Abstract.
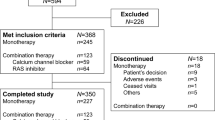
Essential hypertension in pediatric patients may require pharmacological treatment. There is a need for efficacious, safe, and well-tolerated antihypertensive agents with a once-a-day dosing regimen in children and adolescents. The aim of the trial was to evaluate the dose-response and tolerability of the dihydropyridine calcium channel blocker, felodipine extended-release tablets (felodipine ER), given once daily to pediatric patients with essential hypertension. A randomized double-blind, parallel-group, multi-center clinical study comparing felodipine ER (2.5, 5, or 10 mg once daily) and placebo was performed on pediatric patients with a baseline systolic (SBP) or diastolic blood pressure (DBP) above the 95th percentile for age, sex, and height. Of 133 randomized patients, 128 (96.2%) completed the 3 weeks of double-blind treatment. The study population included 50% children 6–12 years of age or Tanner stage ≤3, 39% black, and 40% female patients. All doses of felodipine ER and placebo decreased both SBP and DBP. The 5-mg dose of felodipine ER resulted in a significant decline in trough sitting, standing, and supine DBP (placebo-adjusted, −4.6, −5.06, and −5.05 mmHg, respectively, P<0.05) compared with placebo. However, there was no significant decline in SBP. The 2.5- and 10-mg doses showed no significant differences compared with placebo in lowering BP. The effect of felodipine ER was comparable in patient subgroups by age, race, and gender. The drug was well tolerated. The study failed to show a dose response of felodipine ER in pediatric patients with essential hypertension. This finding might be explained in part by the limitations of study design. The spectrum of side effects in pediatric patients was comparable with that in adults, except for a lower incidence of peripheral edema. Further studies are needed to clarify the role of felodipine ER in the management of children and adolescents with essential hypertension.
Similar content being viewed by others
References
National High Blood Pressure Education Program Working Group on Hypertension Control in Children and Adolescents (1996) Update on the 1987 Task Force Report on High Blood Pressure in Children and Adolescents: a working group report from the national high blood pressure education Program. Pediatric 98:649–658
Morgenstern BZ (1994) Hypertension in pediatric patients: current issues. Mayo Clin Proc 69:1089–1097
Arar MY, Hogg RJ, Arant Jr BS, Seikaly MG (1994) Etiology of sustained hypertension in children in the southwestern United States. Pediatr Nephrol 8:186–189
Sinaiko AR (1996) Hypertension in children. N Engl J Med 335:1968–1973
Sinaiko AR, Gomez-Marin O, Prineas RJ (1989) Prevalence of "significant" hypertension in junior high school-aged children: the children and adolescent blood pressure program. J Pediatr 114:664–669
Gillman MW, Cook NR, Rosner B, Evans DA, Keough ME, Taylor JO, Hennekens CH (1993) Identifying children at high risk for the development of essential hypertension. J Pediatr 122:837–846
Gauthier BG, Trachtman H (1991) The management of primary hypertension in the young. Pediatr Rev Commun 4:217–232
Wells T, Frame V, Soffer B, Shaw W, Zhang Z, Herrera P, Shahinfar S (2002) A double-blind, placebo-controlled, dose-response study of the effectiveness and safety of enalapril for children with hypertension. J Clin Pharmacol 42:870–880
Soffer B, Herrera P, Zhang Z, Miller K, Cano F, Simsolo R, Hernandez H, Shahinfar S (2002) Effects of the ACE inhibitor, lisinopril, in children age 6–16 years with hypertension (abstract). Pediatr Res 51:431A
Sorof JM, Cargo P, Graepel J, Humphrey D, King E, Rolf C, Cunningham RJ (2002) β-Blocker/thiazide combination for treatment of hypertensive children: a randomized, double-blind, placebo-controlled trial. Pediatr Nephrol 17:345–350
Flynn J, Smoyer WE, Bunchman TE (2000) Treatment of hypertensive children with amlodipine. Am J Hypertens 13:1061–1066
Rogan JW, Lyszkiewicz DA, Blowey DL, Khattack S, Arbus GS, Koren G (2000) A randomized prospective crossover trial of amlodipine in pediatric hypertension. Pediatr Nephrol 14:1083–1087
Falkner B, Onesti G, Lowenthal DT, Affrime MB (1982) Effectiveness of centrally acting drugs and diuretics in adolescent hypertension. Clin Pharmacol Ther 32:577–583
Dilman U, Caglar K, Senses A, Kinik E (1983) Nifedipine in hypertensive emergencies of children. Am J Dis Child 137:1162–1165
Evans JHC, Shaw NJ, Brocklebank JT (1988) Sublingual nifedipine in acute severe hypertension. Arch Dis Child 63:975–977
Egger DW, Deming DD, Hamada N, Perkin RM, Sahney S (2002) Evaluation of the safety of short-acting nifedipine in children with hypertension. Pediatr Nephrol 17:35–40
Flynn JT, Pasko DA (2000) Calcium channel blockers: pharmacology and place in therapy of pediatric hypertension. Pediatr Nephrol 15:302–316
