Abstract
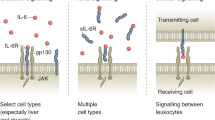
Interleukin-6 (IL-6) is produced locally in working skeletal muscle and can account for the exercise-induced increase in plasma IL-6. The transcription rate for IL-6 in muscle nuclei isolated from muscle biopsies during exercise is very high and is enhanced further when muscle glycogen content is low. Furthermore, cultured human primary muscle cells can increase IL-6 mRNA when incubated with the calcium ionophore ionomycin and it is likely that myocytes produce IL-6 in response to muscle contraction. The biological roles of muscle-derived IL-6 have been investigated in studies in which human recombinant IL-6 was infused in healthy volunteers to mimic closely the IL-6 concentrations observed during prolonged exercise. Using stable isotopes, we have demonstrated that physiological concentrations of IL-6 induce lipolysis. Although we have yet to determine the precise biological action of muscle-derived IL-6, our data support the hypothesis that the role of IL-6 released from contracting muscle during exercise is to act in a hormone-like manner to mobilize extracellular substrates and/or augment substrate delivery during exercise. In addition, IL-6 inhibits low-level TNF-α production, and IL-6 produced during exercise probably inhibits TNF-α-induced insulin resistance in peripheral tissues. Hence, IL-6 produced by skeletal muscle during contraction may play an important role in the beneficial health effects of exercise

Similar content being viewed by others
References
Akira S, Taga T, Kishimoto T (1993) Interleukin-6 in biology and medicine. Adv Immunol 54:1–78
Bartoccioni E, Michaelis D, Hohlfeld R (1994) Constitutive and cytokine-induced production of interleukin-6 by human myoblasts. Immunol Lett 42:135–138
Bastard JP, Jardel C, Bruckert E, Blondy P, Capeau J, Laville M, Vidal H, Hainque B (2000) Elevated levels of interleukin 6 are reduced in serum and subcutaneous adipose tissue of obese women after weight loss. J Clin Endocrinol Metab 85:3338–3342
Begum N, Ragolia L (1996) Effect of tumor necrosis factor-alpha on insulin action in cultured rat skeletal muscle cells. Endocrinology 137:2441–2446
Berridge MJ (1993) Inositol trisphosphate and calcium signalling. Nature 361:315–325
Boden G (1997) Role of fatty acids in the pathogenesis of insulin resistance and NIDDM. Diabetes 46:3–10
Borst SE, Bagby GJ (2002) Neutralization of tumor necrosis factor reverses age-induced impairment of insulin responsiveness in skeletal muscle of Sprague-Dawley rats. Metabolism 51:1061–1064
Bruunsgaard H, Galbo H, Halkjaer-Kristensen J, Johansen TL, MacLean DA, Pedersen BK (1997) Exercise-induced increase in interleukin-6 is related to muscle damage. J Physiol (Lond) 499:833–841
Bruunsgaard H, Andersen-Ranberg K, Jeune B, Pedersen AN, Skinhj P, Pedersen BK (1999) A high plasma concentration of TNF-alpha is associated with dementia in centenarians. J Gerontol 54A:357–364
Castell LM, Poortmans JR, Leclercq R, Brasseur M, Duchateau J, Newsholme EA (1997) Some aspects of the acute phase response after a marathon race, and the effects of glutamine supplementation. Eur J Appl Physiol 75:47–53
Chapman PB, Lester TJ, Casper ES, Gabrilove JL, Wong GY, Kempin SJ, Gold PJ, Welt S, Warren RS, Starnes HF (1987) Clinical pharmacology of recombinant human tumor necrosis factor in patients with advanced cancer. J Clin Oncol 5:1942–1951
