Abstract
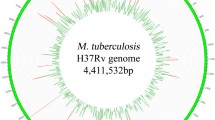
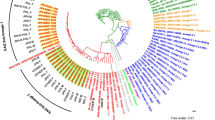
The Beijing genotype of Mycobacterium tuberculosis (MTB) is one of the most successful MTB lineages that has disseminated in the world. In China, the rate of multidrug-resistant (MDR) tuberculosis is significantly higher than the global average rate, and the Beijing genotype strains take the largest share of MDR strains. To study the genetic basis of the epidemiological findings that Beijing genotype has often been associated with tuberculosis outbreaks and drug resistance, we determined the genome sequences of four clinical isolates: two extensively drug resistant (XDR1219, XDR1221) and two multidrug resistant (WX1, WX3), using whole-genome sequencing. A large number of individual and shared SNPs of the four Beijing strains were identified. Our isolates harbored almost all classic drug resistance-associated mutations. The mutations responsible for drug resistance in the two XDR strains were consistent with the clinical quantitative drug resistance levels. COG analysis revealed that Beijing strains have significantly higher abundances of the mutations responsible for cell wall/membrane/envelope biogenesis (COG M), secondary metabolites biosynthesis, transport and catabolism (COG Q), lipid transport and metabolism (COG I) and defense mechanisms (COG V). The shared mutated genes of the four studied Beijing strains were significantly overrepresented in three DNA repair pathways. Our analyses promote the understanding of the genome polymorphism of the Beijing family strains and provide the molecular genetic basis for their wide dissemination capacity and drug resistance.



Similar content being viewed by others
Abbreviations
- MTB:
-
Mycobacterium tuberculosis
- MDR:
-
Multidrug resistant
- COG:
-
Clusters of Orthologous Groups of proteins
- TB:
-
Tuberculosis
- RIF:
-
Rifampicin
- INH:
-
Isoniazid
- MDR-TB:
-
Multidrug-resistant tuberculosis
- XDR-TB:
-
Extensively drug-resistant tuberculosis
- FQs:
-
Fluoroquinolones
- SNPs:
-
Single nucleotide polymorphisms
- MIC:
-
Minimum inhibitory concentration
- ORFs:
-
Open reading frames
- TBDB:
-
TB Drug Resistance Mutation Database
- STR:
-
Streptomycin
- EMB:
-
Ethambutol
- OFX:
-
Ofloxacin
- QRDR:
-
Quinolone resistance-determining region
- KAN:
-
Kanamycin
- AMI:
-
Amikacin
- CAP:
-
Capreomycin
- MMR:
-
Mismatch repair
- BER:
-
Base excision repair
- NER:
-
Nucleotide excision repair
- NHEJ:
-
Non-homologous end joining
- WGS:
-
Whole-genome sequence
- DIM:
-
Phthiocerol dimycocerosates
- PGL:
-
Phenolglycolipids
- ROS:
-
Reactive oxygen species
- RNI:
-
Reactive nitrogen intermediates
References
Almeida Da Silva PE, Palomino JC (2011) Molecular basis and mechanisms of drug resistance in Mycobacterium tuberculosis: classical and new drugs. J Antimicrob Chemother 66(7):1417–1430. doi:10.1093/jac/dkr173
Almeida D, Rodrigues C, Ashavaid TF, Lalvani A, Udwadia ZF, Mehta A (2005) High incidence of the Beijing genotype among multidrug-resistant isolates of Mycobacterium tuberculosis in a tertiary care center in Mumbai, India. Clin Infect Dis 40(6):881–886. doi:10.1086/427940
Andersson DI, Levin BR (1999) The biological cost of antibiotic resistance. Curr Opin Microbiol 2(5):489–493
Aubry A, Pan XS, Fisher LM, Jarlier V, Cambau E (2004) Mycobacterium tuberculosis DNA gyrase: interaction with quinolones and correlation with antimycobacterial drug activity. Antimicrob Agents Chemother 48(4):1281–1288
