Abstract
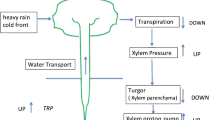
Using non-invasive ion-selective microprobes, that were placed in substomatal cavities, long-distance signalling has been investigated in intact Hordeum vulgare and Vicia faba seedlings. Heat (flame), applied to one leaf (S-leaf), triggers apoplastic ion activity (pH, pCa, pCl) transients in a distant leaf (T-leaf), all largely independent of simultaneously occurring action potential-like voltage changes. While apoplastic pCa and pH increase (Ca2+-, H+-activities decrease), pCl decreases (Cl−-activity increases). As the signal transfer from the S- to the T-leaf is too fast to account for mass flow, the heat-induced pressure change is primarily responsible for changes in voltage (H+ pump deactivation) as well as for the ion fluxes. The pCa transient precedes the pCl- and pH responses, but not the voltage change. Since the apoplastic pCl decrease (Cl− increase) occurs after the pCa increase (Ca2+ decrease) and after the depolarization, we argue that the Cl− efflux is a consequence of the Ca2+ response, but has no part in the depolarization. Kinetic analysis reveals that pH- and pCl changes are interrelated, indicated by the action of the anion channel antagonist NPPB, which inhibits both pCl- and pH changes. It is suggested that efflux of organic anions into the apoplast causes the pH increase rather than the deactivation of the plasma membrane H+ pump. Since there is considerably more information in ion activity changes than in a single action- or variation potential and heat-induced ion fluxes occur more reliably than voltage changes, released by milder stimuli, they are considered systemic signalling components superior to voltage.







Similar content being viewed by others
Abbreviations
- AP:
-
Action potential
- NPPB:
-
5-Nitro-2-(3-phenylpropylamino)benzoic acid
- S-leaf:
-
Stimulated leaf
- T-leaf:
-
Target leaf
- VP:
-
Variation potential
References
Alpi A, Amrhein N, Bertl A, Blatt MR, Blumwald E, Cervone F, Dainty J, De Michelis MI, Epstein E, Galston AW, Goldsmith MHM, Hawes C, Hell R, Hetherington A, Hofte H, Juergens G, Leaver CJ, Moroni A, Murphy A, Oparka K, Perata P, Quader H, Rausch T, Ritzenthaler C, Rivetta A, Robinson DG, Sanders D, Scheres B, Schumacher K, Sentenac H, Slayman CL, Soave C, Somerville C, Taiz L, Thiel G, Wagner R (2007) Plant neurobiology: no brain, no gain? Trends Plant Sci 12:135–136
Brenner ED, Stahlberg R, Mancuso S, Vivanco J, Baluska F, van Volkenburgh E (2006) Plant neurobiology: an integrated view of plant signaling. Trends Plant Sci 11:413–419
Davies E (1987) Action potentials as multifunctional signals in plants: a unifying hypothesis to explain apparently disparate wound responses. Plant Cell Environ 10:623–631
Davies E (2004) New functions for electrical signals in plants. New Phytol 161:607–610
Davies E (2006) Electrical signals in plants. In: Volkov AG (ed) Plant electrophysiology: theory and methods. Springer, Heidelberg, pp 407–422
Dziubinska H, Trebacz K, Zawadzki T (2001) Transmission route for action potentials and variation potentials in Helianthus annuus L. J Plant Physiol 158:1167–1172
Felle HH (2006) Apoplastic pH during low-oxygen stress in barley. Ann Bot 98:1085–1093
Felle HH, Zimmermann MR (2007) Systemic signalling in barley through action potentials. Planta 226:203–214
Felle HH, Hanstein S, Steinmeyer R, Hedrich R (2000) Dynamics of ion activities in the apoplast of the sub-stomatal cavity of intact Vicia faba leaves during stomatal closure evoked by ABA and darkness. Plant J 24:297–304
Felle HH, Herrmann A, Hückelhoven R, Kogel K-H (2004) Apoplastic pH signaling in barley leaves attacked by the powdery mildew fungus Blumeria graminis f. sp. hordei. Mol Plant Microbe Interact 17:118–123
Felle HH, Herrmann A, Hückelhoven R, Kogel K-H (2005) Root-to-shoot signalling: apoplastic alkalinization, a general stress signal and defence factor in barley (Hordeum vulgare). Protoplasma 227:17–24
Fromm J, Spanswick R (1993) Characteristics of action potentials in willow (Salix viminalis L). J Exp Bot 44:1119–1125
Grabov A, Blatt MR (1998) Co-ordination of signalling elements in guard cell ion channel control. J Exp Bot 49:351–360
Grignon C, Sentenac H (1991) pH and ionic conditions in the apoplast. Annu Rev Plant Physiol 42:103–128
Hanstein S, Felle HH (1999) The influence of atmospheric NH3 on the apoplastic pH of green leaves: a non-invasive approach with pH-sensitive microelectrodes. New Phytol 143:333–338
Hanstein S, Felle HH (2002) CO2-triggered chloride release from guard cells in intact fava bean leaves. Kinetics of the onset of stomatal closure. Plant Physiol 130:940–950
He DY, Yazaki Y, Nishizawa Y, Takai R, Sakano K, Shibuya N, Minami E (1998) Gene activation by cytoplasmic acidification in suspension-cultured rice cells in response to the elicitor N-acetylchitoheptaose. Mol Plant Microbe Interact 11:1167–1174
Hedrich R, Marten I, Lohse G, Dietrich P, Winter H, Lohaus G, Heldt HW (1994) Malate-sensitive anion channels enable guard cells to sense changes in the ambient CO2 concentration. Plant J 6:741–748
