Abstract
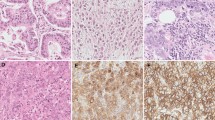
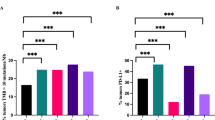
Cancer immunotherapy has significantly improved the management of many malignancies in recent years. Although cervical cancer is the second most common women’s cancer in the world, there are still few information about the role of checkpoint inhibitors in this neoplasm, especially in the neoadjuvant setting. In the present study, we retrieved 38 consecutive patients with squamous cell cervical cancer who underwent platinum-based neoadjuvant chemotherapy (NACT) followed by radical surgery. Pre-therapy biopsies were evaluated for the presence of tumor-infiltrating lymphocytes (TILs), including T (both cytotoxic CD8+ and helper CD4+) and B lymphocytes, macrophages, natural-killer cells, and eosinophils. Immunohistochemistry was performed to characterize the inflammatory cells and to evaluate programmed death-ligand 1 (PD-L1) expression on both neoplastic and inflammatory cells. We divided our study population in three groups using three cut-offs (< 10%, 10–40%, >40%), for both TILs and PD-L1 evaluation. Pathological response to NACT was obtained from the histological reports of the post-therapy surgical specimens. We observed that all cases showed stromal TILs, with a predominance of CD3+/CD4+ T helper cells, thus supporting the strong immunogenic potential of cervical cancer. The vast majority of neoplasms expressed PD-L1: 100% on immune cells and 92% on tumor cells. Firstly, we noticed that the percentage of neoplastic cells PD-L1+ was positively associated with high TIL percentage (p = 0.0073) and with increased PD-L1 expression on inflammatory cells (p = 0.0297). Secondly, we observed a significant correlation between both the percentage (p = 0.0105) of TILs and the expression of PD-L1 (p = 0.01045) on inflammatory cells and pathological response to NACT. These results suggest that cervical cancer could be a good target for immunotherapy, also in the neoadjuvant setting. Furthermore, PD-L1 expression was significantly associated with stromal TILs that interestingly may predict pathological response to NACT.



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Meng Y, Liang H, Hu J, Liu S, Hao X, Wong MSK, Li X, Hu L (2018) PD-L1 expression correlates with tumor infiltrating lymphocytes and response to neoadjuvant chemotherapy in cervical cancer. J Cancer 9(16):2938–2945
Marth C, Landoni F, Mahner S, McCormack M, Gonzalez-Martin A, Colombo N (2017) Cervical cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 28(Supplement 4):iv72–iv83
(2019) Linee guida AIOM. Neoplasie dell’utero: endometrio e cervice. In: Associazione Italiana di Oncologia Medica, cap 13-25, pp 40–71
Goodman A (2015) HPV testing as a screen for cervical cancer. BMJ (Online) 350: h2372
Sahasrabuddhe VV, Parham GP, Mwanahamuntu MH, Vermund SH (2012) Cervical cancer prevention in low- and middle-income countries: Feasible, affordable, essential. Cancer Prev Res (Phila) 5(1):11-7
Liu Y et al (2019) PD-1/PD-L1 inhibitors in cervical cancer. Front Pharmacol 10(FEB):1–8
Guitarte C, Alagkiozidis I, Mize B, Stevens E, Salame G, Lee YC (2014) Glassy cell carcinoma of the cervix: A systematic review and meta-analysis. Gynecol Oncol 133:186–191
Heidi L (2014) THE KILLER WITHIN The immune system can be a powerful weapon against cancer — but researchers are still grappling with how to control it. Nature 508:24–26
Hamanishi J, Mandai M, Matsumura N, Abiko K, Baba T, Konishi I (2016) PD-1/PD-L1 blockade in cancer treatment: perspectives and issues. Int J Clin Oncol 21(3):462-73
Khan H, Gucalp R, Shapira I (2015) Evolving concepts: immunity in oncology from targets to treatments. J Oncol Volume 2015, 15 pages
G. Kwok, T. C. C. Yau, J. W. Chiu, E. Tse, and Y. L. Kwong, Pembrolizumab (Keytruda), Human vaccines and immunotherapeutics. 2016.
