hendrix S, Reis RA, Smet S, Deckers J, Gielen H, Jozefczak M, Loix C, Vercampt H, Vangronsveld J, Keunen E (2016) Hydrogen peroxide, signaling in disguise during metal phytotoxicity. Front Plant Sci 7:470
Donohue K (2009) Completing the cycle: maternal effects as the missing link in plant life histories. Philos Trans R Soc B Biol Sci 364(1520):1059–1074. https://doi.org/10.1098/rstb.2008.0291
Dourado MN, Martins PF, Quecine MC, Piotto FA, Souza LA, Franco MR, Tezotto T, Azevedo RA (2013) Burkholderia sp. SCMS54 reduces cadmium toxicity and promotes growth in tomato. Ann Appl Biol 163:494–507
Dziubaneka G, Baranowskab R, Ćwieląg-Drabeka M, Spychałab A, Piekutb A, Rusina M, Hajokb I (2017) Cadmium in edible plants from Silesia, Poland, and its implication for health risk in populations. Ecotoxicol Environ Saf 142:8–13. https://doi.org/10.1016/j.ecoenv.2017.03.048
Esalq (2016) Climatological Data Series of “Luiz de Queiroz” campus at Piracicaba, SP, Brazil. Department of Biossystems, Escola Superior de Agricultura “Luiz de Queiroz”/Universidade de São Paulo
Fernández R, Bertrand A, Reis R, Mourato MP, Martins LL, González A (2013) Growth and physiological responses to cadmium stress of two populations of Dittrichia viscosa (L.) Greuter. J Hazard Mater 244-245:555–562. https://doi.org/10.1016/j.jhazmat.2012.10.044
Feulgen R, Rossenbeck H (1924) Mikroskopisch-chemischer nachweis einer nucleinsäure vom typus der thymonucleinsaure und die darauf beruhende elektive farbung vom zelikernen in mikroskopischen praparaten. Zts Phys Chem 135:203–248
Fidalgo F, Azenha M, Silva AF, Sousa A, Santiago A, Ferraz P, Teixeira J (2013) Copper-induced stress in Solanum nigrum L. and antioxidant defense system responses. Food Energy Secur 2(1):70–80. https://doi.org/10.1002/fes3.20
Fidalgo F, Freitas R, Ferreira R, Pessoa AM, Teixeira J (2011) Solanum nigrum L. antioxidant defence system isozymes are regulated transcriptionally and posttranslationally in Cd-induced stress. Environ Exp Bot 72(2):312–319. https://doi.org/10.1016/j.envexpbot.2011.04.007
Gallego SM, Pena LB, Barcia RA, Azpilicueta CE, Iannone MF, Rosales EP, Zawoznik MS, Groppa MD, Benavides MP (2012) Unravelling cadmium toxicity and tolerance in plants: insight into regulatory mechanisms. Environ Exp Bot 83:33–46. https://doi.org/10.1016/j.envexpbot.2012.04.006
Giri DN, Sinha AK (1983) Effects of heavy metal salts on susceptibility of rice seedlings to brown spot disease. Ann Appl Biol 103(2):229–235. https://doi.org/10.1111/j.1744-7348.1983.tb02759.x
Gratão PL, Monteiro CC, Antunes AM, Peres LEP, Azevedo RA (2008) Acquired tolerance of tomato (Lycopersicon esculentum cv. Micro-Tom) plants to cadmium-induced stress. Ann Appl Biol 153(3):321–333. https://doi.org/10.1111/j.1744-7348.2008.00299.x
Gratão PL, Monteiro CC, Carvalho RF, Tezotto T, Piotto FA, Peres LEP, Azevedo RA (2012) Biochemical dissection of diageotropica and Never ripe tomato mutants to Cd-stressful conditions. Plant Physiol Biochem 56:79–86. https://doi.org/10.1016/j.plaphy.2012.04.009
Gratão PL, Monteiro CC, Tezotto T, Carvalho RF, Alves LR, Peters LP, Azevedo RA (2015) Cadmium stress antioxidant responses and root-to-shoot communication in grafted tomato plants. Biometals 28(5):803–816. https://doi.org/10.1007/s10534-015-9867-3
Gratão PL, Polle A, Lea PJ, Azevedo RA (2005) Making the life of heavy metal-stressed plants a little easier. Funct Plant Biol 32(6):481–494. https://doi.org/10.1071/FP05016
Hartke S, Silva AA, Moraes MG (2013) Cadmium accumulation in tomato cultivars and its effect on expression of metal transport-related genes. Bull Environ Contam Toxicol 90(2):227–232. https://doi.org/10.1007/s00128-012-0899-x