Ciaraldi TP, Carter L, Mudaliar S, Kern PA, Henry RR (1998) Effects of tumor necrosis factor-alpha on glucose metabolism in cultured human muscle cells from nondiabetic and type 2 diabetic subjects. Endocrinology 139:4793–4800
Crabtree GR (1999) Generic signals and specific outcomes: signaling through Ca2+, calcineurin, and NF-AT. Cell 96:611–614
Creagan ET, Kovach JS, Moertel CG, Frytak S, Kvols LK (1988) A phase I clinical trial of recombinant human tumor necrosis factor. Cancer 62:2467–2471
Croisier JL, Camus G, Venneman I, Deby-Dupont G, Juchmes-Ferir A, Lamy M, Crielaard JM, Deby C, Duchateau J (1999) Effects of training on exercise-induced muscle damage and interleukin 6 production. Muscle Nerve 22:208–212
De Rossi M, Bernasconi P, Baggi F, de Waal MR, Mantegazza R (2000) Cytokines and chemokines are both expressed by human myoblasts: possible relevance for the immune pathogenesis of muscle inflammation. Int Immunol 12:1329–1335
Del Aguila LF, Claffey KP, Kirwan JP (1999) TNF-alpha impairs insulin signaling and insulin stimulation of glucose uptake in C2C12 muscle cells. Am J Physiol 276:E849–E855
Dela F, Larsen JJ, Mikines KJ, Ploug T, Petersen LN, Galbo H (1995) Insulin-stimulated muscle glucose clearance in patients with NIDDM. Effects of one-legged physical training. Diabetes 44:1010–1020
DeRijk RH, Boelen A, Tilders FJ, Berkenbosch F (1994) Induction of plasma interleukin-6 by circulating adrenaline in the rat. Psychoneuroendocrinology 19:155–163
Drenth JP, Uum SH van, Deuren M van, Pesman GJ, Jongekrug J van der ven, Meer JW van der (1995) Endurance run increases circulating IL-6 and IL-1ra but downregulates ex vivo TNF-alpha and IL-1beta production. J Appl Physiol 79:1497–1503
Febbraio MA, Pedersen BK (2002) Muscle-derived interleukin-6: mechanisms for activation and possible biological roles. FASEB J 16:1335–1347
Febbraio MA, Steensberg A, Starkie RL, McConell GK, Kingwell BA (2002a) Contracting skeletal muscle releases IL-6, but not TNF-alpha, in healthy subjects and patients with type 2 diabetes. Diabetes 2002 (In Press)
Febbraio MA, Ott P, Nielsen HB, Steensberg A, Keller C, Krustrup I, Secher NH, Pedersen BK (2002b) Exercise induces hepatosplanchnic release of heat shock protein 72 in humans. J Physiol (Lond) 544:957–962
Fiers W (1991) Tumor necrosis factor. Characterization at the molecular, cellular and in vivo level. FEBS Lett 285:199–212
Fried SK, Bunkin DA, Greenberg AS (1998) Omental and subcutaneous adipose tissues of obese subjects release interleukin-6: depot difference and regulation by glucocorticoid. J Clin Endocrinol Metab 83:847–850
Gadient RA, Patterson PH (1999) Leukemia inhibitory factor, Interleukin 6, and other cytokines using the GP130 transducing receptor: roles in inflammation and injury. Stem Cells 17:127–137
Ghosh A, Greenberg ME (1995) Calcium signaling in neurons: molecular mechanisms and cellular consequences. Science 268:239–247
Gleeson M, Bishop NC (2000) Special feature for the Olympics: effects of exercise on the immune system: modification of immune responses to exercise by carbohydrate, glutamine and anti-oxidant supplements. Immunol Cell Biol 78:554–561
Halse R, Pearson SL, McCormack JG, Yeaman SJ, Taylor R (2001) Effects of tumor necrosis factor-alpha on insulin action in cultured human muscle cells. Diabetes 50:1102–1109
Hardin J, Kroeker K, Chung B, Gall DG (2000) Effect of proinflammatory interleukins on jejunal nutrient transport. Gut 47:184–191
