Abstract
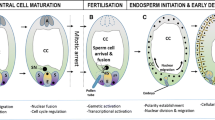
Development of the seed endosperm involves several different types of coordinated cell cycle programs: acytokinetic mitosis, which produces a syncytium soon after fertilization; cellularization through the formation of modified phragmoplasts; cell proliferation, in which mitosis is coupled to cell division; and, in certain species like cereal crops, endoreduplication. Understanding the regulation of these programs and their transitions is challenging, but it has the potential to define important links between the cell cycle, cell differentiation and development, as well as provide tools for the manipulation of seed yield. A relatively large number of mutants display endosperm proliferation defects, and connections with known cell cycle genes are beginning to emerge. For example, it is becoming increasingly evident that the master cell cycle regulators, the cyclin-dependent kinases and retinoblastoma-related families, play key roles in the events leading to endosperm formation and development. Recent studies highlight cross-talk between pathways controlling the cell cycle and genomic imprinting.


Similar content being viewed by others
References
Ach RA, Durfee T, Miller AB, Taranto P, Hanley-Bowdoin L, Zambryski PC, Gruissem W (1997a) RRB1 and RRB2 encode maize retinoblastoma-related proteins that interact with a plant D-type cyclin and geminivirus replication protein. Mol Cell Biol 17:5077–5086
Ach RA, Taranto P, Gruissem W (1997b) A conserved family of WD-40 proteins binds to the retinoblastoma protein in both plants and animals. Plant Cell 9:1595–1606
Artlip TS, Madison JT, Setter TL (1995) Water-deficit in developing endosperm of maize cell division and nuclear DNA endoreduplication. Plant Cell Environ 18:1034–1040
Bennett MD, Smith JB, Barclay I (1975) Early seed development in the Triticeae. Philos Trans R Soc Lond Biol Sci B 272:199–227
Berger F, Grini PE, Schnittger A (2006) Endosperm: an integrator of seed growth and development. Curr Opin Plant Biol 9:664–670
Boisnard-Lorig C, Colon-Carmona A, Bauch M, Hodge S, Doerner P, Bancharel E, Dumas C, Haseloff J, Berger F (2001) Dynamic analyses of the expression of the HISTONE::YFP fusion protein in Arabidopsis show that syncytial endosperm is divided in mitotic domains. Plant Cell 13:495–509
Brink R, Cooper D (1947) The endosperm in seed development. Bot Rev 13:423–477
Brown RC, Lemmon BE, Olsen OA (1994) Endosperm development in barley: microtubule involvement in the morphogenetic pathway. Plant Cell 6:1241–1252
Brown RC, Lemmon BE, Nguyen H, Olsen O-A (1999) Development of endosperm in Arabidopsis thaliana. Sex Plant Reprod 12:32–42
Brown RC, Lemmon BE, Nguyen H (2003) Events during the first four rounds of mitosis establish three developmental domains in the syncytial endosperm of Arabidopsis thaliana. Protoplasma 222:167–174
Canales C, Bhatt AM, Scott R, Dickinson H (2002) EXS, a putative LRR receptor kinase, regulates male germline cell number and tapetal identity and promotes seed development in Arabidopsis. Curr Biol 12:1718–1727
Cavallini A, Natali L, Balconi C, Rizzi E, Motto M, Cionini G, Damato F (1995) Chromosome endoreduplication in endosperm cells of two maize genotypes and their progenies. Protoplasma 189:156–162
Chaudhury AM, Ming L, Miller C, Craig S, Dennis ES, Peacock WJ (1997) Fertilization-independent seed development in Arabidopsis thaliana. Proc Natl Acad Sci USA 94:4223–4228
Chen Z, Hafidh S, Poh SH, Twell D, Berger F (2009) Proliferation and cell fate establishment during Arabidopsis male gametogenesis depends on the Retinoblastoma protein. Proc Natl Acad Sci USA 106:7257–7262
