Abstract
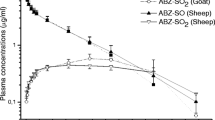
Most pharmacokinetic studies on anthelmintic drugs have been performed on non-parasitized animals. However, it seems likely that the parasite burden could influence the deposition of such drugs. The pharmacokinetics of moxidectin administered orally and by subcutaneous injection was compared in lambs exposed to nematode infection and in parasite naive lambs. Plasma samples were analyzed for moxidectin over 40 days post-treatment. The main pharmacokinetic parameters calculated demonstrated a significant change in drug deposition in infected lambs when compared to controls. The area under the plasma concentration—time curve was decreased 54% and 46% by infection in the subcutaneous and oral groups, respectively. There was also a major decrease in the mean residence time in parasitized lambs. In parallel, the clearance of the drug was increased by infection. Thus, parasite infection dramatically influences the disposition of moxidectin in lambs. These results may contribute to determining a therapeutic strategy adapted to heavily infested animals.


Similar content being viewed by others
References
Alvinerie M, Sutra J F, Badri M, Galtier P (1995) Determination of moxidectin in plasma by high-performance liquid chromatography with automated solid-phase extraction and fluorescence detection. J Chromatogr B Biomed Appl 674:119–124
Alvinerie M, Cabezas I, Rubilar L, Perez R (2000) enhanced plasma availability of moxidectin in fasted horses. J Equine Vet Sci 20: 575–578
Asato G, France D (1990) The American Cynamid Co. U S patent 4916, pp 154–158
Benchaoui HA, McKellar QA (1993) Effect of early treatment with rafoxanide on antipyrine clearance in sheep infected with Fasciola hepatica. Xenobiotica 23:439–448
Biro-Sauveur B, Eeckhoutte C, Baeza E, Boulard C, Galtier P (1995) Comparison of hepatic and extrahepatic drug-metabolizing enzyme activities in rats given single or multiple challenge infections with Fasciola hepatica. Int J Parasitol 25:1193–1200
Craven J, Bjorn H, Hennessy DR, Friis C (2002) The effects of body composition on the pharmacokinetics of subcutaneously injected ivermectin and moxidectin in pigs. J Vet Pharmacol Ther 25:227–232
Dupuy J, Escudero E, Eeckhoutte C, Sutra JF, Galtier P, Alvinerie M (2001) In vitro metabolism of 14C-moxidectin by hepatic microsomes from various species. Vet Res Commun 25:345–354
Echeverria J, Mestorino N, Errecalde JO (2002) Comparative pharmacokinetics of ivermectin after its subcutaneous administration in healthy sheep and sheep infected with mange. J Vet Pharmacol Ther 25:159–160
Galtier P, Alvinerie M, Plusquellec Y, Tufenkji AE, Houin G (1991) Decrease in albendazole sulphonation during experimental fascioliasis in sheep. Xenobiotica 21:917–924
Hennessy DR, Sangster NC, Steel JW, Collins GH (1993) Comparative kinetic disposition of oxfendazole in sheep and goats before and during infection with Haemonchus contortus and Trichostrongylus colubriformis. J Vet Pharmacol Ther 16:245–253
Hennessy DR, Page SW, Gottschall D (2000) The behaviour of doramectin in the gastrointestinal tract, its secretion in bile and pharmacokinetic disposition in the peripheral circulation after oral and intravenous administration to sheep. J Vet Pharmacol Ther 23:203–213
Kawalek JC, Fetterer RH (1990) Effect of Haemonchus contortus infection on the clearance of antipyrine, sulfobromophthalein, chloramphenicol, and sulfathiazole in lambs. Am J Vet Res 51:2044–2049
Kerboeuf D, Hubert J, Cardinaud B, Blond-Riou F (1995) The persistence of the efficacy of injectable or oral moxidectin against Teladorsagia, Haemonchus and Trichostrongylus species in experimentally infected sheep. Vet Rec 137:399–401
Laffont CM, Toutain PL, Alvinerie M, Bousquet-Melou A (2002) Intestinal secretion is a major route for parent ivermectin elimination in the rat. Drug Metab Dispos 30:626–630
Landuyt J, Debackere M, Vercruysse J, McKellar Q (1995) The influence of a heavy infection with sensitive and resistant strains of Ostertagia circumcincta and with Trichostrongylus colubriformis on the pharmacokinetics of febantel in lambs. J Vet Pharmacol Ther 18:180–186
Lanusse C, Lifschitz A, Virkel G, Alvarez L, Sanchez S, Sutra J F, Galtier P, Alvinerie M (1997) Comparative plasma disposition kinetics of ivermectin, moxidectin and doramectin in cattle. J Vet Pharmacol Ther 20:91–99
Lespine A, Bree F, Bassissi F, Sutra JF, Tillement JP, Alvinerie M (2003) Binding of ivermectin and moxidectin to the plasma lipoproteins. 9th International EAVPT Congress, 14–18 July, Lisbon
Lifschitz A, Virkel G, Imperiale F, Sutra JF, Galtier P, Lanusse C, Alvinerie M (1999) Moxidectin in cattle: correlation between plasma and target tissues disposition. J Vet Pharmacol Ther 22:266–273
Lifschitz A, Imperiale F, Virkel G, Munoz Cobenas M, Scherling N, DeLay R, Lanusse C (2000) Depletion of moxidectin tissue residues in sheep. J Agric Food Chem 48:6011-6015
Lifschitz A, Virkel G, Sallovitz J, Imperiale F, Pis A, Lanusse C (2002) Loperamide-induced enhancement of moxidectin availability in cattle. J Vet Pharmacol Ther 25:111–120
Marriner SE, Evans ES, Bogan JA (1985) Effect of parasitism with Ostertagia circumcincta on pharmacokinetics of fenbendazole in sheep. Vet Parasitol 17:239–249
McKellar QA, Jackson F, Coop RL, Jackson E, Scott E (1991) Effect of parasitism with Nematodirus battus on the pharmacokinetics of levamisole, ivermectin and netobimin. Vet Parasitol 39:123–136
Steel J, Symons LEA, Jones WO (1980) Effect of level of larval intake on the productivity and physiological and metabolic responses of lambs infected with Trichostrongylus colubriformis. Aust J Agric 31:821–838
Van Wyk JA, Malan FS, Randles JL (1997) How long before resistance makes it impossible to control some field strains of Haemonchus contortus in South Africa with any of the modern anthelmintics? Vet Parasitol 70:111–122
Wood IB, Bairden K, Duncan JL, Kassai T, Malone JB, Pankavich JA, Reinecke RK, Slocombe O, Taylor SM, Vercruysse J (1995) World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P) second edition of guidelines for evaluating the efficacy of anthelmintics in ruminants (bovine, ovine, caprine). Vet Parasitol 58:181–213
Yamakoa TK, Nokaguana K, Unot T (1981) A pharmacokinetic analysis program (multi) for microcomputer. J Pharmacobiol Dyn 4:879–885
Zulalian J, Stout S, DaCunha A, Garces T, Miller P (1994) Absorption, tissue distribution, metabolism, and excretion of moxidectin in cattle. J Agric Food Chem 42:381–387
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lespine, A., Sutra, JF., Dupuy, J. et al. The influence of parasitism on the pharmacokinetics of moxidectin in lambs. Parasitol Res 93, 121–126 (2004). https://doi.org/10.1007/s00436-004-1084-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-004-1084-x