Abstract
Purpose
Limited data about oral mucositis (OM) in stem cell transplant patients with underlying hematological disease is available in Germany. The purpose of this feasibility study was to determine the incidence, treatment patterns, patients’ adherence, and costs of OM.
Methods
Prospective, noninterventional single-center observational study. Inclusion criteria: allogenic/autologous stem cell transplant patients ≥ 18 years, high-dose chemotherapy. OM assessment: WHO Oral Toxicity Scale. Adherence was measured in patient interviews. Preventive and therapeutic measures were extracted from patients’ charts.
Results
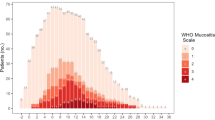
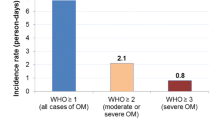
Forty-five patients (25 allogenic, 20 autologous) were enrolled. Twenty-six (58%) patients developed OM (54% grade I/II, 46% grade III/IV). Age ≥ 65 (31% vs 69%, p = 0.021) was associated with a lower OM incidence. A positive history of smoking (1.77 vs 2.69, p = 0.036) was associated with a lower OM grade, patients with unrelated donors (2.63 vs 1.29, p = 0.014) were associated with higher OM grades and females (80% vs 47%, RR = 1.71, p = 0.035) with a higher incidence. OM patients were less adherent to recommended daily mouth rinses (35% vs 68%, p = 0.027). More analgesic treatment (80% vs 32%, p = 0.001) and intravenous opioids (24% vs 0%, p = 0.023) were prescribed in OM patients. Total drug treatment and nutrition costs were 824€ (p = 0.037) higher in autologous transplanted patients.
Conclusion
Initial risk and consecutive OM assessment, determination of patients’ adherence, resource consumption, and costs are prerequisites to evaluate OM care. In the best case, several centers will follow the same methodological approach and the collected data will serve as a basis for benchmarking analyses to optimize OM care where required.


Similar content being viewed by others
References
Passweg JR, Baldomero H, Bader P, Bonini C, Cesaro S, Dreger P, Duarte RF, Dufour C, Kuball J, Farge-Bancel D, Gennery A, Kroger N, Lanza F, Nagler A, Sureda A, Mohty M (2016) Hematopoietic stem cell transplantation in Europe 2014: more than 40 000 transplants annually. Bone marrow transplantation 51(6):786–792. https://doi.org/10.1038/bmt.2016.20
Cherny NI (2009) Stigma associated with "palliative care": getting around it or getting over it. Cancer 115(9):1808–1812. https://doi.org/10.1002/cncr.24212
Cherny N (2011) Best supportive care: a euphemism for no care or a standard of good care? Seminars in oncology 38(3):351–357. https://doi.org/10.1053/j.seminoncol.2011.03.002
Staudenmaier T, Cenzer I, Crispin A, Ostermann H, Berger K (2018) Burden of oral mucositis in stem cell transplant patients-the patients' perspective. Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer 26(5):1577–1584. https://doi.org/10.1007/s00520-017-4000-5
Bellm LA, Epstein JB, Rose-Ped A, Martin P, Fuchs HJ (2000) Patient reports of complications of bone marrow transplantation. Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer 8(1):33–39
Elting LS, Cooksley C, Chambers M, Cantor SB, Manzullo E, Rubenstein EB (2003) The burdens of cancer therapy. Clinical and economic outcomes of chemotherapy-induced mucositis. Cancer 98(7):1531–1539
Scully C, Sonis S, Diz PD (2006) Oral mucositis. Oral Dis 12(3):229–241
Lalla RV, Sonis ST, Peterson DE (2008) Management of oral mucositis in patients with cancer. Dental clinics of North America 52(1):61-viii. https://doi.org/10.1016/j.cden.2007.10.002
Jones JA, Qazilbash MH, Shih YC, Cantor SB, Cooksley CD, Elting LS (2008) In-hospital complications of autologous hematopoietic stem cell transplantation for lymphoid malignancies: clinical and economic outcomes from the Nationwide Inpatient Sample. Cancer 112(5):1096–1105. https://doi.org/10.1002/cncr.23281
Urbain P, Raynor A, Bertz H, Lambert C, Biesalski HK (2012) Role of antioxidants in buccal mucosa cells and plasma on the incidence and severity of oral mucositis after allogeneic haematopoietic cell transplantation. Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer 20(8):1831–1838. https://doi.org/10.1007/s00520-011-1284-8
Chaudhry HM, Bruce AJ, Wolf RC, Litzow MR, Hogan WJ, Patnaik MS, Kremers WK, Phillips GL, Hashmi SK (2016) The incidence and severity of oral mucositis among allogeneic hematopoietic stem cell transplantation patients: a systematic review. Biol Blood Marrow Transplant 22(4):605–616. https://doi.org/10.1016/j.bbmt.2015.09.014