Hasan MK, Liu C, Wang F, Ahammed GJ, Zhou J, Xu M-X, Yu J-Q, Xia XJ (2016) Glutathione-mediated regulation of nitric oxide, S-nitrosothiol and redox homeostasis confers cadmium tolerance by inducing transcription factors and stress response genes in tomato. Chemosphere 161:536–545. https://doi.org/10.1016/j.chemosphere.2016.07.053
Hédiji H, Djebali W, Belkadhi A, Cabasson C, Moing A, Rolin D, Brouquisse R, Gallusci P, Chaïbi W (2015) Impact of long-term cadmium exposure on mineral content of Solanum lycopersicum plants: consequences on fruit production. South Afric J Bot 97:176–181. https://doi.org/10.1016/j.sajb.2015.01.010
Hédiji H, Djebali W, Cabasson C, Maucourt M, Baldet P, Bertrand A, Zoghlami LB, Deborde C, Moing A, Brouquisse R, Chaïbi W, Gallusci P (2010) Effects of long-term cadmium exposure on growth and metabolomic profile of tomato plants. Ecotoxicol Environ Saf 73(8):1965–1974. https://doi.org/10.1016/j.ecoenv.2010.08.014
Herman JJ, Sultan SE (2011) Adaptive transgenerational plasticity in plants: case studies, mechanisms, and implications for natural populations. Front Plant Sci 2:102
Huber DM, Jones JB (2013) The role of magnesium in plant disease. Plant Soil 368(1-2):73–85. https://doi.org/10.1007/s11104-012-1476-0
Hussain MM, Saeed A, Khan AA, Javid S, Fatima B (2015) Differential responses of one hundred tomato genotypes grown under cadmium stress. Genet Mol Res 14(4):13162–13171. https://doi.org/10.4238/2015.October.26.12
Iannone MF, Groppa MD, Benavides MP (2015) Cadmium induces different biochemical responses in wild type and catalase-deficient-tobacco plants. Environ Exp Bot 109:201–211. https://doi.org/10.1016/j.envexpbot.2014.07.008
Kubo K, Kobayashi H, Fujita M, Otaa T, Minamiyama Y, Watanabee Y, Nakajima T, Shinano T (2016) Varietal differences in the absorption and partitioning of cadmium in common wheat (Triticum aestivum L.) Environ Exp Bot 24:79–88
Kudo H, Kudo K, Uemura M, Kawai S (2015) Magnesium inhibits cadmium translocation from roots to shoots, rather than the uptake from roots, in barley. Botany 93(6):345–351. https://doi.org/10.1139/cjb-2015-0002
Kumar P, Edelstein M, Cardarelli M, Ferri E, Colla G (2015) Grafting affects growth, yield, nutrient uptake, and partitioning under cadmium stress in tomato. Hortscience 50:1654–1661
Leme DM, Marin-Morales MA (2009) Allium cepa test in environmental monitoring: a review on its application. Mutat Res 682(1):71–81. https://doi.org/10.1016/j.mrrev.2009.06.002
Lin L, Chen F, Cai Y, Chen ZH, Cao F (2017) Genotypic differences in cadmium transport in developing barley grains. Environ Sci Pollut Res 24(8):7009–7015. https://doi.org/10.1007/s11356-017-8399-5
Maguire JD (1962) Speeds of germination-aid selection and evaluation for seedling emergence and vigor. Crop Sci 2(2):176–177. https://doi.org/10.2135/cropsci1962.0011183X000200020033x
Marcos-Filho J (2016) Seed physiology of cultivated plants, 2nd edn. ABRATES, Londrina
Marschner P (2012) Marschner’s mineral nutrition of higher plants. Academic Press, San Diego
Méndez AAE, Pena LB, Benavides MP, Gallego SM (2016) Priming with NO controls redox state and prevents cadmium-induced general up-regulation of methionine sulfoxide reductase gene family in Arabidopsis. Biochimie 131:128–136. https://doi.org/10.1016/j.biochi.2016.09.021
Mengarda LHG, Lopes JC (2012) Qualidade de sementes e desenvolvimento inicial de plântulas de pimenta malagueta e sua relação com a posição de coleta de frutos. Rev Brasil Sementes 34(4):644–650. https://doi.org/10.1590/S0101-31222012000400016
Niu Y, Chai R, Liu L, Jin G, Liu M, Tang C, Zhang Y (2014) Magnesium availability regulates the development of root hairs in Arabidopsis thaliana (L.) Heynh. Plant Cell Environ 37(12):2795–2813. https://doi.org/10.1111/pce.12362