Choi Y, Gehring M, Johnson L, Hannon M, Harada JJ, Goldberg RB, Jacobsen SE, Fischer RL (2002) DEMETER, a DNA glycosylase domain protein, is required for endosperm gene imprinting and seed viability in Arabidopsis. Cell 110:33–42
Chojecki AJS, Bayliss MW, Gale MD (1986) Cell production and DNA accumulation in the wheat endosperm, and their association with grain weight. Ann Bot 58:809–817
Coelho CM, Dante RA, Sabelli PA, Sun Y, Dilkes BP, Gordon-Kamm WJ, Larkins BA (2005) Cyclin-dependent kinase inhibitors in maize endosperm and their potential role in endoreduplication. Plant Physiol 138:2323–2336
Collinge MA, Spillane C, Kohler C, Gheyselinck J, Grossniklaus U (2004) Genetic interaction of an origin recognition complex subunit and the Polycomb group gene MEDEA during seed development. Plant Cell 16:1035–1046
Cooper DC, Brink RA (1942) The endosperm as a barrier to interspecific hybridization in flowering plants. Science 95:75–76
Costa LM, Gutierrez-Marcos JF (2008) Retinoblastoma makes its mark on imprinting in plants. PLoS Biol 6:1631–1633
Costa LM, Gutierrez-Marcos JF, Brutnell TP, Greenland AJ, Dickinson HG (2003) The globby1–1 (glo1–1) mutation disrupts nuclear and cell division in the developing maize seed causing alterations in endosperm cell fate and tissue differentiation. Development 130:5009–5017
Danilevskaya ON, Hermon P, Hantke S, Muszynski MG, Kollipara K, Ananiev EV (2003) Duplicated fie genes in maize: expression pattern and imprinting suggest distinct functions. Plant Cell 15:425–438
Day RC, Herridge RP, Ambrose BA, Macknight RC (2008) Transcriptome analysis of proliferating Arabidopsis endosperm reveals biological implications for the control of syncytial division, cytokinin signaling, and gene expression regulation. Plant Physiol 148:1964–1984
Dilkes BP, Dante RA, Coelho C, Larkins BA (2002) Genetic analyses of endoreduplication in Zea mays endosperrn: Evidence of sporophytic and zygotic maternal control. Genetics 160:1163–1177
Doan DNP, Linnestad C, Olsen O-A (1996) Isolation of molecular markers from the barley endosperm coenocyte and the surrounding nucellus cell layers. Plant Mol Biol 31:877–886
Doelling JH, Yan N, Kurepa J, Walker J, Vierstra RD (2001) The ubiquitin-specific protease UBP14 is essential for early embryo development in Arabidopsis thaliana. Plant J 27:393–405
Drea S, Leader DJ, Arnold BC, Shaw P, Dolan L, Doonan JH (2005) Systematic spatial analysis of gene expression during wheat caryopsis development. Plant Cell 17:2172–2185
Ebel C, Mariconti L, Gruissem W (2004) Plant retinoblastoma homologues control nuclear proliferation in the female gametophyte. Nature 429:776–780
Engelen-Eigles G, Jones RJ, Phillips RL (2001) DNA endoreduplication in maize endosperm cells is reduced by high temperature during the mitotic phase. Crop Sci 41:1114–1121
Garcia D, Saingery V, Chambrier P, Mayer U, Jurgens G, Berger F (2003) Arabidopsis haiku mutants reveal new controls of seed size by endosperm. Plant Physiol 131:1661–1670
Garcia D, Fitz Gerald JN, Berger F (2005) Maternal control of integument cell elongation and zygotic control of endosperm growth are coordinated to determine seed size in Arabidopsis. Plant Cell 17:52–60
Gehring M, Huh JH, Hsieh T-F, Penterman J, Choi Y, Harada JJ, Goldberg RB, Fischer RL (2006) DEMETER DNA glycosylase establishes MEDEA Polycomb gene self-imprinting by allele-specific demethylation. Cell 124:495–506
