Abstract
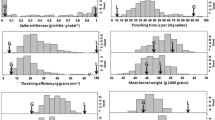
Genetic mutations in genes governing wheat threshability were critical for domestication. Knowing when these genes mutated during wheat evolution will provide more insight into the domestication process and lead to further exploitation of primitive alleles for wheat improvement. We evaluated a population of recombinant inbred lines derived from a cross between the durum variety Rusty and the cultivated emmer accession PI 193883 for threshability, rachis fragility, and other spike-related traits. Quantitative trait loci (QTL) associated with spike length, spikelets per spike, and spike compactness were primarily associated with known genes such as the pleiotropic domestication gene Q. Interestingly, rachis fragility was not associated with the Q locus, suggesting that this trait, usually a pleiotropic effect of the q allele, can be influenced by the genetic background. Threshability QTL were identified on chromosome arms 2AS, 2BS, and 5AL corresponding to the tenacious glume genes Tg2A and Tg2B as well as the Q gene, respectively, further demonstrating that cultivated emmer harbors the primitive non-free-threshing alleles at all three loci. Genetic analysis indicated that the effects of the three genes are mostly additive, with Q having the most profound effects on threshability, and that free-threshing alleles are necessary at all three loci to attain a completely free-threshing phenotype. These findings provide further insight into the timeline and possible pathways of wheat domestication and evolution that led to the formation of modern day domesticated wheats.




Similar content being viewed by others
References
Akpinar BA, Biyiklioglu S, Alptekin B, Havrànkovà M, Vràna J, Doležel J, Distelfeld A, Hernandez P, The IWGSC, Budak H (2018) Chromosome-based survey sequencing reveals the genome organization of wild wheat progenitor Triticum dicoccoides. Plant Biotech J 16:2077–2087
Alvarez MA, Tranquilli G, Lewis S, Kippes N, Dubcovsky J (2016) Genetic and physical mapping of the earliness per se locus Eps-A m 1. in Triticum monococcum identifies EARLY FLOWERING 3 (ELF3) as a candidate gene. Funct Integr Genom 16:365–382
Ankerst M, Breunig MM, Kriegel H-P, Sander J (1999) OPTICS: Ordering points to identify the clustering structure. In: Delis A, Faloutsos C and Ghandeharizadeh S (eds), Proceedings ACM SIGMOD’99 international conference on management of data, ACM Press, Philadelphia, pp 49–60
Avni R, Nave M, Barad O, Baruch K, Twardziok SO, Gundlach H, Hale I, Mascher M, Spannagl M, Wiebe K, Jordan KW, Golan G, Deek J, Ben-Zvi B, Ben-Zvi G, Himmelbach A, MacLachlan RP, Sharpe AG, Fritz A, Ben-David R, Budak H, Fahima T, Korol A, Faris JD, Hernandez A, Mikel MA, Levy AA, Steffenson B, Maccaferri M, Tuberosa R, Cattivelli L, Faccioli P, Ceriotti A, Kashkush K, Pourkheirandish M, Komatsuda T, Eilam T, Sela H, Sharon A, Ohad N, Chamovitz DA, Mayer KFX, Stein N, Ronen G, Peleg Z, Pozniak CJ, Akhunov ED, Distelfeld A (2017) Wild emmer genome architecture and diversity elucidate wheat evolution and domestication. Science 357:93–97
Bullrich L, Appendino ML, Tranquilli G, Lewis S, Dubcovsky J (2002) Mapping of a thermo-sensitive earliness per se gene on Triticum monococcum chromosome 1Am. Theor Appl Genet 105:585–593
Cadalen T, Sourdille P, Charmet G, Tixier MH, Gay G, Boeuf C, Bernard S, Leroy P, Bernard M (1998) Molecular markers linked to genes affecting plant height in wheat using a double-haploid population. Theor Appl Genet 96:933–940