Nogueirol RC, Monteiro FA, Gratão PL, Silva BKA, Azevedo RA (2016) Cadmium application in tomato: nutritional imbalance and oxidative stress. Water Air Soil Poll 227(6):210. https://doi.org/10.1007/s11270-016-2895-y
Norton GJ, Travis AJ, Danku JMC, Salt DE, Hossain M, Islam MR, Price AH (2017) Biomass and elemental concentrations of 22 rice cultivars grown under alternate wetting and drying conditions at three field sites in Bangladesh. Food Energy Secur 6(3):98–112. https://doi.org/10.1002/fes3.110
Park J, Song WY, Ko D, Eom Y, Hansen TH, Schiller M, Lee TG, Martinoia E, Lee Y (2012) The phytochelatin transporters AtABCC1 and AtABCC2 mediate tolerance to cadmium and mercury. Plant J 69(2):278–288. https://doi.org/10.1111/j.1365-313X.2011.04789.x
Pompeu GB, Vilhena MB, Gratão PL, Carvalho RF, Rossi ML, Martinelli AP, Azevedo RA (2017) Abscisic acid-deficient sit tomato mutant responses to cadmium-induced stress. Protoplasma 254(2):771–783. https://doi.org/10.1007/s00709-016-0989-4
Puschenreiter M, Türktas M, Sommer P, Wieshammer G, Laaha G, Wenzel WW, Hauser M-T (2010) Differentiation of metallicolous and non-metallicolous Salix caprea populations based on phenotypic characteristics and nuclear microsatellite (SSR) markers. Plant Cell Environ 33(10):1641–1655. https://doi.org/10.1111/j.1365-3040.2010.02170.x
Rui H, Chen C, Zhanga X, Shena X, Zhang F (2016) Cd-induced oxidative stress and lignification in the roots of two Vicia sativa L. varieties with different Cd tolerances. J Hazard Mater 301:304–313. https://doi.org/10.1016/j.jhazmat.2015.08.052
SAS Institute. (2011). SAS/STAT user’s guide: version 9.3. SAS Institute, Cary
Sasaki A, Yamaji N, Yokosho K, Ma JF (2012) Nramp5 is a major transporter responsible for manganese and cadmium uptake in rice. Plant Cell 24(5):2155–2167. https://doi.org/10.1105/tpc.112.096925
Science Communication Unit. Science for environment policy in-depth report: soil contamination: impacts on human health (2013) Report produced for the European Commission DG Environment. Available at: http://ec.europa.eu/science-environment-policy
Sebastian A, Prasad MNV (2016a) Modulatory role of mineral nutrients on cadmium accumulation and stress tolerance in Oryza sativa L. seedlings. Environ Sci Pollut Res 23(2):1224–1233. https://doi.org/10.1007/s11356-015-5346-1
Sebastian A, Prasad MNV (2016b) Iron plaque decreases cadmium accumulation in Oryza sativa L. and serves as a source of iron. Plant Biol 18(6):1008–1015. https://doi.org/10.1111/plb.12484
Seth CS, Misraa V, Chauhan LKS, Singh RR (2008) Genotoxicity of cadmium on root meristem cells of Allium cepa: cytogenetic and comet assay approach. Ecotoxicol Environ Saf 71(3):711–716. https://doi.org/10.1016/j.ecoenv.2008.02.003
Sharmila P, Kumari PK, Singh K, Prasad NVSRK, Pardha-Saradhi P (2017) Cadmium toxicity-induced proline accumulation is coupled to iron depletion. Protoplasma 254(2):763–770. https://doi.org/10.1007/s00709-016-0988-5
Shi HP, Feng Y, Wang YL, Tsang PKE (2014) Effect of cadmium on cytogenetic toxicity in hairy roots of Wedelia trilobata L. and their alleviation by exogenous CaCl2. Environ Sci Pollut Res 21(2):1436–1443. https://doi.org/10.1007/s11356-013-2015-0
Shi Q, Wang J, Zou J, Jiang Z, Wu H, Wang J, Jiang W, Liu D (2016) Cadmium localization and its toxic effects on root tips of barley. Zemdirbyste-Agricul 103(2):151–158. https://doi.org/10.13080/z-a.2016.103.020
Štolfa I, Pfeiffer TŽ, Špoljarić D, Teklić T, Lončarić Z (2015) Heavy metal-induced oxidative stress in plants: response of the antioxidative system. In: Gupta D, Palma J, Corpas F (eds) Reactive oxygen species and oxidative damage in plants under stress. Switzerland, Springer Inter Pub, pp 127–163. https://doi.org/10.1007/978-3-319-20421-5_6