Grafi G, Larkins BA (1995) Endoreduplication in maize endosperm—involvement of M-phase-promoting factor inhibition and induction of S-phase-related kinases. Science 269:1262–1264
Grafi G, Burnett RJ, Helentjaris T, Larkins BA, DeCaprio JA, Sellers WR, Kaelin WG (1996) A maize cDNA encoding a member of the retinoblastoma protein family: Involvement in endoreduplication. Proc Natl Acad Sci USA 93:8962–8967
Grimanelli D, Perotti E, Ramirez J, Leblanc O (2005) Timing of the maternal-to-zygotic transition during early seed development in maize. Plant Cell 17:1061–1072
Grini PE, Jurgens G, Hulskamp M (2002) Embryo and endosperm development is disrupted in the female gametophytic capulet mutants of Arabidopsis. Genetics 162:1911–1925
Grossniklaus U, Vielle-Calzada J-P, Hoeppner MA, Gagliano WB (1998) Maternal control of embryogenesis by MEDEA, a Polycomb Group gene in Arabidopsis. Science 280:446–450
Guitton AE, Page DR, Chambrier P, Lionnet C, Faure JE, Grossniklaus U, Berger F (2004) Identification of new members of Fertilisation Independent Seed Polycomb Group pathway involved in the control of seed development in Arabidopsis thaliana. Development 131:2971–2981
Gutierrez C (2005) Coupling cell proliferation and development in plants. Nat Cell Biol 7:535–541
Gutierrez-Marcos JF, Costa LM, Pra MD, Scholten S, Kranz E, Perez P, Dickinson HG (2006) Epigenetic asymmetry of imprinted genes in plant gametes. Nat Genet 38:876–878
Hermon P, Srilunchang K-o, Zou J, Dresselhaus T, Danilevskaya O (2007) Activation of the imprinted Polycomb Group Fie1 gene in maize endosperm requires demethylation of the maternal allele. Plant Mol Biol 64:387–395
Holding DR, Springer PS (2002) The Arabidopsis gene PROLIFERA is required for proper cytokinesis during seed development. Planta 214:373–382
Ingouff M, Gerald JNF, Guerin C, Robert H, Sørensen MB, Damme DV, Geelen D, Blanchoin L, Berger F (2005a) Plant formin AtFH5 is an evolutionarily conserved actin nucleator involved in cytokinesis. Nat Cell Biol 7:374–380
Ingouff M, Haseloff J, Berger F (2005b) Polycomb group genes control developmental timing of endosperm. Plant J 42:663–674
Ingouff M, Jullien PE, Berger F (2006) The female gametophyte and the endosperm control cell proliferation and differentiation of the seed coat in Arabidopsis. Plant Cell 18:3491–3501
John PCL (2007) Hormonal regulation of cell cycle progression and its role in development. In: Inze D (ed) Cell cycle control and plant development. Blackwell, Oxford, pp 311–334
Johnston AJ, Matveeva E, Kirioukhova O, Grossniklaus U, Gruissem W (2008) A dynamic reciprocal RBR-PRC2 regulatory circuit controls Arabidopsis gametophyte development. Curr Biol 18:1680–1686
Jones RJ, Schreiber BMN, Roessler JA (1996) Kernel sink capacity in maize: genotypic and maternal regulation. Crop Sci 36:301–306
Jullien PE, Katz A, Oliva M, Ohad N, Berger F (2006) Polycomb Group complexes self-regulate imprinting of the Polycomb Group gene MEDEA in Arabidopsis. Curr Biol 16:486–492
Jullien PE, Mosquna A, Ingouff M, Sakata T, Ohad N, Berger F (2008) Retinoblastoma and its binding partner MSI1 control imprinting in Arabidopsis. Plos Biol 6:1693–1705
Kang I-H, Steffen JG, Portereiko MF, Lloyd A, Drews GN (2008) The AGL62 MADS domain protein regulates cellularization during endosperm development in Arabidopsis. Plant Cell 20:635–647
Kiesselbach TA (1949) The structure and reproduction of corn. Research Bulletin 161. University of Nebraska College of Agriculture, Lincoln, NE
