Abstract
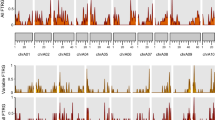
In a rapidly changing climate, flowering time (FL) adaptation is important to maximize seed yield in flax (Linum usitatissimum L.). However, our understanding of the genetic mechanism underlying FL in this multipurpose crop remains limited. With the aim of dissecting the genetic architecture of FL in flax, a genome-wide association study (GWAS) was performed on 200 accessions of the flax core collection evaluated in four environments. Two single-locus and six multi-locus models were applied using 70,935 curated single nucleotide polymorphism (SNP) markers. A total of 40 quantitative trait nucleotides (QTNs) associated with 27 quantitative trait loci (QTL) were identified in at least two environments. The number of QTL with positive-effect alleles in accessions was significantly correlated with FL (r = 0.77 to 0.82), indicating principally additive gene actions. Nine QTL were significant in at least three of the four environments accounting for 3.06–14.71% of FL variation. These stable QTL spanned regions that harbored 27 Arabidopsis thaliana and Oryza sativa FL-related orthologous genes including FLOWERING LOCUS T (Lus10013532), FLOWERING LOCUS D (Lus10028817), transcriptional regulator SUPERMAN (Lus10021215), and gibberellin 2-beta-dioxygenase 2 (Lus10037816). In silico gene expression analysis of the 27 FL candidate gene orthologous suggested that they might play roles in the transition from vegetative to reproductive phase, flower development and fertilization. Our results provide new insights into the QTL architecture of flowering time in flax, identify potential candidate genes for further studies, and demonstrate the effectiveness of combining different GWAS models for the genetic dissection of complex traits.




Similar content being viewed by others

References
Ambreen H, Kumar S, Kumar A, Agarwal M, Jagannath A, Goel S (2018) Association mapping for important agronomic traits in safflower (Carthamus tinctorius L.) core collection using microsatellite markers. Front Plant Sci 9:402. https://doi.org/10.3389/fpls.2018.00402
Andrés F, Coupland G (2012) The genetic basis of flowering responses to seasonal cues. Nat Rev Genet 13:627–639. https://doi.org/10.1038/nrg3291
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B (Methodological) 57:289–300. https://doi.org/10.2307/2346101
Bhatty RS, Cherdkiatgumchai P (1990) Compositional analysis of laboratory-prepared and commercial samples of linseed meal and of hull isolated from flax. J Am Oil Chem Soc. 67:79˗84. https://doi.org/10.1007/BF02540631
Bonnafous F, Fievet G, Blanchet N, Boniface MC, Carrère S, Gouzy J, Legrand L, Marage G, Bret-Mestries E, Munos S, Pouilly N, Vincourt P, Langlade N, Mangin B (2018) Comparison of GWAS models to identify non-additive genetic control of flowering time in sunflower hybrids. Theor Appl Genet 131:319–332. https://doi.org/10.1007/s00122-017-3003-4
Bradbury PJ, Zhang Z, Kroon DE, Casstevens TM, Ramdoss Y, Buckler ES (2007) TASSEL: software for association mapping of complex traits in diverse samples. Bioinformatics 23:2633–2635. https://doi.org/10.1093/bioinformatics/btm308
Cloutier S, You FM, Soto-Cerda BJ (2019) Linum genetic markers, maps, and QTL discovery. In: Cullis CA (ed) Genetics and genomics of linum plant genetics and genomics: crops and models, vol 23. Springer, Cham, pp 97–117
Craufurd PQ, Wheeler TR (2009) Climate change and the flowering time of annual crops. J Exp Bot 60:2529–2539. https://doi.org/10.1093/jxb/erp196
Cu ST, Guild G, Nicolson A, Velu G, Singh R, Stangoulis J (2020) Genetic dissection of zinc, iron, copper, manganese and phosphorus in wheat (Triticum aestivum L) grain and rachis at two developmental stages. Plant Sci 291:110338. https://doi.org/10.1016/j.plantsci.2019.110338