Chalupska D, Lee HY, Faris JD, Evrard A, Chalhoub B, Haselkorn R, Gornicki P (2008) Acc homoeoloci and the evolution of the wheat genomes. Proc Natl Acad Sci USA 105:9691–9696
Debernardi JM, Lin H, Chuck G, Faris JD, Dubcovsky J (2017) microRNA172 plays a crucial role in wheat spike morphogenesis and grain threshability. Development 144:1966–1975
Díaz A, Zikhali M, Turner AS, Isaac P, Laurie D (2012) Copy number variation affecting the Photoperiod-B1 and Vernalization-A1 genes is associated with altered flowering time in wheat (Triticum aestivum). PLoS One 7:e33234. https://doi.org/10.1371/journal.pone.0033234
Doebley JF, Gaut BS, Smith BD (2006) The molecular genetics of crop domestication. Cell 127:1309–1321
Dubcovsky J, Lijavetzky D, Appendino L, Tranquilli G (1998) Comparative RFLP mapping of Triticum monococcum genes controlling vernalization requirement. Theor Appl Genet 97:968–975
Dvorak J, Deal KR, Luo M-C, You FM, von Borstel K, Dehghani H (2012) The origin of spelt and free-threshing hexaploid wheat. J Hered 103:426–441
Dvořák J, Di Terlizzi P, Zhang H-B, Resta P (1993) The evolution of polyploid wheats: identification of the A genome donor species. Genome 36:21–31
Faris JD (2014) Wheat Domestication: Key to agricultural revolution past and future. In: Tuberosa R, Graner A, Frison E (eds) Genomics of plant genetic resources, Vol. 1: Managing, sequencing and mining genetic resources. Springer, Netherlands, pp 439–464
Faris JD, Gill BS (2002) Genomic targeting and high-resolution mapping of the domestication gene Q in wheat. Genome 45:706–718
Faris JD, Fellers JP, Brooks SA, Gill BS (2003) A bacterial artificial chromosome contig spanning the major domestication locus Q in wheat and identification of a candidate gene. Genetics 164:311–321
Faris JD, Simons KJ, Zhang Z, Gill BS (2005) The wheat super domestication gene Q. Wheat Info Serv 100:129–148
Faris JD, Zhang Z, Chao S (2014a) Map-based analysis of the tenacious glume gene Tg-B1 of wild emmer and its role in wheat domestication. Gene 542:198–208
Faris JD, Zhang Z, Garvin DF, Xu SS (2014b) Molecular and comparative mapping of genes governing spike compactness from wild emmer wheat. Mol Genet Genomics 289:641–651
Faris JD, Zhang Q, Chao S, Zhang Z, Xu SS (2014c) Analysis of agronomic and domestication traits in a durum × cultivated emmer wheat population using a high-density single nucleotide polymorphism-based linkage map. Theor Appl Genet 127:2333–2348
Fu D, Szűcs P, Yan L, Helguera M, Skinner JS, von Zitzewitz J, Hayes PM, Dubcovsky J (2005) Large deletions within the first intron in VRN-1 are associated with spring growth habit in barley and wheat. Mol Genet Genomics 273:54–65
Greenwood JR, Finnegan EJ, Watanabe N, Trevaskis B, Swain SM (2017) New alleles of the wheat domestication gene Q reveal multiple roles in growth and reproductive development. Development 144:1959–1965
Hillman GC (1978) On the origins of domestic rye—Secale cereale: the finds from aceramic can hasan III in Turkey. Anatolian Studies 28:157–174
Hu X, Ren J, Ren X, Huang S, Sabiel SAI, Luo M, Nevo E, Fu C, Peng J, Sun D (2015) Association of agronomic traits with SNP markers in durum wheat (Triticum turgidum L. durum (Desf.)). PLoS One 10:e0130854. https://doi.org/10.1371/journal.pone.0130854