Berger K, Schopohl D, Bollig A, Strobach D, Rieger C, Rublee D, Ostermann H (2018) Burden of Oral Mucositis: A Systematic Review and Implications for Future Research. Oncology research and treatment 41(6):399–405. https://doi.org/10.1159/000487085
Lalla RV, Peterson DE (2005) Oral mucositis. Dental Clinics of North America 49(1):167–184. https://doi.org/10.1016/j.cden.2004.07.009
Sonis ST, Oster G, Fuchs H, Bellm L, Bradford WZ, Edelsberg J, Hayden V, Eilers J, Epstein JB, LeVeque FG, Miller C, Peterson DE, Schubert MM, Spijkervet FK, Horowitz M (2001) Oral mucositis and the clinical and economic outcomes of hematopoietic stem-cell transplantation. J Clin Oncol 19(8):2201–2205
Bezinelli LM, de Paula EF, da Graca Lopes RM, Biazevic MG, de Paula EC, Correa L, Hamerschlak N, Michel-Crosato E (2014) Cost-effectiveness of the introduction of specialized oral care with laser therapy in hematopoietic stem cell transplantation. Hematological oncology 32(1):31–39. https://doi.org/10.1002/hon.2050
Villa A, Sonis ST (2015) Mucositis: pathobiology and management. Current opinion in oncology 27(3):159–164. https://doi.org/10.1097/cco.0000000000000180
Bollig A (2016) Die Krankheitslast der oralen Mukositis. Department of hematology-oncology University Hospital of Munich, https://core.ac.uk/download/pdf/79056039.pdf. Access 25.03.2019 18:00.
Biswal BM (2008) Current trends in the management of oral mucositis related to cancer treatment. The Malaysian journal of medical sciences : MJMS 15(3):4–13
World Health Organization (1979) Handbook for reporting results of cancer treatment. World Health Organization, Geneva, Switzerland
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. American journal of clinical oncology 5(6):649–655
Aaronson NKAS, Bergman B, Bullinger M, Cull A, Duez NJ, Filiberti A, Flechtner HFS, de Haes JCJM, Kaasa S, Klee MC, Osoba D, Razavi D, Rofe PBSS, Sneeuw KCA, Sullivan M, Takeda F (1993) The European Organisation for Research and Treatment of Cancer QLQ-C30: A quality-of-life instrument for use in international clinical trials in oncology. Journal of the National Cancer Institute 85:365–376
McCann S, Schwenkglenks M, Bacon P, Einsele H, D'Addio A, Maertens J, Niederwieser D, Rabitsch W, Roosaar A, Ruutu T, Schouten H, Stone R, Vorkurka S, Quinn B, Blijlevens N (2009) The Prospective Oral Mucositis Audit: relationship of severe oral mucositis with clinical and medical resource use outcomes in patients receiving high-dose melphalan or BEAM-conditioning chemotherapy and autologous SCT. Bone marrow transplantation 43(2):141–147. https://doi.org/10.1038/bmt.2008.299
Sakellari I, Angelopoulou M, Tsopra O, Dervenoulas I, Tsirigotis P, Spyridonidis A, Liga M, Tsionos K, Anargyrou K, Pouli A, Anagnostopoulos A (2015) A prospective study of incidence, clinical and quality of life consequences of oral mucositis post palifermin prophylaxis in patients undergoing high-dose chemotherapy and autologous hematopoietic cell transplantation. Annals of hematology 94(10):1733–1740. https://doi.org/10.1007/s00277-015-2437-5
Sobecks RM, Rybicki L, Yurch M, Kalaycio M, Dean R, Andresen S, Pohlman B, Duong H, Bolwell B, Copelan E (2012) Intravenous compared with oral busulfan as preparation for allogeneic hematopoietic progenitor cell transplantation for AML and MDS. Bone marrow transplantation 47(5):633–638. https://doi.org/10.1038/bmt.2011.167
Blijlevens N, Schwenkglenks M, Bacon P, D'Addio A, Einsele H, Maertens J, Niederwieser D, Rabitsch W, Roosaar A, Ruutu T, Schouten H, Stone R, Vokurka S, Quinn B, McCann S (2008) Prospective oral mucositis audit: oral mucositis in patients receiving high-dose melphalan or BEAM conditioning chemotherapy--European Blood and Marrow Transplantation Mucositis Advisory Group. J Clin Oncol 26(9):1519–1525. https://doi.org/10.1200/jco.2007.13.6028
Dodd MJ, Miaskowski C, Shiba GH, Dibble SL, Greenspan D, MacPhail L, Paul SM, Larson P (1999) Risk factors for chemotherapy-induced oral mucositis: dental appliances, oral hygiene, previous oral lesions, and history of smoking. Cancer investigation 17(4):278–284
Patussi C, Sassi LM, Munhoz EC, Zanicotti RT, Schussel JL (2014) Clinical assessment of oral mucositis and candidiasis compare to chemotherapic nadir in transplanted patients. Brazilian oral research 28:1–7