Kiesselbach TA (1999) The structure and reproduction of corn. 50th anniversary edition. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Kiyosue T, Ohad N, Yadegari R, Hannon M, Dinneny J, Wells D, Katz A, Margossian L, Harada JJ, Goldberg RB, Fischer RL (1999) Control of fertilization-independent endosperm development by the MEDEA polycomb gene in Arabidopsis. Proc Natl Acad Sci USA 96:4186–4191
Kladnik A, Chourey PS, Pring DR, Dermastia M (2006) Development of the endosperm of Sorghum bicolor during the endoreduplication-associated growth phase. J Cereal Sci 43:209–215
Kohler C, Hennig L, Bouveret R, Gheyselinck J, Grossniklaus U, Gruissem W (2003) Arabidopsis MSI1 is a component of the MEA/FIE Polycomb group complex and required for seed development. EMBO J 22:4804–4814
Kowles RV, Phillips RL (1985) DNA amplification patterns in maize endosperm nuclei during kernel development. Proc Natl Acad Sci USA 82:7010–7014
Kowles RV, Phillips RL (1988) Endosperm development in maize. Int Rev Cyt 112:97–136
Kowles RV, McMullen MD, Yerk G, Phillips RL, Kraemer S, Srienc F (1992) Endosperm mitotic-activity and endoreduplication in maize affected by defective kernel mutations. Genome 35:68–77
Kowles RV, Yerk GL, Haas KM, Phillips RL (1997) Maternal effects influencing DNA endoreduplication in developing endosperm of Zea mays. Genome 40:798–805
Larkins BA, Dilkes BP, Dante RA, Coelho CM, Woo YM, Liu Y (2001) Investigating the hows and whys of DNA endoreduplication. J Exp Bot 52:183–192
Laudencia-Chingcuanco D, Stamova B, You F, Lazo G, Beckles D, Anderson O (2007) Transcriptional profiling of wheat caryopsis development using cDNA microarrays. Plant Mol Biol 63:651–668
Leblanc O, Pointe C, Hernandez M (2002) Cell cycle progression during endosperm development in Zea mays depends on parental dosage effects. Plant J 32:1057–1066
Leiva-Neto JT, Grafi G, Sabelli PA, Woo YM, Dante RA, Maddock S, Gordon-Kamm WJ, Larkins BA (2004) A dominant negative mutant of cyclin-dependent kinase A reduces endoreduplication but not cell size or gene expression in maize endosperm. Plant Cell 16:1854–1869
Lepiniec L, Devic M, Berger F (2005) Genetic and molecular control of seed development in Arabidopsis. In: Lester D (ed) Plant functional genomics. Food Products Press, Birghamton, pp 511–564
Lersten NR (2004) Flowering plant embryology. Blackwell, Ames, Iowa
Lid S, Al R, Krekling T, Meeley R, Ranch J, Opsahl-Ferstad H-G, Olsen O-A (2004) The maize disorganized aleurone layer 1 and 2 (dil1, dil2) mutants lack control of the mitotic division plane in the aleurone layer of developing endosperm. Planta 218:370–378
Liu C-M, Meinke DW (1998) The titan mutants of Arabidopsis are disrupted in mitosis and cell cycle control during seed development. Plant J 16:21–31
Liu C-M, McElver J, Tzafrir I, Joosen R, Wittich P, Patton D, Van Lammeren AAM, Meinke DW (2002) Condensin and cohesin knockouts in Arabidopsis exhibit a titan seed phenotype. Plant J 29:405–415
Liu X, Fu J, Gu D, Liu W, Liu T, Peng Y, Wang J, Wang G (2008) Genome-wide analysis of gene expression profiles during the kernel development of maize (Zea mays L.). Genomics 91:378–387
Luo M, Bilodeau P, Koltunow A, Dennis ES, Peacock WJ, Chaudhury AM (1999) Genes controlling fertilization-independent seed development in Arabidopsis thaliana. Proc Natl Acad Sci USA 96:296–301
Luo M, Bilodeau P, Dennis ES, Peacock WJ, Chaudhury A (2000) Expression and parent-of-origin effects for FIS2, MEA, and FIE in the endosperm and embryo of developing Arabidopsis seeds. Proc Natl Acad Sci USA 97:10637–10642
Luo M, Dennis ES, Berger F, Peacock WJ, Chaudhury A (2005) MINISEED3 (MINI3), a WRKY family gene, and HAIKU2 (IKU2), a leucine-rich repeat (LRR) KINASE gene, are regulators of seed size in Arabidopsis. Proc Natl Acad Sci USA 102:17531–17536