Miguel P, Sanmamed F, Chen L (2014) Inducible expression of B7-H1 (PD-L1) and its selective role in tumor site immune modulation. Cancer J 20(4):256–261
Frenel JS, le Tourneau C, O’Neil B, Ott PA, Piha-Paul SA, Gomez-Roca C, van Brummelen EMJ, Rugo HS, Thomas S, Saraf S, Rangwala R, Varga A (2017) Safety and efficacy of pembrolizumab in advanced, programmed death ligand 1-positive cervical cancer: results from the phase IB KEYNOTE-028 trial. In: Journal of Clinical Oncology, vol 35, pp 4035–4041
Schellens JHM, Marabelle A, Zeigenfuss S, Ding J, Pruitt SK, Chung HC (2017) Pembrolizumab for previously treated advanced cervical squamous cell cancer: preliminary results from the phase 2 KEYNOTE-158 study. J Clin Oncol 35:15_suppl, 5514-5514
Hollebecque A et al (2017) An open-label, multicohort, phase I/II study of nivolumab in patients with virus-associated tumors (CheckMate 358): efficacy and safety in recurrent or metastatic (R/M) cervical, vaginal, and vulvar cancers. J Clin Oncol 35:15_suppl, 5504-5504
Naumann RW, Hollebecque A, Meyer T, Devlin MJ, Oaknin A, Kerger J, López-Picazo JM, Machiels JP, Delord JP, Evans TRJ, Boni V, Calvo E, Topalian SL, Chen T, Soumaoro I, Li B, Gu J, Zwirtes R, Moore KN (2019) Safety and efficacy of nivolumab monotherapy in recurrent or metastatic cervical, vaginal, or vulvar carcinoma: results from the phase I/II CheckMate 358 Trial. J Clin Oncol 37(31):2825–2834
Papadopoulos KP, Crittenden MR, Johnson ML, Lockhart AC, Moore KN, Falchook GS, Formenti S, Carvajal RD, Leidner RS, Naing A, Rosen LS, Weiss GJ, Caldwell Iii W, Gao B, Paccaly A, Stankevich E, Trail P, Fury MG, Lowy I (2016) A first-in-human study of REGN2810, a monoclonal, fully human antibody to programmed death-1 (PD-1), in combination with immunomodulators including hypofractionated radiotherapy (hfRT). J Clin Oncol 34:3024
Hendry S et al (2017) Assessing tumor-infiltrating lymphocytes in solid tumors: a practical review for pathologists and proposal for a standardized method from the International Immuno-Oncology Biomarkers Working Group: Part 2: TILs in melanoma, Gastrointestinal Tract Carcinom. Adv Anat Pathol 24(6): 311–335
Heeren AM, Punt S, Bleeker MCG, Gaarenstroom KN, van der Velden J, Kenter GG, de Gruijl TD, Jordanova ES (2016) Prognostic effect of different PD-L1 expression patterns in squamous cell carcinoma and adenocarcinoma of the cervix. Mod Pathol 29(7):753–763
Roach C, Zhang N, Corigliano E, Jansson M, Toland G, Ponto G, Dolled-Filhart M, Emancipator K, Stanforth D, Kulangara K (2016) Development of a companion diagnostic PD-L1 immunohistochemistry assay for pembrolizumab therapy in non-small-cell Lung Cancer. Appl Immunohistochem Mol Morphol 24:392–397
Reck M et al (2016) Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell Lung Cancer. N Engl J Med
Dunn GP, Old LJ, Schreiber RD (2004) The immunobiology of cancer immunosurveillance and immunoediting. Immunity 21(2):137–148
Karim R, Jordanova ES, Piersma SJ, Kenter GG, Chen L, Boer JM, Melief CJM, van der Burg SH (2009) Tumor-expressed B7-H1 and B7-DC in relation to PD-1+ T-cell infiltration and survival of patients with cervical carcinoma. Clin Cancer Res 15:6341–6347
Enwere EK, Kornaga EN, Dean M, Koulis TA, Phan T, Kalantarian M, Köbel M, Ghatage P, Magliocco AM, Lees-Miller SP, Doll CM (2017) Expression of PD-L1 and presence of CD8-positive T cells in pre-treatment specimens of locally advanced cervical cancer. Mod Pathol 30(4):577–586
