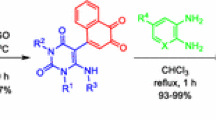
Abstract
A facile and selective one-pot N-acylation of nucleobases via carboxylic acids using cyanuric chloride (2,4,6-trichloro-1,3,5-triazine) is described. In this protocol, the reaction of diverse carboxylic acids with pyrimidine and/or purine nucleobases is efficiently achieved in the presence of cyanuric chloride and NaH/Et3N in DMF/MeCN (1:1, v/v) to afford the corresponding N-acyl nucleobases in good yields.
Graphical Abstract

Similar content being viewed by others
References
Kleeman A, Engel J, Kutscher B, Reichert D (1999) Pharmaceutical substances, 3rd edn. Thieme, Stuttgart
De Clercq E (1993) In: Johnsson NG (ed) Advances in antiviral drug design, vol 1. JAI Press, Greenwich, p 88
Pathak T (2002) Chem Rev 102:1623
Agrofoglio LA, Gillaizeau I, Saito Y (2003) Chem Rev 103:1875
Huryn DM, Okabe M (1992) Chem Rev 92:1745
Khalafi-Nezhad A, Soltani Rad MN, Moosavi-Movahedi AA, Kosari M (2007) Helv Chim Acta 90:730
Khalafi-Nezhad A, Soltani Rad, MN, Khoshnood A (2004) Synthesis 583
Khalafi-Nezhad A, Zarea A, Soltani Rad MN, Mokhtari B Parhami A (2005) Synthesis 419
Choi JR, Cho DG, Roh KY, Hwang JT, Ahn S, Jang HS, Cho WY, Kim KW, Cho YG, Kim J, Kim YZ (2004) J Med Chem 47:2864
Amblard F, Nolan SP, Schinazi RF, Agrofoglio LA (2005) Tetrahedron 61:537
Bryant JD, Keyser GE, Barrio JR (1979) J Org Chem 44:3733
Herdewijn P (2008) Modified nucleosides. In: Biochemistry, biotechnology and medicine. Wiley-VCH, Weinheim
Chu CK (2002) Recent advances in nucleosides: chemistry and chemotherapy. Elsevier Science, Amsterdam
Romeo G, Chiacchio U, Corsaro A, Merino P (2010) Chem Rev 110:3337
Mieczkowski A, Roy V, Agrofoglio LA (2010) Chem Rev 110:1828
Cruickshank KA, Jiricny J, Reese CB (1984) Tetrahedron Lett 25:681
Ti GS, Gaffney BL, Jones RA (1982) J Am Chem Soc 104:1316
Zhu XF, Williams HJ, Scott AI (2003) Synth Commun 33:1233
Zhang ZL, Han P, Ma XY, Liu J, Wang XW, Liu JY (2005) Chin Chem Lett 16:287
Nováček A, Hesoun D, Gut J (1965) Collect Czech Chem Commun 30:1890
Spector LB, Keller EB (1958) J Biol Chem 232:185
Bhat V, Ugarkar BG, Sayeed VA, Grimm K, Kosora N, Domenico PA, Stocker E (1989) Nucleosides Nucleotides 8:179
Kumar A, Singh D, Wani MJ, Joshi NS, Thombre PS, Rawat AS (2000) Process for preparation of acyclic nucleosides. (1997) Chem Abstr 127:346617. US Patent 6,043,364, March 28 2000
Blotny G (2006) Tetrahedron 62:9507 and references cited therein
Soltani Rad MN, Behrouz S (2011) Synlett 2562
Soltani Rad MN, Khalafi-Nezhad A, Asrari Z, Behrouz S, Amini Z, Behrouz M (2009) Synthesis 3983
Soltani Rad MN, Khalafi-Nezhad A, Asrari Z, Behrouz S (2010) Synthesis 2599
Soltani Rad MN, Khalafi-Nezhad A, Behrouz S (2009) Helv Chim Acta 92:1760
Soltani Rad MN, Khalafi-Nezhad A, Behrouz S (2010) Beilstein J Org Chem 6:49
Soltani Rad MN, Khalafi-Nezhad A, Behrouz S, Faghihi MA, Zare A, Parhami A (2008) Tetrahedron 64:1778
Soltani Rad MN, Khalafi-Nezhad A, Behrouz S, Asrari Z, Behrouz M, Amini Z (2009) Synthesis 3067
Soltani Rad MN, Behrouz S, Doroodmand MM, Moghtaderi N (2011) Synthesis 3915
Soltani Rad MN, Khalafi-Nezhad A, Divar M, Behrouz S (2010) Phosphorus. Sulfur Silicon Relat Elem 185:1943
Venkataraman K, Wagle DR (1979) Tetrahedron Lett 20:3037
Bandgar BP, Pandit SS (2002) Tetrahedron Lett 43:3413
Kaminski ZJ, Paneth P, Rudzinski J (1998) J Org Chem 63:4248
Rayle HL, Fellmeth L (1999) Org Process Res Dev 3:172
Gambacorta’ A, Farah ME, Tofani D (1999) Tetrahedron Lett 55:12615
Pal A, Salandria KJ, Arico JW, Schlegel MK, McLaughlin LW (2013) Chem Commun 49:2936
Acknowledgments
We wish to thank the Shiraz University of Technology and Shiraz University Research Councils for partial support of this work.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Soltani Rad, M.N., Behrouz, S., Asrari, Z. et al. A simple and regioselective one-pot procedure for the direct N-acylation of some purine and pyrimidine nucleobases via carboxylic acids using cyanuric chloride. Monatsh Chem 145, 1933–1940 (2014). https://doi.org/10.1007/s00706-014-1270-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-014-1270-1