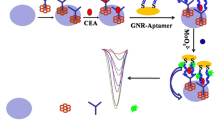
Abstract
A magnetic bead (MB)-based sandwich biorecognition reactions is combined with a gold nanoprobe-induced homogenous synthesis of molybdophosphate to develop a novel bioassay method for the electrochemical detection of the tumor biomarker of carcinoembryonic antigen (CEA). The nanoprobe is prepared through the specific loading of numerous alkaline phosphatase (ALP)-functionalized gold nanoparticles (Au NPs) on a double-stranded DNA (dsDNA) produced by the CEA aptamer-triggered hybridization chain reaction (HCR). Both the large amounts of PO43− produced by the ALP catalytic hydrolysis of pyrophosphate and the phosphate backbones of dsDNA can react with the added MoO42− to generate electroactive molybdophosphates. So, the gold nanoprobe was used for signal tracing of the sandwich bioassay of CEA at a constructed antibody-functionalized MB platform. The sensitive electrochemical measurement of molybdophosphate produced from the quantitatively captured nanoprobes at a carbon nanotube-modified electrode (measured at about 0.12 V vs. Ag/AgCl, 3 M KCl) enabled the convenient signal transduction of the method. Due to the dually enhanced synthesis of molybdophosphate by the HCR and multi-enzyme Au NP nanotags, this method shows a wide linear range from 0.05 pg mL−1 to 10 ng mL−1 along with a low detection limit of 0.027 pg mL−1. In addition, the MB-based biorecognition reaction and the homogeneous synthesis of molybdophosphate are much convenient in manipulations. These excellent performances decide the extensive application potentials of the method.

A magnetic bead-based bioassay method was simply developed for the electrochemical detection of carcinoembryonic antigen. The dually enhanced homogenous synthesis of molybdophosphate by hybridization chain reaction (HCR) and enzyme nanotags and the sensitive electrochemical measurement of molybdophosphate at a carbon nanotube (CNT)-electrode enable ultrasensitive signal transduction of the method.





Similar content being viewed by others
References
Labib M, Sargent EH, Kelley SO (2016) Electrochemical methods for the analysis of clinically relevant biomolecules. Chem Rev 116:9001–9090
Ghorbani F, Abbaszadeh H, Mehdizadeh A, Ebrahimi-Warkiani M, Rashidi MR (2019) Biosensors and nanobiosensors for rapid detection of autoimmune diseases: a review. Microchim Acta 186:838
Devi RV, Doble M, Verma RS (2015) Nanomaterials for early detection of cancer biomarker with special emphasis on gold nanoparticles in immunoassays/sensors. Biosens Bioelectron 68:688–698
Song S, Qin Y, He Y, Huang Q, Fan C, Chen HY (2010) Functional nanoprobes for ultrasensitive detection of biomolecules. Chem Soc Rev 39:4234–4243
Pei X, Zhang B, Tang J, Liu B, Lai W, Tang D (2013) Sandwich type immunosensors and immunoassays exploiting nanostructure labels: a review. Anal Chim Acta 758:1–18
Luo Y, Wang Y, Yan H, Wu Y, Zhu C, Du D, Lin Y (2018) SWCNTs@GQDs composites as nanocarriers for enzyme-free dual-signal amplification electrochemical immunoassay of cancer biomarker. Anal Chim Acta 1042:44–51
Xu L, Liu Z, Lei S, Huang D, Zou L, Ye B (2019) A sandwich-type electrochemical aptasensor for the carcinoembryonic antigen via biocatalytic precipitation amplification and by using gold nanoparticle composites. Microchim Acta 186:473
Parsa SF, Vafajoo A, Rostami A, Salarian R, Rabiee M, Rabiee N, Rabiee G, Tahririd M, Yadegari A, Vashaee D, Tayebi L, Hamblin MR (2018) Early diagnosis of disease using microbead array technology: a review. Anal Chim Acta 1032:1–17
Li B, Lai G, Lin B, Yu A, Yang N (2018) Enzyme-induced biomineralization of cupric subcarbonate for ultrasensitive colorimetric immunosensing of carcinoembryonic antigen. Sensors Actuators B Chem 262:789–795
Jie G, Ge J, Gao X, Li C (2018) Amplified electrochemiluminescence detection of CEA based on magnetic Fe3O4@ Au nanoparticles-assembled Ru@SiO2 nanocomposites combined with multiple cycling amplification strategy. Biosens Bioelectron 118:115–121
Liu Z, Lei S, Zou L, Li G, Xu L, Ye B (2019) A label-free and double recognition-amplification novel strategy for sensitive and accurate carcinoembryonic antigen assay. Biosens Bioelectron 131:113–118
He Y, Chen S, Huang L, Wang Z, Wu Y, Fu F (2018) Combination of magnetic-beads-based multiple metal nanoparticles labeling with hybridization chain reaction amplification for simultaneous detection of multiple cancer cells with inductively coupled plasma mass spectrometry. Anal Chem 91:1171–1177
Li B, Qin L, Zhou J, Cai X, Lai G, Yu A (2019) Hybridization chain reaction-enhanced enzyme biomineralization for ultrasensitive colorimetric biosensing of a protein biomarker. Analyst 144:5003–5009
Yu X, Zhang ZL, Zheng SY (2015) Highly sensitive DNA detection using cascade amplification strategy based on hybridization chain reaction and enzyme-induced metallization. Biosens Bioelectron 66:520–526
Chen X, Wang Y, Zhang J, Zhang Y (2019) DNA concatemer-silver nanoparticles as a signal probe for electrochemical prostate-specific antigen detection. Analyst 144:6313–6320
Liu Y, Ding Y, Gao Y, Liu R, Hu X, Lv Y (2018) Enzyme-free amplified DNA assay: five orders of linearity provided by metal stable isotope detection. Chem Commun 54:13782–13785