Bifani PJ, Mathema B, Kurepina NE, Kreiswirth BN (2002) Global dissemination of the Mycobacterium tuberculosis W-Beijing family strains. Trends Microbiol 10(1):45–52
Bodmer T, Zurcher G, Imboden P, Telenti A (1995) Mutation position and type of substitution in the beta-subunit of the RNA polymerase influence in vitro activity of rifamycins in rifampicin-resistant Mycobacterium tuberculosis. J Antimicrob Chemother 35(2):345–348
Castresana J (2000) Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol Biol Evol 17(4):540–552
Cole ST, Brosch R, Parkhill J, Garnier T, Churcher C, Harris D, Gordon SV, Eiglmeier K, Gas S, Barry CE 3rd, Tekaia F, Badcock K, Basham D, Brown D, Chillingworth T, Connor R, Davies R, Devlin K, Feltwell T, Gentles S, Hamlin N, Holroyd S, Hornsby T, Jagels K, Krogh A, McLean J, Moule S, Murphy L, Oliver K, Osborne J, Quail MA, Rajandream MA, Rogers J, Rutter S, Seeger K, Skelton J, Squares R, Squares S, Sulston JE, Taylor K, Whitehead S, Barrell BG (1998) Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 393(6685):537–544. doi:10.1038/31159
Comas I, Borrell S, Roetzer A, Rose G, Malla B, Kato-Maeda M, Galagan J, Niemann S, Gagneux S (2012) Whole-genome sequencing of rifampicin-resistant Mycobacterium tuberculosis strains identifies compensatory mutations in RNA polymerase genes. Nat Genet 44(1):106–110. doi:10.1038/ng.1038
Darling AE, Mau B, Perna NT (2010) progressiveMauve: multiple genome alignment with gene gain, loss and rearrangement. PLoS ONE 5(6):e11147. doi:10.1371/journal.pone.0011147
Delcher AL, Harmon D, Kasif S, White O, Salzberg SL (1999) Improved microbial gene identification with GLIMMER. Nucleic Acids Res 27(23):4636–4641
Domenech P, Reed MB, Dowd CS, Manca C, Kaplan G, Barry CE 3rd (2004) The role of MmpL8 in sulfatide biogenesis and virulence of Mycobacterium tuberculosis. J Biol Chem 279(20):21257–21265. doi:10.1074/jbc.M400324200
Drobniewski F, Balabanova Y, Nikolayevsky V, Ruddy M, Kuznetzov S, Zakharova S, Melentyev A, Fedorin I (2005) Drug-resistant tuberculosis, clinical virulence, and the dominance of the Beijing strain family in Russia. JAMA 293(22):2726–2731. doi:10.1001/jama.293.22.2726
Ebrahimi-Rad M, Bifani P, Martin C, Kremer K, Samper S, Rauzier J, Kreiswirth B, Blazquez J, Jouan M, van Soolingen D, Gicquel B (2003) Mutations in putative mutator genes of Mycobacterium tuberculosis strains of the W-Beijing family. Emerg Infect Dis 9(7):838–845. doi:10.3201/eid0907.020589
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32(5):1792–1797. doi:10.1093/nar/gkh340
Finken M, Kirschner P, Meier A, Wrede A, Bottger EC (1993) Molecular basis of streptomycin resistance in Mycobacterium tuberculosis: alterations of the ribosomal protein S12 gene and point mutations within a functional 16S ribosomal RNA pseudoknot. Mol Microbiol 9(6):1239–1246
Fleischmann RD, Alland D, Eisen JA, Carpenter L, White O, Peterson J, DeBoy R, Dodson R, Gwinn M, Haft D, Hickey E, Kolonay JF, Nelson WC, Umayam LA, Ermolaeva M, Salzberg SL, Delcher A, Utterback T, Weidman J, Khouri H, Gill J, Mikula A, Bishai W, Jacobs WR Jr, Venter JC, Fraser CM (2002) Whole-genome comparison of Mycobacterium tuberculosis clinical and laboratory strains. J Bacteriol 184(19):5479–5490
Gagneux S (2013) Genetic Diversity in Mycobacterium tuberculosis. Curr Top Microbiol Immunol. doi:10.1007/82_2013_329