Hauner H, Petruschke T, Russ M, Rohrig K, Eckel J (1995) Effects of tumour necrosis factor alpha (TNF alpha) on glucose transport and lipid metabolism of newly-differentiated human fat cells in cell culture. Diabetologia 38:764–771
Helge JW, Stallknecht B, Pedersen BK, Galbo H, Kiens B, Richter EA (2002) The effect of graded exercise on IL-6 release and glucose uptake in skeletal muscle. J Physiol (Lond) (In Press) DOI 10.1113/jphysiol.2002.030437
Hellsten Y, Frandsen U, Orthenblad N, Sjodin N, Richter EA (1997) Xanthine oxidase in human skeletal muscle following eccentric exercise: a role of inflammation. J Physiol (Lond) 498:239–248
Holst JJ, Binderup M (2002) [Fatty tissue and insulin resistance: resistin and adiponectin]. Ugeskr Laeger 164:2173–2176
Hotamisligil GS (1999a) The role of TNFalpha and TNF receptors in obesity and insulin resistance. J Intern Med 245:621–625
Hotamisligil GS (1999b) Mechanisms of TNF-alpha-induced insulin resistance. Exp Clin Endocrinol Diabetes 107:119–125
Hotamisligil GS, Shargill NS, Spiegelman BM (1993) Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science 259:87–91
Hotamisligil GS, Budavari A, Murray D, Spiegelman BM (1994) Reduced tyrosine kinase activity of the insulin receptor in obesity-diabetes. Central role of tumor necrosis factor-alpha. J Clin Invest 94:1543–1549
Jonsdottir I, Schjerling P, Ostrowski K, Asp S, Richter EA, Pedersen BK (2000) Muscle contractions induces interleukin-6 mRNA production in rat skeletal muscles. J Physiol (Lond) 528:157–163
Katsuki A, Sumida Y, Murashima S, Murata K, Takarada Y, Ito K, Fujii M, Tsuchihashi K, Goto H, Nakatani K, Yano Y (1998) Serum levels of tumor necrosis factor-alpha are increased in obese patients with non-insulin-dependent diabetes mellitus. J Clin Endocrinol Metab 83:859–862
Keller C, Steensberg A, Pilegaard H, Osada T, Saltin B, Pedersen BK, Neufer PD (2001) Transcriptional activation of the IL-6 gene in human contracting skeletal muscle: influence of muscle glycogen content. FASEB J 15:2748–2750
Keller C, Hellsten Y, Pilegaard H, Febbraio MA, Pedersen BK (2002) Human muscle cells express IL-6 via a Ca2+-dependent pathway. J Physiol (Lond) (In Press)
Klouche M, Bhakdi S, Hemmes M, Rose-John S (1999) Novel path to activation of vascular smooth muscle cells: up-regulation of gp130 creates an autocrine activation loop by IL-6 and its soluble receptor. J Immunol 163:4583–4589
Liao J, Keiser JA, Scales WE, Kunkel SL, Kluger MJ (1995a) Role of corticosterone in TNF and IL-6 production in isolated perfused rat liver. Am J Physiol 268:R699–R706
Liao J, Keiser JA, Scales WE, Kunkel SL, Kluger MJ (1995b) Role of epinephrine in TNF and IL-6 production from isolated perfused rat liver. Am J Physiol 268:R896–R901
Matthys P, Mitera T, Heremans H, Van Damme J, Billiau A (1995) Anti-gamma interferon and anti-interleukin-6 antibodies affect staphylococcal enterotoxin B-induced weight loss, hypoglycemia, and cytokine release in d-galactosamine-sensitized and unsensitized mice. Infect Immun 63:1158–1164
McCarthy DA, Dale MM (1988) The leucocytosis of exercise. A review and model. Sports Med 6:333–363
Michie HR, Spriggs DR, Manogue KR, Sherman ML, Revhaug A, O'Dwyer ST, Arthur K, Dinarello CA, Cerami A, Wolff SM (1988) Tumor necrosis factor and endotoxin induce similar metabolic responses in human beings. Surgery 104:280–286