Xie S, Yuan Y, Chai Y, Yuan R (2015) Tracing phosphate ions generated during loop-mediated isothermal amplification for electrochemical detection of nosema bombycis genomic DNA PTP1. Anal Chem 87:10268–10274
Xu J, Cao X, Xia J, Gong S, Wang Z, Lu L (2016) Phosphomolybdic acid functionalized graphene loading copper nanoparticles modified electrodes for non-enzymatic electrochemical sensing of glucose. Anal Chim Acta 934:44–51
Tang Q, Zhang L, Tan X, Jiao L, Wei Q, Li H (2019) Bioinspired synthesis of organic-inorganic hybrid nanoflowers for robust enzyme-free electrochemical immunoassay. Biosens Bioelectron 133:94–99
Shen C, Li X, Rasooly A, Guo L, Zhang K, Yang M (2016) A single electrochemical biosensor for detecting the activity and inhibition of both protein kinase and alkaline phosphatase based on phosphate ions induced deposition of redox precipitates. Biosens Bioelectron 85:220–225
Hua X, Zhou X, Guo S, Zheng T, Yuan R, Xu W (2019) Determination of Alzheimer biomarker DNA by using an electrode modified with in-situ precipitated molybdophosphate catalyzed by alkaline phosphatase-encapsulated DNA hydrogel and target recycling amplification. Microchim Acta 186:158
Hu L, Hu S, Guo L, Shen C, Yang M, Rasooly A (2017) DNA generated electric current biosensor. Anal Chem 89:2547–2552
Feng K, Liu J, Deng L, Yu H, Yang M (2018) Amperometric detection of microRNA based on DNA-controlled current of a molybdophosphate redox probe and amplification via hybridization chain reaction. Microchim Acta 185:28
Li X, Shen C, Yang M, Rasooly A (2018) Polycytosine DNA electric-current-generated immunosensor for electrochemical detection of human epidermal growth factor receptor 2 (HER2). Anal Chem 90:4764–4769
Si Z, Xie B, Chen Z, Tang C, Li T, Yang M (2017) Electrochemical aptasensor for the cancer biomarker CEA based on aptamer induced current due to formation of molybdophosphate. Microchim Acta 184:3215–3221
Nie Y, Yang M, Ding Y (2018) Gold nanoparticle enhanced hybridization chain reaction as a method for signal amplification. Application to electrochemical immunodetection of the ovarian cancer biomarker carbohydrate antigen 125. Microchim Acta 185:331
Yi J, Liu Z, Liu J, Liu H, Xia F, Tian D, Zhou C (2020) A label-free electrochemical aptasensor based on 3D porous CS/rGO/GCE for acetamiprid residue detection. Biosens Bioelectron 148:111827
Li B, Lai G, Zhang H, Hu S, Yu A (2017) Copper chromogenic reaction based colorimetric immunoassay for rapid and sensitive detection of a tumor biomarker. Anal Chim Acta 963:106–111
Lai G, Zheng M, Hu W, Yu A (2017) One-pot loading high-content thionine on polydopamine-functionalized mesoporous silica nanosphere for ultrasensitive electrochemical immunoassay. Biosens Bioelectron 95:15–20
Karami K, Bayat P, Allafchian AR, Amiri R, Rezaei B, Laurents DV (2019) A new glucose biosensor based on nickel/KH550 nanocomposite deposited on the GCE: an electrochemical study. J Electroanal Chem 839:9–15
Lai G, Zhang H, Yong J, Yu A (2013) In situ deposition of gold nanoparticles on polydopamine functionalized silica nanosphere for ultrasensitive nonenzymatic electrochemical immunoassay. Biosens Bioelectron 47:178–183
Zhao L, Li C, Qi H, Gao Q, Zhang C (2016) Electrochemical lectin-based biosensor array for detection and discrimination of carcinoembryonic antigen using dual amplification of gold nanoparticles and horseradish peroxidase. Sensors Actuators B Chem 235:575–582
Yang Z, Lan Q, Li J, Wu J, Tang Y, Hu X (2017) Efficient streptavidin-functionalized nitrogen-doped graphene for the development of highly sensitive electrochemical immunosensors. Biosens Bioelectron 89:312–318
Nakhjavani SA, Afsharan H, Khalilzadeh B, Ghahremani MH, Carrara S, Omidi Y (2019) Gold and silver bio/nano-hybrids-based electrochemical immunosensor for ultrasensitive detection of carcinoembryonic antigen. Biosens Bioelectron 141:111439
Jia Y, Li Y, Zhang S, Wang P, Liu Q, Dong Y (2020) Mulberry-like Au@PtPd porous nanorods composites as signal amplifers for sensitive detection of CEA. Biosens Bioelectron 149:111842
Akter R, Rhee CK, Rahman MA (2014) Sensitivity enhancement of an electrochemical immunosensor through the electrocatalysis of magnetic bead-supported non-enzymatic labels. Biosens Bioelectron 54:351–357
Funding
This work was financially supported by the National Natural Science Foundation of China (21475033) and Science and Technology Foundation for Excellent Creative Research Group of Hubei Provincial Department of Education (T201810).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All serum sample experiments were approved by the Ethics Committee of Hubei Normal University, and informed consent was obtained from human participants of this study.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 378 kb)
Rights and permissions
About this article
Cite this article
Cai, X., Lv, F., Lai, G. et al. Dually enhanced homogenous synthesis of molybdophosphate by hybridization chain reaction and enzyme nanotags for the electrochemical bioassay of carcinoembryonic antigen. Microchim Acta 187, 361 (2020). https://doi.org/10.1007/s00604-020-04342-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-020-04342-3