Mahood DJ, Dose AM, Loprinzi CL, Veeder MH, Athmann LM, Therneau TM, Sorensen JM, Gainey DK, Mailliard JA, Gusa NL et al (1991) Inhibition of fluorouracil-induced stomatitis by oral cryotherapy. J Clin Oncol 9(3):449–452
Villa A, Woo SB (2017) Leukoplakia-a diagnostic and management algorithm. Journal of oral and maxillofacial surgery : official journal of the American Association of Oral and Maxillofacial Surgeons 75(4):723–734. https://doi.org/10.1016/j.joms.2016.10.012
McCarthy GM, Awde JD, Ghandi H, Vincent M, Kocha WI (1998) Risk factors associated with mucositis in cancer patients receiving 5-fluorouracil. Oral oncology 34(6):484–490
Sonis ST, Fey EG (2002) Oral complications of cancer therapy. Oncology (Williston Park, NY) 16(5):680–686 discussion 686, 691-682, 695
Robien K, Schubert MM, Bruemmer B, Lloid ME, Potter JD, Ulrich CM (2004) Predictors of oral mucositis in patients receiving hematopoietic cell transplants for chronic myelogenous leukemia. J Clin Oncol 22(7):1268–1275
Zalcberg J, Kerr D, Seymour L, Palmer M (1998) Haematological and non-haematological toxicity after 5-fluorouracil and leucovorin in patients with advanced colorectal cancer is significantly associated with gender, increasing age and cycle number. Tomudex International Study Group. European journal of cancer (Oxford, England : 1990) 34(12):1871–1875
Vokurka S, Bystricka E, Koza V, Scudlova J, Pavlicova V, Valentova D, Visokaiova M, Misaniova L (2006) Higher incidence of chemotherapy induced oral mucositis in females: a supplement of multivariate analysis to a randomized multicentre study. Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer 14(9):974–976. https://doi.org/10.1007/s00520-006-0031-z
Yamagata K, Arai C, Sasaki H, Takeuchi Y, Onizawa K, Yanagawa T, Ishibashi N, Karube R, Shinozuka K, Hasegawa Y, Chiba S, Bukawa H (2012) The effect of oral management on the severity of oral mucositis during hematopoietic SCT. Bone marrow transplantation 47(5):725–730. https://doi.org/10.1038/bmt.2011.171
Elad S, Raber-Durlacher JE, Brennan MT, Saunders DP, Mank AP, Zadik Y, Quinn B, Epstein JB, Blijlevens NM, Waltimo T, Passweg JR, Correa ME, Dahllof G, Garming-Legert KU, Logan RM, Potting CM, Shapira MY, Soga Y, Stringer J, Stokman MA, Vokurka S, Wallhult E, Yarom N, Jensen SB (2015) Basic oral care for hematology-oncology patients and hematopoietic stem cell transplantation recipients: a position paper from the joint task force of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) and the European Society for Blood and Marrow Transplantation (EBMT). Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer 23(1):223–236. https://doi.org/10.1007/s00520-014-2378-x
Rubenstein EB, Peterson DE, Schubert M, Keefe D, McGuire D, Epstein J, Elting LS, Fox PC, Cooksley C, Sonis ST (2004) Clinical practice guidelines for the prevention and treatment of cancer therapy-induced oral and gastrointestinal mucositis. Cancer 100(9 Suppl):2026–2046. https://doi.org/10.1002/cncr.20163
Lalla RV, Bowen J, Barasch A, Elting L, Epstein J, Keefe DM, McGuire DB, Migliorati C, Nicolatou-Galitis O, Peterson DE, Raber-Durlacher JE, Sonis ST, Elad S (2014) MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 120(10):1453–1461
Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft DK, AWMF) (2017) Supportive Therapie bei onkologischen PatientInnen, Langversion 1.1 Leitlinienreport, 2017, AWMF Registernummer: 032/054OL, http://leitlinienprogramm-onkologie.de/Supportive-Therapie.95.0.html. Access: 21.01.2018
Min YH, Lee JW, Shin Y-W, Jo M-W, Sohn G, Lee J-H, Lee G, Jung KH, Sung J, Ko BS, Yu J-H, Kim HJ, Son BH, Ahn SH (2014) Daily collection of self-reporting sleep disturbance data via a smartphone app in breast cancer patients receiving chemotherapy: a feasibility study. Journal of medical Internet research 16(5):e135–e135. https://doi.org/10.2196/jmir.3421
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of human rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Ethics committee of the Faculty of Medicine, Ludwig-Maximilians-University Munich, 345-16) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Berger, K., Staudenmaier, T., Cenzer, I. et al. Epidemiology, patient adherence, and costs of oral mucositis in routine care in stem cell transplantation. Support Care Cancer 28, 3113–3123 (2020). https://doi.org/10.1007/s00520-019-05107-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-019-05107-2