Mikines KJ, Sonne B, Farrell PA, Tronier B, Galbo H (1989) Effect of training on the dose-response relationship for insulin action in men. J Appl Physiol 66:695–703
Mishima Y, Kuyama A, Tada A, Takahashi K, Ishioka T, Kibata M (2001) Relationship between serum tumor necrosis factor-alpha and insulin resistance in obese men with Type 2 diabetes mellitus. Diabetes Res Clin Pract 52:119–123
Mizuhara H, O'Neill E, Seki N, Ogawa T, Kusunoki C, Otsuka K, Satoh S, Niwa M, Senoh H, Fujiwara H (1994) T cell activation-associated hepatic injury: mediation by tumor necrosis factors and protection by interleukin 6. J Exp Med 179:1529–1537
Moldoveanu AI, Shephard RJ, Shek PN (2000) Exercise elevates plasma levels but not gene expression of IL-1beta, IL-6, and TNF-alpha in blood mononuclear cells. J Appl Physiol 89:1499–1504
Nagaraju K, Raben N, Merritt G, Loeffler L, Kirk K, Plotz P (1998) A variety of cytokines and immunologically relevant surface molecules are expressed by normal human skeletal muscle cells under proinflammatory stimuli. Clin Exp Immunol 113:407–414
Nehlsen-Canarella SL, Fagoaga OR, Nieman DC (1997) Carbohydrate and the cytokine response to 2.5 hours of running. J Appl Physiol 82:1662–1667
Nielsen HB, Secher N, Pedersen BK (1996) Lymphocytes and NK cell activity during repeated bouts of maximal exercise. Am J Physiol 271:R222–R227
Nieman DC, Fagoaga OR, Butterworth DE, Warren BJ, Utter A, Davis JM, Henson DA, Nehlsen-Canarella SL (1997a) Carbohydrate supplementation affects blood granulocyte and monocyte trafficking but not function after 2.5 hours of running. J Appl Physiol 82:1385–1394
Nieman DC, Henson DA, Garner EB, Butterworth DE, Warren BJ, Utter A, Davis JM, Fagoaga OR, Nehlsen-Cannarella SL (1997b) Carbohydrate affects natural killer cell redistribution but not activity after running. Med Sci Sports Exerc 29:1318–1324
Nieman DC, Nehlsen-Canarella SL, Fagoaga OR, Henson DA, Utter A, Davis JM, Williams F, Butterworth DE (1998a) Effects of mode and carbohydrate on the granulocyte and monocyte response to intensive prolonged exercise. J Appl Physiol 84:1252–1259
Nieman DC, Nehlsen-Canarella SL, Fagoaga OR, Henson DA, Utter A, Davis JM, Williams F, Butterworth DE (1998b) Influence of mode and carbohydrate on the cytokine response to heavy exertion. Med Sci Sports Exerc 30:671–678
Nonogaki K, Fuller GM, Fuentes NL, Moser AH, Staprans I, Grunfeld C, Feingold KR (1995) Interleukin-6 stimulates hepatic triglyceride secretion in rats. Endocrinology 136:2143–2149
Northoff H, Weinstock C, Berg A (1994) The cytokine response to strenuous exercise. Int J Sports Med 15 (Suppl 3):S167–S171
Olson EN, Williams RS (2000) Remodeling muscles with calcineurin. Bioessays 510–519
Ostrowski K, Hermann C, Bangash A, Schjerling P, Nielsen JN, Pedersen BK (1998a) A trauma-like elevation in plasma cytokines in humans in response to treadmill running. J Physiol (Lond) 508:889–894
Ostrowski K, Rohde T, Zacho M, Asp S, Pedersen BK (1998b) Evidence that interleukin-6 is produced in human skeletal muscle during prolonged running. J Physiol (Lond) 508:949–953
Ostrowski K, Rohde T, Asp S, Schjerling P, Pedersen BK (1999) Pro- and anti-inflammatory cytokine balance and strenuous exercise in humans. J Physiol (Lond) 515:287–291
Ostrowski K, Schjerling P, Pedersen BK (2000) Physical activity and plasma interleukin-6 in humans: effect of intensity of exercise. Eur J Appl Physiol 83:512–515