Lur HS, Setter TL (1993) Role of auxin in maize endosperm development—timing of nuclear DNA endoreduplication, zein expression, and cytokinin. Plant Physiol 103:273–280
Mambelli S, Setter TL (1998) Inhibition of maize endosperm cell division and endoreduplication by exogenously applied abscisic acid. Physiol Plant 104:266–277
Mathieu O, Reinders J, Caikovski M, Smathajitt C, Paszkowski J (2007) Transgenerational stability of the Arabidopsis epigenome is coordinated by CG methylation. Cell 130:851–862
Mayer U, Herzog U, Berger F, Inze D, Jurgens G (1999) Mutations in the pilz group genes disrupt the microtubule cytoskeleton and uncouple cell cycle progression from cell division in Arabidopsis embryo and endosperm. Eur J Cell Biol 78:100–108
Menand B, Desnos T, Nussaume L, Berger F, Bouchez D, Meyer C, Robaglia C (2002) Expression and disruption of the Arabidopsis TOR (target of rapamycin) gene. Proc Natl Acad Sci USA 99:6422–6427
Mosquna A, Katz A, Shochat S, Grafi G, Ohad N (2004) Interaction of FIE, a Polycomb protein, with pRb: a possible mechanism regulating endosperm development. Mol Genet Genom 271:651–657
Nacry P, Mayer U, Jurgens G (2000) Genetic dissection of cytokinesis. Plant Mol Biol 43:719–733
Neuffer MG, Coe EH, Wessler SR (1997) Mutants of maize. Cold Spring Harbor Laboratory Press, New York
Nowack MK, Grini PE, Jakoby MJ, Lafos M, Koncz C, Schnittger A (2006) A positive signal from the fertilization of the egg cell sets off endosperm proliferation in angiosperm embryogenesis. Nat Genet 38:63–67
Nowack MK, Shirzadi R, Dissmeyer N, Dolf A, Endl E, Grini PE, Schnittger A (2007) Bypassing genomic imprinting allows seed development. Nature 447:312–315
Ohad N, Margossian L, Hsu Y-C, Williams C, Repetti P, Fischer RL (1996) A mutation that allows endosperm development without fertilization. Proc Natl Acad Sci USA 93:5319–5324
Ohad N, Yadegari R, Margossian L, Hannon M, Michaeli D, Harada JJ, Goldberg RB, Fischer RL (1999) Mutations in FIE, a WD polycomb group gene, allow endosperm development without fertilization. Plant Cell 11:407–416
Olsen OA (2001) Endosperm development: cellularization and cell fate specification. Ann Rev Plant Physiol Plant Mol Biol 52:233–267
Olsen OA (2004) Nuclear endosperm development in cereals and Arabidopsis thaliana. Plant Cell 16:214–227
Olsen O-A, Linnestad C, Nichols SE (1999) Developmental biology of the cereal endosperm. Trends Plant Sci 4:253–257
Otegui M (2007) Endosperm cell walls: formation, composition, and functions. In: Olsen OA (ed) Endosperm. Springer-Verlag, Berlin-Heidelberg, pp 159–177
Otegui M, Staehelin LA (2000) Syncytial-type cell plates: a novel kind of cell plate involved in endosperm cellularization of Arabidopsis. Plant Cell 12:933–947
Pignocchi C, Minns GE, Nesi N, Koumproglou R, Kitsios G, Benning C, Lloyd CW, Doonan JH, Hills MJ (2009) ENDOSPERM DEFECTIVE1 Is a novel microtubule-associated protein essential for seed development in Arabidopsis. Plant Cell 21:90–105
Randolph LF (1936) Developmental morphology of the caryopsis in maize. J Agric Res 53:881–916
Ronceret A, Guilleminot J, Lincker F, Gadea-Vacas J, Delorme V, Bechtold N, Pelletier G, Delseny M, Chabouté M-E, Devic M (2005) Genetic analysis of two Arabidopsis DNA polymerase epsilon subunits during early embryogenesis. Plant J 44:223–236
Rossi V, Locatelli S, Lanzanova C, Boniotti MB, Varotto S, Pipal A, Goralik-Schramel M, Lusser A, Gatz C, Gutierrez C, Motto M (2003) A maize histone deacetylase and retinoblastoma-related protein physically interact and cooperate in repressing gene transcription. Plant Mol Biol 51:401–413