Huang S, Sirikhachornkit A, Su X, Faris J, Gill B, Haselkorn R, Gornicki P (2002) Genes encoding acetyl-CoA carboxylase and 3-phosphoglycerate kinase of the Triticum/ Aegilops complex and the evolutionary history of polyploid wheat. Proc Natl Acad Sci USA 99:8133–8138
Joehanes R, Nelson JC (2008) QGene 4.0, an extensible Java QTL-analysis platform. Bioinformatics 24:2788–2789
Kamran A, Iqbal M, Spaner D (2014) Flowering time in wheat (Triticum aestivum L.): a key factor for global adaptability. Euphytica 197:1. https://doi.org/10.1007/s10681-014-1075-7
Kato K, Miura H, Sawada S (1999) QTL mapping of genes controlling ear emergence time and plant height on chromosome 5A of wheat. Theor Appl Genet 98:472–477
Kato K, Miura H, Sawada S (2000) Mapping QTLs controlling grain yield and its components on chromosome 5A of wheat. Theor Appl Genet 101:1114–1121
Kato K, Sonokawa R, Miura H, Sawada S (2003) Dwarfing effect associated with the threshability gene Q on wheat chromosome 5A. Plant Breed 122:489–492
Kerber ER, Dyck PL (1969) Inheritance in hexaploid wheat of leaf rust resistance and other characters derived from Aegilops squarrosa. Can J Genet Cytol 11:639–647
Kerber ER, Rowland GG (1974) Origin of the free threshing character in hexaploid wheat. Can J Genet Cytol 16:145–154
Kislev ME (1980) Triticum parvicoccum, the oldest naked wheat. Isr J Bot 28:95–107
Klindworth DL, Miller JD, Xu SS (2006) Registration of Rusty durum wheat. Crop Sci 46:1012–1013
Kosambi DD (1943) The estimation of map distances from recombination values. Ann Eugen 12:172–175
Leighty CE, Boshnakian S (1921) Genetic behaviour of the spelt form in crosses between Triticum spelta and Triticum aestivum. J Agric Res 7:335–364
Levene H (1960) Robust tests for equality of variances. In: Olkin I, Hotelling H et al (eds) Contributions to probability and statistics: essays in honor of harold hotelling. Stanford University Press, Stanford, CA, pp 278–292
Li W, Gill BS (2006) Multiple genetic pathways for seed shattering in the grasses. Funct Integr Genomics 6:300–309
Lorieux M (2012) MapDisto: fast and efficient computation of genetic linkage maps. Mol Breeding 30:1231–1235
Mackey J (1954) Neutron and X-ray experiments in wheat and a revision of the speltoid problem. Hereditas 40:65–180
Muramatsu M (1979) Presence of vulgare gene, Q, in a dense-spike variety of Triticum dicoccum Schübl. Report of the Plant Germ-Plasm Institute, Kyoto University, No 4, pp 39–41
Muramatsu M (1985) Spike type in two cultivars of Triticum dicoccum with the spelta gene q compared with the Q-bearing variety liguliforme. Jpn J Breed 35:255–267
Muramatsu M (1986) The vulgare super gene, Q: its universality in durum wheat and its phenotypic effects in tetraploid and hexaploid wheats. Can J Genet Cytol 28:30–41
Nalam VJ, Vales MI, Watson CJW, Kianian SF, Riera-Lizarazu O (2006) Map-based analysis of genes affecting the brittle rachis character in tetraploid wheat (Triticum turgidum L.). Theor Appl Genet 112:373–381
Nesbitt M (2001) Wheat evolution: integrating archaeological and biological evidence. In: Caligari PDS, Brandham PE (eds) Wheat taxonomy: the legacy of John Percival. Linnean Society, London, pp 37–59 (Linnean Special Issue 3)
Nesbitt M, Samuel D (1996) From staple crop to extinction? The archaeology and history of hulled wheats. In: Padulosi S, Hammer K, Heller J (eds) Hulled wheats, promoting the conservation and use of underutilized and neglected crops 4: proceedings of the first international workshop on hulled wheats. Castelvecchio Pascoli, Tuscany, Italy, pp 41–100