Gomez-Marin O, Prineas RJ, Rastam L (1992) Cuff bladder width and blood pressure measurements in children and adolescents. J Hypertens 10:1235–1241
Chesney RW, Adamson P, Wells T, Wilson JT, Walson PD (2001) The testing of antihypertensive medications in children: report of the antihypertensive agent guidelines subcommittee of the pediatric pharmacology research units. Pediatrics 107:558–561
Sorof JM, Urbina EM, Cunningham RJ, Hogg RJ, Moxey-Mims M, Eissa MA, Rolf C, and the Ziac Pediatric Hypertension Study Group (2001) Screening eligibility in the study of antihypertensive medication in children: experience from the Ziac pediatric hypertension study. Am J Hypertens 14:783–787
Weaver MG, Park MK, Lee DH (1990) Differences in blood pressure levels obtained by auscultatory and oscillometric methods. Am J Dis Child 144:911–914
Blowey DL, Moncica I, Scolnik D, Arbus GS, Hebert D, Balfe JW, Koren G (1996) The pharmacokinetics of extended release felodipine in children. Eur J Clin Pharmacol 50:147–148
Moncica I, Oh PI, Qamar I ul, Scolnik D, Arbus GS, Hebert D, Balfe JW, Koren G (1995) A crossover comparison of extended release felodipine with prolonged action nifedipine in hypertension. Arch Dis Child 73:154–156
Anderson GD (2002) Children versus adults: pharmacokinetic and adverse-effect differences. Epilepsia 43 [Suppl 3]:53–59
Gauthier BG, Trachtman H (1997) Short-acting nifedipine. Pediatr Nephrol 11:786–787
Alderman MH, Cohen H, Roque R, Madhavan S (1997) Effect of long-acting and short-acting calcium antagonists on cardiovascular outcomes in hypertensive patients. Lancet 349:594–598
Furberg CD, Psaty BM, Meyer JV (1995) Nifedipine: dose-related increase in mortality in patients with coronary heart disease. Circulation 92:1326–1331
Opie LH (2001) Calcium channel blockers in hypertension: reappraisal after new trial and major meta-analyses. Am J Hypertens 14:1074–1081
Acknowledgement.
The authors would like to thank Michaelene Llewellyn and Scott Rosenberger for their efforts and support in managing this multi-center clinical trial.
Author information
Authors and Affiliations
Corresponding author
Additional information
The complete list of investigators and sites for the Plendil Pediatric Clinical Trial is listed in the Appendix
Appendix
Appendix
List of local principal investigators and participating sites where patients were enrolled in the study: Mazan Arar (transferred from Ihasan Elshibabi), University Hospital-Children's Kidney Center, San Antonio, Tex.; Noosha Baqi, State University of New York Health Science Center, Brooklyn, N.Y.; Mark Benfield (transferred from Judith Lynn Pugh), Children's Hospital of Alabama, Birmingham, Ala.; Stuart Berger, Children's Hospital of Wisconsin, Madison, Wis.; Robert J. Cunningham, Cleveland Clinic Foundation, Cleveland, Ohio; Ian G. Dresner, Children's Hospital Medical Center of Akron, Akron, Ohio; Bonita Falkner, Thomas Jefferson University, Philadelphia, Pa.; Eduardo Garin, University of South Florida Ambulatory Medical Clinics, Tampa, Fla.; Jens Goebel, University of Kentucky, Lexington, Ky.; Coral D. Hanevold, Children's Medical Center, Augusta, Ga.; Randall Jenkins, Norwest Pediatric Kidney Specialists, Portland, Ore.; Pedro Jose, Georgetown University Medical Center, Washington D.C.; Vijay Kusnoor, South East Texas Cardiology Associates, Beaumont, Tex.; John Mahan, Clinical Study Center, Columbus, Ohio; Tej Matoo, Children's Hospital of Michigan, Detroit, Mich.; Joel Melnick, Children's Memorial Hospital, Chicago, Ill.; Susan R. Mendley, University of Maryland Medical School, Baltimore, Md.; Kenneth Miller, Nephrology and Hypertension Consultants, Park Ridge, Ill.; Shashi Nagaraj, Wake Forest University, Winston-Salem, N.C.; Ronald Portman, Hermann Hospital, Houston, Tex.; Jorge A. Ramirez, Nemours Children's Clinic-Orlando, Orlando, Fla.; Irene Restaino, Children's Hospital of the King's Daughters, Norfolk, Va.; George A. Richard, Shands Hospital, Gainesville, Fla.; Ari Simckes, Children's Mercy Hospital, Kansas City, Mo.; W. Frank Tenny (transferred from Abdullah Sakarcan), Louisiana State University Health Sciences Center, Shreveport, La.; Frederick Strife, Children's Hospital Medical Center, Cincinnati, Ohio; Howard Trachtman, Schneider Children's Hospital, New Hyde Park, N.Y.; Lynn Weiss, Robert Wood Johnson Medical School, New Brunswick, N.J.; Matti Vehaskari, Children's Hospital, New Orleans, La.; Shefali Vyas (transferred from Andrea L. Zuckerman), New York Medical Center, Valhalla, N.Y.
Rights and permissions
About this article
Cite this article
Trachtman, H., Frank, R., Mahan, J.D. et al. Clinical trial of extended-release felodipine in pediatric essential hypertension. Pediatr Nephrol 18, 548–553 (2003). https://doi.org/10.1007/s00467-003-1134-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-003-1134-0