Herde O, Pena-Cortes H, Fuss H, Willmitzer L, Fisahn J (1999) Effects of mechanical wounding, current application, and heat treatment on chlorophyll fluorescence and pigment position in tomato plants. Physiol Plant 105:179–184
Johannes E, Felle HH (1985) Transport of basic amino acids in Riccia fluitans: evidence for a second binding site. Planta 166:244–251
Julien JL, Desbiez M-O, De Jaegher G, Frachisse JM (1991) Characteristics of the wave of depolarization induced by wounding in Bidens pilosa L. J Exp Bot 42:131–137
Kikuyama M, Tazawa M (2001) Mechanosensitive Ca2+ release from intracellular stores in Nitella flexilis. Plant Cell Physiol 42:358–365
Kinraide TB, Etherton B (1980) Electrical evidence for different mechanisms of uptake for basic, neutral, and basic amino acids in oat coleoptiles. Plant Physiol 65:1085–1089
Krasznai Z, Morisawa M, Krasznai ZT, Morisawa S, Inaba K, Bazsane ZK, Rubovsky B, Bodnar B, Borsos A, Marian T (2003) Gadolinium, a mechano-sensitive channel blocker, inhibits osmosis-initiated motility of sea- and freshwater fish sperm, but does not affect human or ascidian sperm motility. Cell Mot Cytoskel 55:232–243
Lautner S, Grams TEE, Matyssek R, Fromm J (2005) Characteristics of electrical signals in poplar and responses in photosynthesis. Plant Physiol 138:2200–2209
Lew RR (1998) Mapping fungal ion channel locations. Fung Gen Biol 24:69–76
Malone M (1996) Rapid, long-distance signal transmission in higher plants. Adv Bot Res 22:163–228
Mancuso S (1999) Hydraulic and electrical transmission of wound-induced signals in Vitis vinifera. Aust J Plant Physiol 26:55–61
Oja V, Savchenko G, Jakob B, Heber U (1999) pH and buffer capacities of apoplastic cell compartments in leaves. Planta 209:239–249
Pantoja O, Smith JAC (2002) Sensitivity of the plant vacuolar malate channel to pH, Ca2+ and anion channel blockers. J Membr Biol 1:31–42
Pickard BG (1973) Action potentials in higher plants. Bot Rev 39:172–201
Rhodes JD, Thain JF, Wildon DC (1996) The pathway for systemic electrical signal conduction in the wounded tomato plant. Planta 200:50–57
Rhodes JD, Thain JF, Wildon DC (1997) Evidence for physically distinct systemic signalling pathways in the wounded tomato plant. Ann Bot 84:109–116
Roblin G (1985) Analysis of the variation potential induced by wounding in plants. Plant Cell Physiol 26:255–261
Ryan PR, Dong B, Watt M, Kataoka T, Delhaize E (2003) Strategies to isolate transporters that facilitate organic anion efflux from plant cells. Plant Soil 248:61–69
Schmidt C, Schroeder JI (1994) Anion selectivity of slow anion channels in the plasma membrane of guard cells–large nitrate permeability. Plant Physiol 106:383–391
Shepard VA, Beilby MJ, Shimmen T (2002) Mechanosensory ion channels in charophyte cells: the response to touch and salinity stress. Eur Biophys J 31:341–355
Shiina T, Tazawa M (1987) Ca2+ activated Cl− channel in plasmalemma of Nitellopsis obtusa. J Membr Biol 99:137–146
Stahlberg R, Cosgrove DJ (1997) The propagation of the slow wave potential in pea epicotyls. Plant Physiol 113:209–217
Stanković B, Davies E (1996) Both action potentials and variation potentials induce proteinase inhibitor gene expression in tomato. FEBS Lett 390:275–279
Stewart PA (1983) Modern quantitative acid-base chemistry. Can J Physiol Pharmacol 61:1444–1461
Thibault G, Amiri F, Garcia R (1999) The regulation of natriuretic peptide secretion by the heart. Annu Rev Physiol 61:193–217
Trebacz K, Simonis W, Schönknecht G (1994) Cytoplasmic Ca2+, K+, Cl−, and NO3 − activities in the liverwort Conocephalum conicum L. at rest and during action potentials. Plant Physiol 106:1073–1084
Trewavas A (2007) Response to Alpi et al.: Plant neurobiology–all metaphors have a value. Trends Plant Sci 12:231–233
Ullrich CI, Novacky AJ (1992) Recent aspects of ion-induced pH changes. Curr Top Plant Biochem Physiol 11:231–248
Vodeneev VA, Opritov VA, Pyatygin SS (2006) Reversible changes of extracellular pH during action potential generation in a higher plant Cucurbita pepo. Russ J Plant Physiol 53:481–487
Walling LL (2000) The myriad plant responses to herbivores. J Plant Growth Reg 19:195–216
Wayne R (1994) The excitability of plant cells: with special emphasis on Characean internodal cells. Bot Rev 60:265–367
Wildon DC, Thain JF, Minchin PEH, Gubb IR, Reilly AJ, Skipper YD, Doherty HM, O’Donnell PJ, Bowles DJ (1992) Electrical signalling and systemic proteinase inhibitor induction in the wounded plant. Nature 360:62–65
Wilkinson S (1999) pH as a stress signal. Plant Growth Reg 29:87–99
Zhang W, Fan L-M, Wu W-H (2007) Osmo-sensitive and stretch-activated calcium-permeable channels in Vicia faba guard cells are regulated by actin dynamics. Plant Physiol 143:1140–1151
Acknowledgments
The financial support of this work by the “Deutsche Forschungsgemeinschaft” (Fe 213/15-1,2) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zimmermann, M.R., Felle, H.H. Dissection of heat-induced systemic signals: superiority of ion fluxes to voltage changes in substomatal cavities. Planta 229, 539–547 (2009). https://doi.org/10.1007/s00425-008-0850-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-008-0850-x