Nitcher R, Pearce S, Tranquilli G, Zhang X, Dubcovsky J (2014) Effect of the hope FT-B1 allele on wheat heading time and yield components. J Hered 105:666–675
Pallotta MA, Warner P, Fox RL, Kuchel H, Jefferies SJ, Langridge P (2003) Marker assisted wheat breeding in the southern region of Australia. In: Pogna NE, Romano M, Pogna EA, Galterio Z (eds) Proceedings of the 10th international wheat genetics symposium, Paestum, Italy, 1–6 September, 2003. Instituto Sperimentale per la Cerealicoltura, Sant’Angelo Lodigiano, pp 789–791
Peleg Z, Fahima T, Korol AB, Abbo S, Saranga Y (2011) Genetic analysis of wheat domestication and evolution under domestication. J Exp Botany 62:5051–5061
Peng J, Ronin Y, Fahima T, Röder MS, Li Y, Nevo E, Korol A (2003) Domestication quantitative trait loci in Triticum dicoccoides, the progenitor of wheat. Proc Natl Acad Sci USA 100:2489–2494
Peng J, Sun D, Nevo E (2011) Wild emmer wheat, Triticum dicoccoides, occupies a pivotal position in wheat domestication process. Aust J Crop Sci 5:1127–1143
Röder MS, Korzun V, Gill BS, Ganal MW (1998a) The physical mapping of microsatellite markers in wheat. Genome 41:278–283
Röder MS, Korzun V, Wendehake K, Plaschke J, Tixier MH, Leroy P, Ganal MW (1998b) A microsatellite map of wheat. Genetics 149:2007–2023
SAS Institute (2011) SAS/STAT 9.3 User’s Guide. SAS Institute Inc., Cary
Sears ER (1954) The aneuploids of common wheat. Mo Agr Exp Stn Res Bull 572:1–59
Sears ER (1956) The systematics, cytology and genetics of wheat. handb pflanzenzücht, vol. 2, 2nd edn, pp 164–187
Shiferaw B, Smale M, Braun H-J, Duveiller E, Reynolds M, Muricho G (2013) Crops that feed the world 10. Past successes and future challenges to the role played by wheat in global food security. Food Sec 5:291–317
Simonetti MC, Bellomo MP, Laghetti G, Perrino P, Simeone R, Blanco A (1999) Quantitative trait loci influencing free-threshing habit in tetraploid wheats. Genet Res Crop Evol 46:267–271
Simons KJ, Fellers JP, Trick HN, Zhang Z, Tai Y-S, Gill BS, Faris JD (2006) Molecular characterization of the major wheat domestication gene Q. Genetics 172:547–555
Singh HB, Anderson E, Pal BP (1957) Studies in the genetics of Triticum vavilovii Jackub. Agron J 49:4–11
Snedecor GW, Cochran WG (1989) Statistical methods, Eighth Edition. Iowa State University Press, Ames
Somers DJ, Isaac P, Edwards K (2004) A high-density microsatellite consensus map for bread wheat (Triticum aestivum L.). Theor Appl Genet 109:1105–1114
Song QJ, Shi JR, Singh S, Fickus EW, Costa JM, Lewis J, Gill BS, Ward R, Cregan PB (2005) Development and mapping of microsatellite (SSR) markers in wheat. Theor Appl Genet 110:550–560
Sorrells ME, Gustafson JP, Somers D, Chao S, Benscher D, Guedira-Brown G, Huttner E, Kilian A, McGuire PE, Ross K, Tanaka J, Wenzl P, Williams K, Qualset CO (2011) Reconstruction of the synthetic W7984 × Opata M85 wheat reference population. Genome 54:875–882
Sourdille P, Cadalen T, Guyomarc’h H, Snape JW, Perretant MR, Charmet G, Boeuf C, Bernard S, Bernard M (2003) An update of the Courtot × Chinese spring intervarietal molecular marker linkage map for the QTL detection of agronomic traits in wheat. Theor Appl Genet 106:530–538
Thanh PT, Vladutu CI, Kianian SF, Thanh PT, Ishii T, Nitta M, Nasuda S, Mori N (2013) Molecular genetic analysis of domestication traits in emmer wheat. I: map construction and QTL analysis using an F2 population. Biotechnology 27:3627–3637