Das M, Haberer G, Panda A, Das Laha S, Ghosh TC, Schäffner AR (2016) Expression pattern similarities support the prediction of orthologs retaining common functions after gene duplication events. Plant Physiol 171:2343–2357. https://doi.org/10.1104/pp.15.01207
Davis SJ (2009) Integrating hormones into the floral-transition pathway of Arabidopsis thaliana. Plant Cell Environ 32:1201–1210. https://doi.org/10.1111/j.1365-3040.2009.01968.x
Devlin PF, Patel SR, Whitelam GC (1998) Phytochrome E influences internode elongation and flowering time in Arabidopsis. Plant Cell 10:1479–1487. https://doi.org/10.1105/tpc.10.9.1479
Diederichsen A, Kusters PM, Kessler D, Bainas Z, Gugel RK (2013) Assembling a core collection from the flax world collection maintained by Plant Gene Resources of Canada. Genet Resour Crop Evol 60:1479–1485. https://doi.org/10.1007/s10722-012-9936-1
Ding R, Yang M, Quan J, Li S, Zhuang Z, Zhou S, Zheng E, Hong L, Li Z, Cai G, Huang W, Wu Z, Yang J (2019) Single-locus and multi-locus genome-wide association studies for intramuscular fat in Duroc pigs. Front Genet 10:619. https://doi.org/10.3389/fgene.2019.00619
Elliott RC, Betzner AS, Huttner E, Oakes MP, Tucker WQ, Gerentes D, Perez P, Smyth DR (1996) AINTEGUMENTA, an APETALA2-like gene of Arabidopsis with pleiotropic roles in ovule development and floral organ growth. Plant Cell 8:155–168. https://doi.org/10.1105/tpc.8.2.155
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
VSN International. 2015. Genstat for Windows 18th Edition, VSNI Hemel Hempstead England UK, Available online: https//www.vsni.co.uk/software/genstathttp://www.Genstat.co.uk" http://www.Genstat.co.uk (accessed on 10 May 2015).
Grabowski PP, Evans J, Daum C, Deshpande S, Barry KW, Kennedy M, Ramstein G, Kaeppler SM, Buell CR, Jiang Y, Casler MD (2017) Genome-wide associations with flowering time in switchgrass using exome-capture sequencing data. New Phytol 213:154–169. https://doi.org/10.1111/nph.14101
He Y, Michaels SD, Amasino RM (2003) Regulation of flowering time by histone acetylation in Arabidopsis. Science 302:1751–1754. https://doi.org/10.1126/science.1091109
He L, Xiao J, Rashid KY, Yao Z, Li P, Jia G, Wang X, Cloutier S, You FM (2019) Genome-wide association studies for pasmo resistance in flax (Linum usitatissimum L.). Front Plant Sci 9:1982. https://doi.org/10.3389/fpls.2018.01982
Huang X, Wei X, Sang T, Zhao Q, Feng Q, Zhao Y, Li C, Zhu C, Lu T, Zhang Z, Li M, Fan D, Guo Y, Wang A, Wang L, Deng L, Li W, Lu Y, Weng Q, Liu K, Huang T, Zhou T, Jing Y, Li W, Lin Z, Buckler ES, Qian Q, Zhang QF, Li J, Han B (2010) Genome-wide association studies of 14 agronomic traits in rice landraces. Nat Genet 42:961–967. https://doi.org/10.1038/ng.695
Ionescu IA, Møller BL, Sánchez-Pérez R (2017) Chemical control of flowering time. J Exp Bot 68:369–382. https://doi.org/10.1093/jxb/erw427
Jagadish SVK, Craufurd PQ, Wheeler TR (2008) Phenotyping parents of mapping populations of rice for heat tolerance during anthesis. Crop Sci 48:1140–1146. https://doi.org/10.2135/cropsci2007.10.0559
Jagadish SVK, Bahuguna RN, Djanaguiraman M, Gamuyao R, Prasad PVV, Craufurd PQ (2016) Implications of high temperature and elevated CO2 on flowering time in plants. Front Plant Sci 7:913. https://doi.org/10.3389/fpls.2016.00913
Kong L, Lu S, Wang Y, Fang C, Wang F, Nan H, Su T, Li S, Zhang F, Li X, Zhao X, Yuan X, Liu B, Kong F (2018) Quantitative trait locus mapping of flowering time and maturity in soybean using next-generation sequencing-based analysis. Front Plant Sci 11:9–995. https://doi.org/10.3389/fpls.2018.00995
Kryuchkova-Mostacci N, Robinson-Rechavi M (2016) Tissue-specificity of gene expression diverges slowly between orthologs, and rapidly between paralogs. PLoS Comput Biol 12:e1005274. https://doi.org/10.1371/journal.pcbi.1005274