Ostrowski K, Rohde T, Asp S, Schjerling P, Pedersen BK (2001) Chemokines are elevated in plasma after strenuous exercise in humans. Eur J Appl Physiol 84:244–245
Patton JS, Shepard HM, Wilking H, Lewis G, Aggarwal BB, Eessalu TE, Gavin LA, Grunfeld C (1986) Interferons and tumor necrosis factors have similar catabolic effects on 3T3 L1 cells. Proc Natl Acad Sci USA 83:8313–8317
Pedersen BK, Hoffman-Goetz L (2000) Exercise and the immune system: regulation, integration and adaption. Physiol Rev 80:1055–1081
Pedersen BK, Ostrowski K, Rohde T, Bruunsgaard H (1998) The cytokine response to strenuous exercise. Can J Physiol Pharmacol 76:505–511
Pedersen BK, Steensberg A, Schjerling P (2001) Muscle-derived interleukin-6: possible biological effects. J Physiol (Lond) 536:329–337
Pickup JC, Chusney GD, Thomas SM, Burt D (2000) Plasma interleukin-6, tumour necrosis factor alpha and blood cytokine production in type 2 diabetes. Life Sci 67:291–300
Qi C, Pekala PH (2000) Tumor necrosis factor-alpha-induced insulin resistance in adipocytes. Proc Soc Exp Biol Med 223:128–135
Rao A, Luo C, Hogan PG (1997) Transcription factors of the NFAT family: regulation and function. Annu Rev Immunol 15:707–747
Rhind SG, Castellani JW, Brenner IK, Shepard RJ, Zamecnik J, Montain SJ, Young AJ, Shek PN (2001) Intracellular monocyte and serum cytokine expression is modulated by exhausting exercise and cold exposure. Am J Physiol 281:R66–R75
Ridker PM, Rifai N, Stampfer MJ, Hennekens CH (2000) Plasma concentration of interleukin-6 and the risk of future myocardial infarction among apparently healthy men. Circulation 101:1767–1772
Rohde T, MacLean DA, Richter EA, Kiens B, Pedersen BK (1997) Prolonged submaximal eccentric exercise is associated with increased levels of plasma IL-6. Am J Physiol 273:E85–E91
Saghizadeh M, Ong JM, Garvey WT, Henry RR, Kern PA (1996) The expression of TNF alpha by human muscle: relationship to insulin resistance. J Clin Invest 97:1111–1116
Sethi JK, Hotamisligil GS (1999) The role of TNF alpha in adipocyte metabolism. Semin Cell Dev Biol 10:19–29
Spriggs DR, Sherman ML, Michie H, Arthur KA, Imamura K, Wilmore D, Frei E III, Kufe DW (1988) Recombinant human tumor necrosis factor administered as a 24-hour intravenous infusion. A phase I and pharmacologic study. J Natl Cancer Inst 80:1039–1044
Starkie RL (2001) Circulating monocytes are not the source of elevations in plasma IL-6 and TNF-alpha levels after prolonged running. Am J Physiol 280:C769–C774
Starkie RL, Angus DJ, Rolland J, Hargreaves M, Febbraio M (2000) Effect of prolonged submaximal exercise and carbohydrate ingestion on monocyte intracellular cytokine production in humans. J Physiol (Lond) 528:647–655
Starkie RL, Arkinstall MJ, Koukoulas I, Hawley JA, Febbraio MA (2001) Carbohydrate ingestion attenuates the increase in plasma interleukin-6, but not skeletal muscle interleukin-6 mRNA, during exercise in humans. J Physiol (Lond) 533:585–591
Starnes HF Jr, Warren RS, Jeevanandam M, Gabrilove JL, Larchian W, Oettgen HF, Brennan MF (1988) Tumor necrosis factor and the acute metabolic response to tissue injury in man. J Clin Invest 82:1321–1325
Steensberg A, Hall G van, Osada T, Sacchetti M, Saltin B, Pedersen BK (2000) Production of IL-6 in contracting human skeletal muscles can account for the exercise-induced increase in plasma IL-6. J Physiol (Lond) 529:237–242
