Abstract
Background and aims
Various techniques have been investigated to enhance peripheral nerve regeneration including the application of low-intensity electrical stimulation (ES) and the administration of growth factors, especially brain-derived neurotrophic factor (BDNF). The purpose of this study was to investigate the effects of combining short-term (ES) and recombinant adenoviral vector-mediated BDNF (BDNF-Ad) transfer, in comparison to each sole modality, on peripheral nerve regeneration in a rat model with crush-injured sciatic nerve.
Methods
Sixty male Sprague–Dawley rats (250-300 g) were equally distributed into four groups; the control group, the ES group, the BDNF-Ad group, and the combination group (n = 15 each). A standard crush injury was introduced to the sciatic nerve. The control group received no treatment after injury, the ES group received 30 minutes of low-intensity ES, the BDNF-Ad group received an injection of recombinant BDNF-Ad (concentration = 1011 pfu/μl, 3 μl/rat) after injury, and the combination group received both ES and BDNF-Ad. The rats were followed-up for 3 weeks.
Results
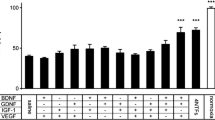
At the end of the follow-up period, the sciatic function index (ES =−39, BDNF-Ad =−38) and number of the retrogradely labeled sensory neurons were significantly increased in the ES group and the BDNF-Ad group (ES = 326, BDNF-Ad = 264), but not in the combined treatment group, compared to the control group (SFI = −53, retrogradely labeled neurons = 229). Axonal counts were highest in the ES group (7,208 axons), axonal densities in the BDNF group (10,598 axons/mm2), and the myelin thickness was greater in both groups as compared to the control group. The combined treatment group showed no signs of superior recovery compared to the other groups.
Conclusions
Both the ES and the BDNF-Ad treatments were effective techniques enhancing the sciatic nerve regeneration following a crush injury in rats. Nevertheless, the combined treatment with ES and BDNF-Ad produces neither a synergistic effect nor an improvement in this injury model.






Similar content being viewed by others
References
Al-Majed AA, Brushart TM, Gordon T (2000) Electrical stimulation accelerates and increases expression of BDNF and trkB mRNA in regenerating rat femoral motoneurons. Eur J Neurosci 12:4381–4390
Al-Majed AA, Neumann CM, Brushart TM, Gordon T (2000) Brief electrical stimulation promotes the speed and accuracy of motor axonal regeneration. J Neurosci 20:2602–2608
Alrashdan MS, Park JC, Sung MA, Yoo SB, Jahng JW, Lee TH, Kim SJ, Lee JH (2010) Thirty minutes of low intensity electrical stimulation promotes nerve regeneration after sciatic nerve crush injury in a rat model. Acta Neurol Belg 110:168–179
Asensio-Pinilla E, Udina E, Jaramillo J, Navarro X (2009) Electrical stimulation combined with exercise increase axonal regeneration after peripheral nerve injury. Exp Neurol 219:258–265
Bain JR, Mackinnon SE, Hunter DA (1989) Functional evaluation of complete sciatic, peroneal, and posterior tibial nerve lesions in the rat. Plast Reconstr Surg 83:129–138
Boyd JG, Gordon T (2002) A dose-dependent facilitation and inhibition of peripheral nerve regeneration by brain-derived neurotrophic factor. Eur J Neurosci 15:613–626
Boyd JG, Gordon T (2003) Glial cell line-derived neurotrophic factor and brain-derived neurotrophic factor sustain the axonal regeneration of chronically axotomized motoneurons in vivo. Exp Neurol 183:610–619
Chan JR, Cosgaya JM, Wu YJ, Shooter EM (2001) Neurotrophins are key mediators of the myelination program in the peripheral nervous system. Proc Natl Acad Sci USA 98:14661–14668
Chikar JA, Colesa DJ, Swiderski DL, Di Polo A, Raphael Y, Pfingst BE (2008) Over-expression of BDNF by adenovirus with concurrent electrical stimulation improves cochlear implant thresholds and survival of auditory neurons. Hear Res 245:24–34
Davidson BL, Allen ED, Kozarsky KF, Wilson JM, Roessler BJ (1993) A model system for in vivo gene transfer into the central nervous system using an adenoviral vector. Nat Genet 3:219–223
Dijkhuizen PA, Pasterkamp RJ, Hermens WT, de Winter F, Giger RJ, Verhaagen J (1998) Adenoviral vector-mediated gene delivery to injured rat peripheral nerve. J Neurotrauma 15:387–397
Fu SY, Gordon T (1997) The cellular and molecular basis of peripheral nerve regeneration. Mol Neurobiol 14:67–116