Sabelli PA, Larkins BA (2006) Grasses like mammals? Redundancy and compensatory regulation within the retinoblastoma protein family. Cell Cycle 5:352–355
Sabelli PA, Larkins BA (2008) The endoreduplication cell cycle: regulation and function. In: Verma DPS, Hong Z (eds) Cell division control in plants. Springer-Verlag, Berlin, pp 75–100
Sabelli PA, Larkins BA (2009) The development of endosperm in grasses. Plant Physiol 149:14–26
Sabelli PA, Dante RA, Leiva-Neto JT, Jung R, Gordon-Kamm WJ, Larkins BA (2005a) RBR3, a member of the retinoblastoma-related family from maize, is regulated by the RBR1/E2F pathway. Proc Natl Acad Sci USA 102:13005–13012
Sabelli PA, Leiva-Neto JT, Dante RA, Nguyen H, Larkins BA (2005b) Cell cycle regulation during maize endosperm development. Maydica 50:485–496
Sabelli PA, Nguyen H, Larkins BA (2007) Cell cycle and endosperm development. In: Inze D (ed) Cell cycle control and plant development. Blackwell, Oxford, pp 294–310
Sabelli PA, Hoerster G, Lizarraga LE, Brown SW, Gordon-Kamm WJ, Larkins BA (2009) Positive regulation of minichromosome maintenance gene expression, DNA replication, and cell transformation by a plant retinoblastoma gene. Proc Natl Acad Sci USA 106:4042–4047
Scanlon MJ, Henderson DC, Bernstein B (2002) SEMAPHORE1 functions during the regulation of ancestrally duplicated knox genes and polar auxin transport in maize. Development 129:2663–2673
Scott R, Spielman M, Bailey J, Dickinson H (1998) Parent-of-origin effects on seed development in Arabidopsis thaliana. Development 125:3329–3341
Segui-Simarro J, Otegui MS, Austin RJ, Staehelin LA (2007) Plant cytokinesis—insights gained from electron tomography studies. In: Verma DPS, Hong Z (eds) Cell division control in plants. Springer-Verlag, Berlin, pp 251–287
Setter TL, Flannigan BA (2001) Water deficit inhibits cell division and expression of transcripts involved in cell proliferation and endoreduplication in maize endosperm. J Exp Bot 52:1401–1408
Shen W-H (2002) The plant E2F-Rb pathway and epigenetic control. Trends Plant Sci 7:505–511
Shen W-H, Parmentier Y, Hellmann H, Lechner E, Dong A, Masson J, Granier F, Lepiniec L, Estelle M, Genschik P (2002) Null mutation of AtCUL1 causes arrest in early embryogenesis in Arabidopsis. Mol Biol Cell 13:1916–1928
Sheridan WF, Neuffer MG (1980) Defective kernel mutants of maize II. Morphological and embryo culture studies. Genetics 95:945–960
Sørensen MB, Chaudhury AM, Robert H, Bancharel E, Berger F (2001) Polycomb group genes control pattern formation in plant seed. Curr Biol 11:277–281
Sørensen MB, Mayer U, Lukowitz W, Robert H, Chambrier P, Jurgens G, Somerville C, Lepiniec L, Berger F (2002) Cellularisation in the endosperm of Arabidopsis thaliana is coupled to mitosis and shares multiple components with cytokinesis. Development 129:5567–5576
Springer PS, Holding DR, Groover A, Yordan C, Martienssen RA (2000) The essential Mcm7 protein PROLIFERA is localized to the nucleus of dividing cells during the G(1) phase and is required maternally for early Arabidopsis development. Development 127:1815–1822
Sreenivasulu N, Altschmied L, Radchuk V, Gubatz S, Wobus U, Weschke W (2004) Transcript profiles and deduced changes of metabolic pathways in maternal and filial tissues of developing barley grains. Plant J 37:539–553
Steinborn K, Maulbetsch C, Priester B, Trautmann S, Pacher T, Geiges B, Kuttner F, Lepiniec L, Stierhof Y-D, Schwarz H, Jurgens G, Mayer U (2002) The Arabidopsis PILZ group genes encode tubulin-folding cofactor orthologs required for cell division but not cell growth. Genes Dev 16:959–971