Steensberg A, Febbraio MA, Osada T, Schjerling P, Hall G van, Saltin B, Pedersen BK (2001a) Interleukin-6 production in contracting human skeletal muscle is influenced by pre-exercise muscle glycogen content. J Physiol (Lond) 537:633–639
Steensberg A, Toft AD, Bruunsgaard H, Sandmand M, Halkjaer-Kristensen J, Pedersen BK (2001b) Strenuous exercise decreases the percentage of type 1 T cells in the circulation. J Appl Physiol 91:1708–1712
Steensberg A, Vissing J, Pedersen BK (2001c) Lack of IL-6 production during exercise in patients with mitochondrial myopathy. Eur J Appl Physiol 84:155–157
Steensberg A, Toft AD, Schjerling P, Halkjaer-Kristensen J, Pedersen BK (2001d) Plasma interleukin-6 during strenuous exercise: role of epinephrine. Am J Physiol 281:C1001–C1004
Stouthard JM, Romijn JA, van der PT, Endert E, Klein S, Bakker PJ, Veenhof CH, Sauerwein HP (1995) Endocrinologic and metabolic effects of interleukin-6 in humans. Am J Physiol 268:E813–E819
Stouthard JM, Oude Elferink RP, Sauerwein HP (1996) Interleukin-6 enhances glucose transport in 3T3-L1 adipocytes. Biochem Biophys Res Commun 220:241–245
Suzuki K, Yamada M, Kurakake S, Okamura N, Yamaya K, Liu Q, Kudoh S, Kowatari K, Nakaji S, Sugawara K (2000) Circulating cytokines and hormones with immunosuppressive but neutrophil-priming potentials rise after endurance exercise in humans. Eur J Appl Physiol 81:281–287
Toft AD, Ostrowski K, Asp S, Møller K, Iversen S, Hermann C, Søndergaard SR, Pedersen BK (2000) The effects of n-3 PUFA on the cytokine response to strenuous exercise. J Appl Physiol 89:2401–2405
Toft AD, Jensen LB, Bruunsgaard H, Ibfelt T, Halkjaer-Kristensen J, Febbraio M, Pedersen BK (2002) Cytokine response to eccentric exercise in young and elderly humans. Am J Physiol 283:C289–C295
Ullum H, Haahr PM, Diamant M, Palmo J, Halkjaer-Kristensen J, Pedersen BK (1994) Bicycle exercise enhances plasma IL-6 but does not change IL-1alpha, IL-1beta, IL-6, or TNF-alpha pre-mRNA in BMNC. J Appl Physiol 77:93–97
Vozarova B, Weyer C, Hanson K, Tataranni PA, Bogardus C, Pratley RE (2001) Circulating interleukin-6 in relation to adiposity, insulin action, and insulin secretion. Obes Res 9:414–417
Wallenius V, Wallenius K, Ahren B, Rudling M, Carlsten H, Dickson SL, Ohlsson C, Jansson JO (2002) Interleukin-6-deficient mice develop mature-onset obesity. Nat Med 8:75–79
Yan SF, Tritto I, Pinsky D, Liao H, Huang J, Fuller G, Brett J, May L, Stern D (1995) Induction of interleukin 6 (IL-6) by hypoxia in vascular cells. Central role of the binding site for nuclear factor-IL-6. J Biol Chem 270:11463–11471
Youd JM, Rattigan S, Clark MG (2000) Acute impairment of insulin-mediated capillary recruitment and glucose uptake in rat skeletal muscle in vivo by TNF-alpha. Diabetes 49:1904–1909
Yudkin JS, Kumari M, Humphries SE, Mohammed-Ali V (2000) Inflammation, obesity, stress and coronary heart disease: is interleukin-6 the link? Atherosclerosis 148:209–214
Acknowledgements
The work from our research laboratories on IL-6 has been supported by The Danish National Research Foundation (#504-14) and The Danish Medical Research Council (22-01-0019).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pedersen, B.K., Steensberg, A., Keller, P. et al. Muscle-derived interleukin-6: lipolytic, anti-inflammatory and immune regulatory effects. Pflugers Arch - Eur J Physiol 446, 9–16 (2003). https://doi.org/10.1007/s00424-002-0981-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-002-0981-z