Glynn JR, Whiteley J, Bifani PJ, Kremer K, van Soolingen D (2002) Worldwide occurrence of Beijing/W strains of Mycobacterium tuberculosis: a systematic review. Emerg Infect Dis 8(8):843–849. doi:10.3201/eid0805.020002
Gordhan BG, Andersen SJ, De Meyer AR, Mizrahi V (1996) Construction by homologous recombination and phenotypic characterization of a DNA polymerase domain polA mutant of Mycobacterium smegmatis. Gene 178(1–2):125–130
Hazbon MH, Brimacombe M, Bobadilla del Valle M, Cavatore M, Guerrero MI, Varma-Basil M, Billman-Jacobe H, Lavender C, Fyfe J, Garcia–Garcia L, Leon CI, Bose M, Chaves F, Murray M, Eisenach KD, Sifuentes-Osornio J, Cave MD, Ponce de Leon A, Alland D (2006) Population genetics study of isoniazid resistance mutations and evolution of multidrug-resistant Mycobacterium tuberculosis. Antimicrob Agents Chemother 50(8):2640–2649. doi:10.1128/AAC.00112-06
Hazra TK, Izumi T, Venkataraman R, Kow YW, Dizdaroglu M, Mitra S (2000) Characterization of a novel 8-oxoguanine-DNA glycosylase activity in Escherichia coli and identification of the enzyme as endonuclease VIII. J Biol Chem 275(36):27762–27767. doi:10.1074/jbc.M004052200
Hershberg R, Lipatov M, Small PM, Sheffer H, Niemann S, Homolka S, Roach JC, Kremer K, Petrov DA, Feldman MW, Gagneux S (2008) High functional diversity in Mycobacterium tuberculosis driven by genetic drift and human demography. PLoS Biol 6(12):e311. doi:10.1371/journal.pbio.0060311
Huet G, Constant P, Malaga W, Laneelle MA, Kremer K, van Soolingen D, Daffe M, Guilhot C (2009) A lipid profile typifies the Beijing strains of Mycobacterium tuberculosis: identification of a mutation responsible for a modification of the structures of phthiocerol dimycocerosates and phenolic glycolipids. J Biol Chem 284(40):27101–27113. doi:10.1074/jbc.M109.041939
Ilina EN, Shitikov EA, Ikryannikova LN, Alekseev DG, Kamashev DE, Malakhova MV, Parfenova TV, Afanas’ev MV, Ischenko DS, Bazaleev NA, Smirnova TG, Larionova EE, Chernousova LN, Beletsky AV, Mardanov AV, Ravin NV, Skryabin KG, Govorun VM (2013) Comparative genomic analysis of Mycobacterium tuberculosis drug resistant strains from Russia. PLoS ONE 8(2):e56577. doi:10.1371/journal.pone.0056577
Ioerger TR, Koo S, No EG, Chen X, Larsen MH, Jacobs WR Jr, Pillay M, Sturm AW, Sacchettini JC (2009) Genome analysis of multi- and extensively-drug-resistant tuberculosis from KwaZulu-Natal, South Africa. PloS One 4(11):e7778. doi:10.1371/journal.pone.0007778
Kamerbeek J, Schouls L, Kolk A, van Agterveld M, van Soolingen D, Kuijper S, Bunschoten A, Molhuizen H, Shaw R, Goyal M, van Embden J (1997) Simultaneous detection and strain differentiation of Mycobacterium tuberculosis for diagnosis and epidemiology. J Clin Microbiol 35(4):907–914
Kubica T, Agzamova R, Wright A, Aziz MA, Rakishev G, Bismilda V, Richter E, Rusch-Gerdes S, Niemann S (2005) The Beijing genotype is a major cause of drug-resistant tuberculosis in Kazakhstan. Int J Tubercul Lung Dis 9(6):646–653
Margulies M, Egholm M, Altman WE, Attiya S, Bader JS, Bemben LA, Berka J, Braverman MS, Chen YJ, Chen Z, Dewell SB, Du L, Fierro JM, Gomes XV, Godwin BC, He W, Helgesen S, Ho CH, Irzyk GP, Jando SC, Alenquer ML, Jarvie TP, Jirage KB, Kim JB, Knight JR, Lanza JR, Leamon JH, Lefkowitz SM, Lei M, Li J, Lohman KL, Lu H, Makhijani VB, McDade KE, McKenna MP, Myers EW, Nickerson E, Nobile JR, Plant R, Puc BP, Ronan MT, Roth GT, Sarkis GJ, Simons JF, Simpson JW, Srinivasan M, Tartaro KR, Tomasz A, Vogt KA, Volkmer GA, Wang SH, Wang Y, Weiner MP, Yu P, Begley RF, Rothberg JM (2005) Genome sequencing in microfabricated high-density picolitre reactors. Nature 437(7057):376–380. doi:10.1038/nature03959