Sun YJ, Dilkes BP, Zhang CS, Dante RA, Carneiro NP, Lowe KS, Jung R, Gordon-Kamm WJ, Larkins BA (1999a) Characterization of maize (Zea mays L.) Wee1 and its activity in developing endosperm. Proc Natl Acad Sci USA 96:4180–4185
Sun YJ, Flannigan BA, Setter TL (1999b) Regulation of endoreduplication in maize (Zea mays L.) endosperm. Isolation of a novel B1-type cyclin and its quantitative analysis. Plant Mol Biol 41:245–258
Tzafrir I, McElver JA, Liu C-m, Yang LJ, Wu JQ, Martinez A, Patton DA, Meinke DW (2002) Diversity of TITAN functions in Arabidopsis seed development. Plant Physiol 128:38–51
Ungru A, Nowack MK, Reymond M, Shirzadi R, Kumar M, Biewers S, Grini PE, Schnittger A (2008) Natural variation in the degree of autonomous endosperm formation reveals independence and constraints of embryo growth during seed development in Arabidopsis thaliana. Genetics 179:829–841
Varotto S, Locatelli S, Canova S, Pipal A, Motto M, Rossi V (2003) Expression profile and cellular localization of maize Rpd3-type histone deacetylases during plant development. Plant Physiol 133:606–617
Verma DPS, Hong Z (2001) Plant callose synthase complexes. Plant Mol Biol 47:693–701
Vielle-Calzada JP, Thomas J, Spillane C, Coluccio A, Hoeppner MA, Grossniklaus U (1999) Maintenance of genomic imprinting at the Arabidopsis medea locus requires zygotic DDM1 activity. Genes Dev 13:2971–2982
Vilhar B, Kladnik A, Blejec A, Chourey PS, Dermastia M (2002) Cytometrical evidence that the loss of seed weight in the miniature1 seed mutant of maize is associated with reduced mitotic activity in the developing endosperm. Plant Physiol 129:23–30
Wan Y, Poole R, Huttly A, Toscano-Underwood C, Feeney K, Welham S, Gooding M, Mills C, Edwards K, Shewry P, Mitchell R (2008) Transcriptome analysis of grain development in hexaploid wheat. BMC Genomics 9:121
Weijers D, Franke-van Dijk M, Vencken R-J, Quint A, Hooykaas P, Offringa R (2001) An Arabidopsis Minute-like phenotype caused by a semi-dominant mutation in a RIBOSOMAL PROTEIN S5 gene. Development 128:4289–4299
Williams L, Grafi G (2000) The retinoblastoma protein—a bridge to heterochromatin. Trends Plant Sci 5:239–240
Xie Q, Sanz-Burgos P, Hannon GJ, Gutierrez C (1996) Plant cells contain a novel member of the retinoblastoma family of growth regulatory proteins. EMBO J 15:4900–4908
Yu L-X, Setter TL (2003) Comparative transcriptional profiling of placenta and endosperm in developing maize kernels in response to water deficit. Plant Physiol 131:568–582
Zhao J, Grafi G (2000) The High Mobility Group I/Y protein is hypophosphorylated in end reduplicating maize endosperm cells and is involved in alleviating histone H1-mediated transcriptional repression. J Biol Chem 275:27494–27499
Zhou Y, Zhang X, Kang X, Zhao X, Zhang X, Ni M (2009) SHORT HYPOCOTYL UNDER BLUE1 associates with MINISEED3 and HAIKU2 promoters in vivo to regulate Arabidopsis seed development. Plant Cell 21:106–117
Acknowledgments
We thank the US Department of Energy (grant # DE-FG02-96ER20242) and Pioneer Hi-Bred Inc. for supporting the endosperm cell cycle project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Scott Russell.
Rights and permissions
About this article
Cite this article
Sabelli, P.A., Larkins, B.A. The contribution of cell cycle regulation to endosperm development. Sex Plant Reprod 22, 207–219 (2009). https://doi.org/10.1007/s00497-009-0105-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-009-0105-4