Stroinski A (1997) Effects of cadmium on the host-pathogen system. V. Effect of exogenous dicyclohexylamine on potato tubers (Solanum tuberosum L.), cadmium and Phytophthora infestans relations. Plant Physiol 150(1-2):178–183. https://doi.org/10.1016/S0176-1617(97)80199-X
Teklić T, Lončarić Z, Kovačević V, Singh BR (2013) Metallic trace elements in cereal grain—a review: how much metal do we eat? Food Energy Secur 2(2):81–95. https://doi.org/10.1002/fes3.24
Thomine S, Wang R, Ward JM, Crawford NM, Schroeder JI (2000) Cadmium and iron transport by members of a plant metal transporter family in Arabidopsis with homology to Nramp genes. Proc Natl Acad Sci U S A 97(9):4991–4996. https://doi.org/10.1073/pnas.97.9.4991
Truyens S, Beckers B, Thijs S, Weyens N, Cuypers A, Vangronsveld J (2016) Cadmium-induced and trans-generational changes in the cultivable and total seed endophytic community of Arabidopsis thaliana. Plant Biol 18(3):376–381. https://doi.org/10.1111/plb.12415
Truyens S, Jambon I, Croes S, Janssen J, Weyens N, Mench M, Carleer R, Cuypers A, Vangronsveld J (2014) The effect of long-term Cd and Ni exposure on seed endophytes of Agrostis capillaris and their potential application in phytoremediation of metal-contaminated soils. Int J Phytoremed 16(7-8):643–659. https://doi.org/10.1080/15226514.2013.837027
Truyens S, Weyens N, Cuypers A, Vangronsveld J (2013) Changes in the population of seed bacteria of transgenerationally Cd-exposed Arabidopsis thaliana. Plant Biol 15(6):971–981. https://doi.org/10.1111/j.1438-8677.2012.00711.x
Uraguchi S, Kiyono M, Sakamoto T, Watanabe I, Kuno K (2009) Contributions of apoplasmic cadmium accumulation, antioxidative enzymes and induction of phytochelatins in cadmium tolerance of the cadmium-accumulating cultivar of black oat (Avena strigosa Schreb.) Planta 230(2):267–276. https://doi.org/10.1007/s00425-009-0939-x
Wu D, Yamaji N, Yamane M, Kashino-Fujii M, Sato K, Ma JF (2016) The HvNramp5 transporter mediates uptake of cadmium and manganese, but not iron. Plant Physiol 172(3):1899–1910. https://doi.org/10.1104/pp.16.01189
Yan YF, Choi DH, Kim DS, Li BWJ (2010) Genotypic variation of cadmium accumulation and distribution in rice. J Crop Sci Biotech 13(2):69–73. https://doi.org/10.1007/s12892-010-0036-5
Zhang S, Zhang H, Qin R, Jiang W, Liu D (2009) Cadmium induction of lipid peroxidation and effects on root tip cells and antioxidant enzyme activities in Vicia faba L. Ecotoxicology 18(7):814–823. https://doi.org/10.1007/s10646-009-0324-3
Zhu Z-J, Sun J-W, Fang X-Z, Qian Q-Q, Yang Z-E (2004) Genotypic differences in effects of cadmium exposure on plant growth and contents of cadmium and elements in 14 cultivars of bai cai. J Environ Sci Health B 39(4):675–687. https://doi.org/10.1081/PFC-200026905
Acknowledgements
R.A.A. thanks Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq-Brazil, 303749/2016-4) for the research fellowship. We are also grateful to Dr. Salete Gaziola for technical assistance, and Dr. Cláudio Roberto Segatelli and Aparecido da Silva for the help during crop management.
Funding
This work was funded by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP—Grants 2009/54676-0 and 2013/15217-5).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Bhumi Nath Tripathi
Electronic supplementary material
ESM 1
(DOCX 23 kb)
Rights and permissions
About this article
Cite this article
Carvalho, M.E.A., Piotto, F.A., Nogueira, M.L. et al. Cadmium exposure triggers genotype-dependent changes in seed vigor and germination of tomato offspring. Protoplasma 255, 989–999 (2018). https://doi.org/10.1007/s00709-018-1210-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-018-1210-8