Mariam DH, Mengistu Y, Hoffner SE, Andersson DI (2004) Effect of rpoB mutations conferring rifampin resistance on fitness of Mycobacterium tuberculosis. Antimicrob Agents Chemother 48(4):1289–1294
Maus CE, Plikaytis BB, Shinnick TM (2005) Molecular analysis of cross-resistance to capreomycin, kanamycin, amikacin, and viomycin in Mycobacterium tuberculosis. Antimicrob Agents Chemother 49(8):3192–3197. doi:10.1128/AAC.49.8.3192-3197.2005
Miyoshi-Akiyama T, Matsumura K, Kobayashi N, Maeda S, Kirikae T (2011) Genome sequence of clinical isolate Mycobacterium tuberculosis NCGM2209. J Bacteriol 193(23):6792. doi:10.1128/JB.06233-11
Mizrahi V, Andersen SJ (1998) DNA repair in Mycobacterium tuberculosis. What have we learnt from the genome sequence? Mol Microbiol 29(6):1331–1339
Musser JM (1995) Antimicrobial agent resistance in mycobacteria: molecular genetic insights. Clin Microbiol Rev 8(4):496–514
Nair J, Rouse DA, Bai GH, Morris SL (1993) The rpsL gene and streptomycin resistance in single and multiple drug-resistant strains of Mycobacterium tuberculosis. Mol Microbiol 10(3):521–527
Okamoto S, Tamaru A, Nakajima C, Nishimura K, Tanaka Y, Tokuyama S, Suzuki Y, Ochi K (2007) Loss of a conserved 7-methylguanosine modification in 16S rRNA confers low-level streptomycin resistance in bacteria. Mol Microbiol 63(4):1096–1106. doi:10.1111/j.1365-2958.2006.05585.x
Ramaswamy S, Musser JM (1998) Molecular genetic basis of antimicrobial agent resistance in Mycobacterium tuberculosis: 1998 update. Tubercle Lung Dis 79(1):3–29. doi:10.1054/tuld.1998.0002
Ramaswamy SV, Amin AG, Goksel S, Stager CE, Dou SJ, El Sahly H, Moghazeh SL, Kreiswirth BN, Musser JM (2000) Molecular genetic analysis of nucleotide polymorphisms associated with ethambutol resistance in human isolates of Mycobacterium tuberculosis. Antimicrob Agents Chemother 44(2):326–336
Ramaswamy SV, Reich R, Dou SJ, Jasperse L, Pan X, Wanger A, Quitugua T, Graviss EA (2003) Single nucleotide polymorphisms in genes associated with isoniazid resistance in Mycobacterium tuberculosis. Antimicrob Agents Chemother 47(4):1241–1250
Rodrigues C, Jani J, Shenai S, Thakkar P, Siddiqi S, Mehta A (2008) Drug susceptibility testing of Mycobacterium tuberculosis against second-line drugs using the Bactec MGIT 960 System. Int J Tubercul Lung Dis 12(12):1449–1455
Rusch-Gerdes S, Pfyffer GE, Casal M, Chadwick M, Siddiqi S (2006) Multicenter laboratory validation of the BACTEC MGIT 960 technique for testing susceptibilities of Mycobacterium tuberculosis to classical second-line drugs and newer antimicrobials. J Clin Microbiol 44(3):688–692. doi:10.1128/JCM.44.3.688-692.2006
Sassetti CM, Boyd DH, Rubin EJ (2003) Genes required for mycobacterial growth defined by high density mutagenesis. Mol Microbiol 48(1):77–84
Shi R, Itagaki N, Sugawara I (2007) Overview of anti-tuberculosis (TB) drugs and their resistance mechanisms. Mini Rev Med Chem 7(11):1177–1185
Somoskovi A, Parsons LM, Salfinger M (2001) The molecular basis of resistance to isoniazid, rifampin, and pyrazinamide in Mycobacterium tuberculosis. Respir Res 2(3):164–168
Takiff HE, Salazar L, Guerrero C, Philipp W, Huang WM, Kreiswirth B, Cole ST, Jacobs WR Jr, Telenti A (1994) Cloning and nucleotide sequence of Mycobacterium tuberculosis gyrA and gyrB genes and detection of quinolone resistance mutations. Antimicrob Agents Chemother 38(4):773–780