Lan S, Zheng C, Hauck K, McCausland M, Duguid SD, Booker HM, Cloutier S, You FM (2020) Genomic prediction accuracy of seven breeding selection traits improved by QTL identification in flax. Int J Mol Sci 21:1577. https://doi.org/10.3390/ijms21051577
Long Y, Shi J, Qiu D, Li R, Zhang C, Wang J, Hou J, Zhao J, Shi L, Park BS, Choi SR, Lim YP, Meng J (2007) Flowering time quantitative trait loci analysis of oilseed Brassica in multiple environments and genomewide alignment with Arabidopsis. Genetics 177:2433–2444. https://doi.org/10.1534/genetics.107.080705
Martin T, Fraser HB (2018) Comparative expression profiling reveals widespread coordinated evolution of gene expression across eukaryotes. Nat Commun 9:4963. https://doi.org/10.1038/s41467-018-07436-y
Mouradov A, Cremer F, Coupland G (2002) Control of flowering time: Interacting pathways as a basis for diversity. Plant Cell s111–s130. https://doi.org/10.1105/tpc.001362
Myles S, Peiffer J, Brown PJ, Ersoz ES, Zhang Z, Costich DE, Buckler ES (2009) Association mapping: critical considerations shift from genotyping to experimental design. Plant Cell 21:2194–2202. https://doi.org/10.1105/tpc.109.068437
Pagnussat GC, Yu HJ, Ngo QA, Rajani S, Mayalagu S, Johnson CS, Capron A, Xie LF, Ye D, Sundaresan V (2005) Genetic and molecular identification of genes required for female gametophyte development and function in Arabidopsis. Development 132:603–614. https://doi.org/10.1242/dev.01595
Price A, Patterson N, Plenge R, Weinblatt M, Shadick N, Reich D (2006) Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet 38:904–909. https://doi.org/10.1038/ng1847
Pritchard JK, Stephens M, Rosenberg NA, Donnelly P (2000) Association mapping in structured populations. Am J Hum Genet 67:170–181. https://doi.org/10.1086/302959
Quackenbush J (2003) Microarrays–guilt by association. Science 302:240–241. https://doi.org/10.1126/science.1090887
Rabetafika HN, Van Remoortel V, Danthine S, Paquot M, Bleckler C (2011) Flaxseed proteins food: uses and health benefits. Int J Food Sci Technol. 46:221˗228. https://doi.org/10.1111/j.1365-2621.2010.02477.x.
Raman H, Raman R, Qiu Y, Yadav AS, Sureshkumar S, Borg L, Rohan M, Wheeler D, Owen O, Menz I, Balasubramanian S (2019) GWAS hints at pleiotropic roles for FLOWERING LOCUS T in flowering time and yield-related traits in canola. BMC Genomics 20:636. https://doi.org/10.1186/s12864-019-5964-y
Ravichandran S, Ragupathy R, Edwards T, Domaratzki M, Cloutier S (2019) MicroRNA-guided regulation of heat stress response in wheat. BMC Genomics 20:488. https://doi.org/10.1186/s12864-019-5799-6
Ren WL, Wen YJ, Dunwell JM, Zhang YM (2018) pKWmEB: integration of Kruskal-Wallis test with empirical Bayes under polygenic background control for multi-locus genome-wide association study. Heredity 120:208–218. https://doi.org/10.1038/s41437-017-0007-4
Sasaki E, Frommlet F, Nordborg M (2018) GWAS with heterogeneous data: estimating the fraction of phenotypic variation mediated by gene expression data. G3-Genes Genom Genet 8:3059–3068. https://doi.org/10.1101/175430.
Schwarz G (1978) Estimating the dimension of a model. Ann Statist 6:461–464
Sehgal D, Singh R, Rajpal VR (2016) Chapter 2: quantitave trait loci mapping in plants: concepts and approaches. In: Rajpal VR, Rao SR, Raina SN (eds) Molecular breeding for sustainable crop improvement, vol 11. Springer, Cham, pp 31–59. https://doi.org/10.1007/978-3-319-27090-6_2
Sertse D, You FM, Ravichandran S, Cloutier S (2019) The complex genetic architecture of early root and shoot traits in flax revealed by genome-wide association analyses. Front Plant Sci 10:1483. https://doi.org/10.3389/fpls.2019.01483
Soto-Cerda BJ, Cloutier S (2012) Association mapping in plant genomes. In: Caliskan M (ed) Genetic diversity in plants. InTech, Rijeka, pp 29–54.