Torada A, Koike M, Mochida K, Ogihara Y (2006) SSR-based linkage map with new markers using an intraspecific population of common wheat. Theor Appl Genet 112:1042–1051
Tzarfati R, Saranga Y, Barak V, Gopher A, Korol AB, Abbo S (2013) Threshing efficiency as an incentive for rapid domestication of emmer wheat. Ann Bot 112:829–837
Wang S, Wong D, Forrest K, Allen A, Chao S, Huang BE, Maccaferri M, Salvi S, Milner SG, Cattivelli L, Mastrangelo AM, Whan A, Stephen S, Barker G, Wieseke R, Plieske J, International Wheat Genome Sequencing Consortium, Lillemo M, Mather D, Appels R, Dolferus R, Brown-Guedira G, Korol A, Akhunova AR, Feuillet C, Salse J, Morgante M, Pozniak C, Luo M-C, Dvorak J, Morell M, Dubcovsky J, Ganal M, Tuberosa R, Lawley C, Mikoulitch I, Cavanagh C, Edwards KJ, Hayden M, Akhunov E (2014) Characterization of polyploid wheat genomic diversity using a high-density 90 000 single nucleotide polymorphism array. Plant Biotechnol J 12:787–796
Yan L, Loukoianov A, Tranquilli G, Helguera M, Fahima T, Dubcovsky J (2003) Positional cloning of wheat vernalization gene VRN1. Proc Natl Acad Sci 100:6263–6268
Yan L, Helguera M, Kato K, Fukuyama S, Sherman J, Dubcovsky J (2004) Allelic variation at the VRN-1 promoter region in polyploid wheat. Theor Appl Genet 109:1677–1686
Zhang ZC, Belcram H, Gornicki P, Charles M, Just J, Huneau C, Magdelenat G, Couloux A, Samain S, Gill BS, Rasmussen JB, Barbe V, Faris JD, Chalhoub B (2011) Duplication and partitioning in evolution and function of homoeologous Q loci governing domestication characters in polyploid wheat. Proc Natl Acad Sci USA 108:18737–18742
Zou J, Semagn K, Iqbal M, Chen H, Asif M, N’Diaye A, Navabi A, Perez-Lara E, Pozniak C, Yang RC, Randhawa H, Spaner D (2017) QTLs associated with agronomic traits in the Attila × CDC Go spring wheat population evaluated under conventional management. PLoS ONE 12:e0171528. https://doi.org/10.1371/journal.pone.0171528
Acknowledgements
The authors thank Megan E. Overlander and Samantha Steckler for greenhouse and technical assistance. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Funding
This research was supported in part by funds to S.S.X. provided through a grant from the Bill & Melinda Gates Foundation to Cornell University for the Borlaug Global Rust Initiative (BGRI) Durable Rust Resistance in Wheat (DRRW) Project and the U.S. Department of Agriculture–Agriculture Research Service (USDA–ARS) Current Research Information System (CRIS) Project No. 3060-21000-038-00D.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Jyoti Sharma declares that she has no conflict of interest, Katherine Running declares that she has no conflict of interest, Steven Xu declares that he has no conflict of interest, Qijun Zhang declares that he has no conflict of interest, Amanda Peters Haugrud declares that she has no conflict of interest, Sapna Sharma declares that she has no conflict of interest, Phillip McClean declares that he has no conflict of interest, and Justin Faris declares that he has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by Shawn Kaeppler.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, J.S., Running, K.L.D., Xu, S.S. et al. Genetic analysis of threshability and other spike traits in the evolution of cultivated emmer to fully domesticated durum wheat. Mol Genet Genomics 294, 757–771 (2019). https://doi.org/10.1007/s00438-019-01544-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-019-01544-0