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 24(8):1596–1599. doi:10.1093/molbev/msm092
Telenti A, Philipp WJ, Sreevatsan S, Bernasconi C, Stockbauer KE, Wieles B, Musser JM, Jacobs WR Jr (1997) The EMB operon, a gene cluster of Mycobacterium tuberculosis involved in resistance to ethambutol. Nat Med 3(5):567–570
Tracevska T, Jansone I, Baumanis V, Marga O, Lillebaek T (2003) Prevalence of Beijing genotype in Latvian multidrug-resistant Mycobacterium tuberculosis isolates. Int J Tubercul Lung Dis 7(11):1097–1103
Wade MM, Zhang Y (2004) Mechanisms of drug resistance in Mycobacterium tuberculosis. Front Biosci 9:975–994
Wang J, Liu Y, Zhang CL, Ji BY, Zhang LZ, Shao YZ, Jiang SL, Suzuki Y, Nakajima C, Fan CL, Ma YP, Tian GW, Hattori T, Ling H (2011) Genotypes and characteristics of clustering and drug susceptibility of Mycobacterium tuberculosis isolates collected in Heilongjiang Province, China. J Clin Microbiol 49(4):1354–1362. doi:10.1128/JCM.02274-10
Williams DL, Spring L, Collins L, Miller LP, Heifets LB, Gangadharam PR, Gillis TP (1998) Contribution of rpoB mutations to development of rifamycin cross-resistance in Mycobacterium tuberculosis. Antimicrob Agents Chemother 42(7):1853–1857
Wilson M, DeRisi J, Kristensen HH, Imboden P, Rane S, Brown PO, Schoolnik GK (1999) Exploring drug-induced alterations in gene expression in Mycobacterium tuberculosis by microarray hybridization. Proc Natl Acad Sci USA 96(22):12833–12838
Yang B, Koga H, Ohno H, Ogawa K, Fukuda M, Hirakata Y, Maesaki S, Tomono K, Tashiro T, Kohno S (1998) Relationship between antimycobacterial activities of rifampicin, rifabutin and KRM-1648 and rpoB mutations of Mycobacterium tuberculosis. J Antimicrob Chemother 42(5):621–628
Yang C, Luo T, Sun G, Qiao K, Sun G, DeRiemer K, Mei J, Gao Q (2012) Mycobacterium tuberculosis Beijing strains favor transmission but not drug resistance in China. Clin Infect Dis 55(9):1179–1187. doi:10.1093/cid/cis670
Yuen LK, Leslie D, Coloe PJ (1999) Bacteriological and molecular analysis of rifampin-resistant Mycobacterium tuberculosis strains isolated in Australia. J Clin Microbiol 37(12):3844–3850
Zhang Y, Chen C, Liu J, Deng H, Pan A, Zhang L, Zhao X, Huang M, Lu B, Dong H, Du P, Chen W, Wan K (2011) Complete genome sequences of Mycobacterium tuberculosis strains CCDC5079 and CCDC5080, which belong to the Beijing family. J Bacteriol 193(19):5591–5592. doi:10.1128/JB.05452-11
Zheng H, Lu L, Wang B, Pu S, Zhang X, Zhu G, Shi W, Zhang L, Wang H, Wang S, Zhao G, Zhang Y (2008) Genetic basis of virulence attenuation revealed by comparative genomic analysis of Mycobacterium tuberculosis strain H37Ra versus H37Rv. PLoS ONE 3(6):e2375. doi:10.1371/journal.pone.0002375
Acknowledgments
We thank Xuping Fu for many helpful discussions and for his help with the data analysis. This work received support from the National Key Technologies R & D Program of China during the twelfth Five-Year Plan period (No. 2013ZX10003003-004-002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Hohmann.
Wei Wu and Huajun Zheng contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, W., Zheng, H., Zhang, L. et al. A genome-wide analysis of multidrug-resistant and extensively drug-resistant strains of Mycobacterium tuberculosis Beijing genotype. Mol Genet Genomics 288, 425–436 (2013). https://doi.org/10.1007/s00438-013-0758-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-013-0758-4