Soto-Cerda BJ, Duguid S, Booker H, Rowland G, Diederichsen A, Cloutier S (2014) Association mapping of seed quality traits using the Canadian flax (Linum usitatissimum L.) core collection. Theor Appl Genet 127:881–896. https://doi.org/10.1007/s00122-014-2264-4
Soto-Cerda BJ, Cloutier S, Quian R, Gajardo HA, Olivos M, You FM (2018) Genome-wide association analysis of mucilage and hull content in flax (Linum usitatissimum L.) seeds. Int J Mol Sci 19:2870. https://doi.org/10.3390/ijms19102870
Soto-Cerda BJ, Cloutier S, Gajardo HA, Aravena G, Quian R, You FM (2020) Drought response of flax accessions and identification of quantitative trait nucleotides (QTNs) governing agronomic and root traits by genome-wide association analysis. Mol Breed 40:15. https://doi.org/10.1007/s11032-019-1096-y
Srikanth A, Schmid M (2011) Regulation of flowering time: all roads lead to Rome. Cell Mol Life Sci 68:2013–2037. https://doi.org/10.1007/s00018-011-0673-y
Tamba CL, Zhang YM (2018) A fast mrMLM algorithm for multi-locus genome-wide association studies. bioRxiv [Preprint]. https://doi.org/10.1101/341784.
Tamba CL, Ni YL, Zhang YM (2017) Iterative sure independence screening EM-Bayesian LASSO algorithm for multi-locus genome-wide association studies. PLoS Comput Biol 13:e1005357. https://doi.org/10.1371/journal.pcbi.1005357
Tasma IM, Lorenzen LL, Green DE, Shoemaker RC (2001) Mapping genetic loci for flowering time, maturity, and photoperiod insensitivity in soybean. Mol Breed 8:25–35. https://doi.org/10.1023/A:1011998116037
Turck F, Fornara F, Coupland G (2008) Regulation and identity of florigen: FLOWERING LOCUS T moves center stage. Annu Rev Plant Biol 59:573–594. https://doi.org/10.1146/annurev.arplant.59.032607.092755
Verhage L, Angenent GC, Immink RG (2014) Research on floral timing by ambient temperature comes into blossom. Trends Plant Sci 19:583–591. https://doi.org/10.1016/j.tplants.2014.03.009
Vercruysse J, Van Bel M, Osuna-Cruz CM, Kulkarni SR, Storme V, Nelissen H, Gonzales N, Inzé D, Vandepoele K (2020) Comparative transcriptomics enables the identification of functional orthologous genes involved in early leaf growth. Plant Biotecnol J 18:553–567. https://doi.org/10.1111/pbi.13223
Vos PG, Paulo MJ, Voorrips RE, Visser RGF, van Eck HJ, van Eeuwijk FA (2017) Evaluation of LD decay and various LD-decay estimators in simulated and SNP-array data of tetraploid potato. Theor Appl Genet 130:123–135. https://doi.org/10.1007/s00122-016-2798-8
Wang Z, Hobson N, Galindo L, Zhu S, Shi D, McDill J, Yang L, Hawkins S, Neutelings G, Datla R, Lambert G, Galbraith DW, Grassa CJ, Geraldes A, Cronk QC, Cullis C, Dash PK, Kumar PA, Cloutier S, Sharpe AG, Wong GKS, Wang J, Deyholos MK (2012a) The genome of flax (Linum usitatissimum) assembled de novo from short shotgun sequence reads. Plant J 72:461–473. https://doi.org/10.1111/j.1365-313X.2012.05093.x
Wang X, Wu F, Xie Q, Wang H, Wang Y, Yue Y, Gahura O, Ma S, Liu L, Cao Y, Jiao Y, Puta F, McClung CR, Xu X, Ma L (2012b) SKIP is a component of the spliceosome linking alternative splicing and the circadian clock in Arabidopsis. Plant Cell 24:3278–3295. https://doi.org/10.1105/tpc.112.100081
Wang SB, Feng JY, Ren WL, Huang B, Zhou L, Wen YJ, Zhang J, Dunwell JM, Xu S, Zhang YM (2016) Improving power and accuracy of genome-wide association studies via multi-locus mixed linear model methodology. Sci Rep 6:19444. https://doi.org/10.1038/srep19444
Wen YJ, Zhang H, Ni YL, Huang B, Zhang J, Feng JY, Wang SB, Dunwell JM, Zhang YM, Wu R (2018) Methodological implementation of mixed linear models in multi-locus genome-wide association studies. Brief Bioinform 19:700–712. https://doi.org/10.1093/bib/bbw145
Wheeler TR, Craufurd PQ, Ellis RH, Porter JR, Prasad PVV (2000) Temperature variability and the yield of annual crops. Agric Ecosyst Environ 82:159–167. https://doi.org/10.1016/S0167-8809(00)00224-3
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag New York. ISBN 978-3-319-24277-4. https://ggplot2.tidyverse.org.