Geremia NM, Gordon T, Brushart TM, Al-Majed AA, Verge VM (2007) Electrical stimulation promotes sensory neuron regeneration and growth-associated gene expression. Exp Neurol 205:347–359
Glatzel M, Flechsig E, Navarro B, Klein MA, Paterna JC, Bueler H, Aguzzi A (2000) Adenoviral and adeno-associated viral transfer of genes to the peripheral nervous system. Proc Natl Acad Sci USA 97:442–447
Gordon T, Sulaiman O, Boyd JG (2003) Experimental strategies to promote functional recovery after peripheral nerve injuries. J Peripher Nerv Syst 8:236–250
Hammarberg H, Piehl F, Risling M, Cullheim S (2000) Differential regulation of trophic factor receptor mRNAs in spinal motoneurons after sciatic nerve transection and ventral root avulsion in the rat. J Comp Neurol 426:587–601
Herz J, Gerard RD (1993) Adenovirus-mediated transfer of low density lipoprotein receptor gene acutely accelerates cholesterol clearance in normal mice. Proc Natl Acad Sci USA 90:2812–2816
Kline DG, Hudson AR (1995) Nerve injuries: operative results for major nerve injuries, entrapments, and tumors. W.B. Saunders, Philadelphia
Koda M, Kamada T, Hashimoto M, Murakami M, Shirasawa H, Sakao S, Ino H, Yoshinaga K, Koshizuka S, Moriya H, Yamazaki M (2007) Adenovirus vector-mediated ex vivo gene transfer of brain-derived neurotrophic factor to bone marrow stromal cells promotes axonal regeneration after transplantation in completely transected adult rat spinal cord. Eur Spine J 16:2206–2214
Liu Q, Muruve DA (2003) Molecular basis of the inflammatory response to adenovirus vectors. Gene Ther 10:935–940
Mendonca AC, Barbieri CH, Mazzer N (2003) Directly applied low-intensity direct electric current enhances peripheral nerve regeneration in rats. J Neurosci Meth 129:183–190
Meyer M, Matsuoka I, Wetmore C, Olson L, Thoenen H (1992) Enhanced synthesis of brain-derived neurotrophic factor in the lesioned peripheral nerve: different mechanisms are responsible for the regulation of BDNF and NGF mRNA. J Cell Biol 119:45–54
Nix WA, Hopf HC (1983) Electrical stimulation of regenerating nerve and its effect on motor recovery. Brain Res 272:21–25
Novikov L, Novikova L, Kellerth JO (1997) Brain-derived neurotrophic factor promotes axonal regeneration and long-term survival of adult rat spinal motoneurons in vivo. Neuroscience 79:765–774
Obata K, Noguchi K (2006) BDNF in sensory neurons and chronic pain. Neurosci Res 55:1–10
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108
Sunderland S (1978) Nerves and nerve injuries. Churchill Livingstone, Edinburgh, NY, distributed by Longman
Thomas CE, Birkett D, Anozie I, Castro MG, Lowenstein PR (2001) Acute direct adenoviral vector cytotoxicity and chronic, but not acute, inflammatory responses correlate with decreased vector-mediated transgene expression in the brain. Mol Ther 3:36–46
Watanabe TS, Ohtori S, Koda M, Aoki Y, Doya H, Shirasawa H, Yamazaki M, Moriya H, Takahashi K, Yamashita T (2006) Adenoviral gene transfer in the peripheral nervous system. J Orthop Sci 11:64–69
Yan Q, Elliott J, Snider WD (1992) Brain-derived neurotrophic factor rescues spinal motor neurons from axotomy-induced cell death. Nature 360:753–755
Yang Y, Nunes FA, Berencsi K, Furth EE, Gonczol E, Wilson JM (1994) Cellular immunity to viral antigens limits E1-deleted adenoviruses for gene therapy. Proc Natl Acad Sci USA 91:4407–4411
Zhang JY, Luo XG, Xian CJ, Liu ZH, Zhou XF (2000) Endogenous BDNF is required for myelination and regeneration of injured sciatic nerve in rodents. Eur J Neurosci 12:4171–4180
Zhang Y, Zheng Y, Zhang YP, Shields LBE, Hu X, Yu P, Burke DA, Wang H, Jun C, Byers J, Whittemore SR, Shields CB (2010) Enhanced adenoviral gene delivery to motor and dorsal root ganglion neurons following injection into demyelinated peripheral nerves. J Neurosci Res 88:2374–2384
Zhou XF, Rush RA (1996) Endogenous brain-derived neurotrophic factor is anterogradely transported in primary sensory neurons. Neuroscience 74:945–953
Acknowledgements
This study was supported by a grant from the Korea Healthcare technology R&D Project, Ministry for Health, Welfare & Family Affairs, Republic of Korea. (A101578)
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alrashdan, M.S., Sung, MA., Kim Kwon, Y. et al. Effects of combining electrical stimulation with BDNF gene transfer on the regeneration of crushed rat sciatic nerve. Acta Neurochir 153, 2021–2029 (2011). https://doi.org/10.1007/s00701-011-1054-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-011-1054-x