Nedergaard BS, Ladekarl M, Thomsen HF, Nyengaard JR, Nielsen K (2007) Low density of CD3+, CD4+ and CD8+ cells is associated with increased risk of relapse in squamous cell cervical cancer. Br J Cancer 97:1135–1138
Borcoman E, Le Tourneau C (2017) Pembrolizumab in cervical cancer: Latest evidence and clinical usefulness. Ther Adv Med Oncol 9(6):431-439
Yang W, Lu YP, Yang YZ, Kang JR, Jin YD, Wang HW (2017) Expressions of programmed death (PD)-1 and PD-1 ligand (PD-L1) in cervical intraepithelial neoplasia and cervical squamous cell carcinomas are of prognostic value and associated with human papillomavirus status. J Obstet Gynaecol Res 43(10):1602–1612
Nicol AF, de Andrade CV, Gomes SC Jr, Brusadelli MG, Lodin HM, Wells SI, Nuovo GJ (Feb. 2019) The distribution of novel biomarkers in carcinoma-in-situ, microinvasive, and squamous cell carcinoma of the uterine cervix. Ann Diagn Pathol 38:115–122
Van Driel WJ, Hogendoorn PCW, Jansen FW, Zwinderman AH, Trimbos JB, Fleuren GJ (1996) Tumor-associated eosinophilic infiltrate of cervical cancer is indicative for a less effective immune response. Hum Pathol 27(9):904–911
Hirsch FR, McElhinny A, Stanforth D, Ranger-Moore J, Jansson M, Kulangara K, Richardson W, Towne P, Hanks D, Vennapusa B, Mistry A, Kalamegham R, Averbuch S, Novotny J, Rubin E, Emancipator K, McCaffery I, Williams JA, Walker J, Longshore J, Tsao MS, Kerr KM (2017) PD-L1 Immunohistochemistry assays for lung cancer: results from phase 1 of the blueprint PD-L1 IHC assay comparison project. J Thorac Oncol 12:208–222
Mezache L, Paniccia B, Nyinawabera A, Nuovo GJ (2015) Enhanced expression of PD L1 in cervical intraepithelial neoplasia and cervical cancers. Mod Pathol 28(12):1594–1602
Liu C et al (2017) Increased expression of PD-L1 by the human papillomavirus 16 E7 oncoprotein inhibits anticancer immunity. Mol Med Rep 15(3):1063-1070
U.S FOOD AND DRUG ADMINISTRATION, FDA approves pembrolizumab for advanced cervical cancer with disease progression during or after chemotherapy, 2018. [Online]. Available: https://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm610572.htm. [Accessed: 13-Jun-2018].
Author information
Authors and Affiliations
Contributions
D’Alessandris Nicoletta and Pernazza Angelina carried out the pathologic analysis and manuscript writing; Palaia Innocenza and Di Pinto Anna collected and critically analyzed clinical data; Tomao Federica contributed to statistical analysis; Musacchio Lucia collected clinical data; Leopizzi Martina and Di Maio Valeria performed statistical analysis; Pecorella Irene contributed to the pathological analysis; Benedetti Panici Pierluigi revised the work; Della Rocca Carlo contributed to the analysis of the pathologic findings, revised, and edited the work for important intellectual content.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study complies with ethical standards of our Institution. All patients gave their informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
D’Alessandris, N., Palaia, I., Pernazza, A. et al. PD-L1 expression is associated with tumor infiltrating lymphocytes that predict response to NACT in squamous cell cervical cancer. Virchows Arch 478, 517–525 (2021). https://doi.org/10.1007/s00428-020-02922-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-020-02922-5