Xiao D, Shen HR, Zhao JJ, Wei YP, Liu DR, Hou XL, Bonnema G (2019) Genetic dissection of flowering time in Brassica rapa responses to temperature and photoperiod. Plant Sci 280:110–119. https://doi.org/10.1016/j.plantsci.2018.10.027
Xu L, Hu K, Zhang Z, Guan C, Chen S, Hua W, Li J, Wen J, Yi B, Shen J, Ma C, Tu J, Fu T (2016) Genome-wide association study reveals the genetic architecture of flowering time in rapeseed (Brassica napus L.). DNA Res 23:43–52. https://doi.org/10.1093/dnares/dsv035
You FM, Cloutier S (2020) Mapping quantitative trait loci onto chromosome-Scale pseudomolecules in flax. Methods Protoc 3:E28. https://doi.org/10.3390/mps3020028
Yu J, Pressoir G, Briggs WH, Bi IV, Yamasaki M, Doebley JF, McMullen MD, Gaut BS, Nielsen DM, Holland JB, Kresovich S, Buckler ES (2006) A unified mixed-model method for association mapping that accounts for multiple levels of relatedness. Nat Genet 38:203–208. https://doi.org/10.1038/ng1702
Zhang J, Feng JY, Ni YL, Wen YJ, Niu Y, Tamba CL, Yue C, Song Q, Zhang YM (2017) pLARmEB: integration of least angle regression with empirical Bayes for multilocus genome-wide association studies. Heredity 118:517–524. https://doi.org/10.1038/hdy.2017.8
Zhang YM, Jia Z, Dunwell JM (2019) Editorial: The applications of new multi-locus GWAS methodologies in the genetic dissection of complex traits. Front Plant Sci 10:100. https://doi.org/10.3389/fpls.2019.00100
Zhang J, Qi Y, Wang L, Wang L, Yan X, Dang Z, Li W, Zhao W, Pei X, Li X, Liu M, Tan M, Wang L, Long Y, Wang J, Zhang X, Dang Z, Zheng H, Liu T (2020) Genomic comparison and population diversity analysis provide insights into the domestication and improvement of flax. iScience 23:100967. https://doi.org/10.1016/j.isci.2020.100967
Zhuang Z, Ding R, Peng L, Wu J, Ye Y, Zhou S, Wang X, Quan J, Zheng E, Cai G, Huang W, Yang J, Wu Z (2020) Genome-wide association analyses identify known and novel loci for teat number in Duroc pigs using single-locus and multi-locus models. BMC Genomics 21:344. https://doi.org/10.1186/s12864-020-6742-6
Zimmermann P, Hirsch-Hoffmann M, Hennig L, Gruissem W (2004) GENEVESTIGATOR. Arabidopsis microarray database and analysis toolbox. Plant Physiol 136:2621–2632. https://doi.org/10.1104/pp.104.046367
Acknowledgements
This work was funded by Fondo Nacional de Desarrollo Científico y Tecnológico (FONDECYT) project N°1161133, and supported by the Agriaquaculture Nutritional Genomic Center (CGNA) and the Gobierno Regional de La Araucania, Chile. CGNA acknowledges the collaboration of Agriculture and Agri-Food Canada (AAFC) and the Total Utilization Flax GENomics (TUFGEN) project formerly funded by Genome Canada and other stakeholders of the Canadian flax industry. Madeleine Levesque-Lemay is also acknowledged for her helpful assistance in manuscript preparation.
Author information
Authors and Affiliations
Contributions
BJSC and SC conceived and designed the study. GA and BJSC conducted the phenotyping of FL. SC performed curation of next-generation sequencing data. BJSC conducted GWA analyses. BJSC and GA prepared tables and figures. BJSC and SC drafted the manuscript. All authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study did not involve human participants.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Additional information
Communicated by Stefan Hohmann.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Soto-Cerda, B.J., Aravena, G. & Cloutier, S. Genetic dissection of flowering time in flax (Linum usitatissimum L.) through single- and multi-locus genome-wide association studies. Mol Genet Genomics 296, 877–891 (2021). https://doi.org/10.1007/s00438-021-01785-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-021-01785-y