Abstract
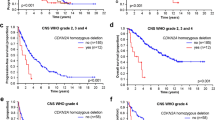
Glycosaminoglycans are major components of brain extracellular matrix (ECM), although heparan sulfate (HS) contribution in brain physiology and carcinogenesis remains underinvestigated. This study examined HS content and distribution in glioblastoma multiforme (GBM) tissues in the context of potential molecular mechanisms underlying its deregulation in brain tumours. Totally, 42 tissue samples and paraffin-embedded tissues for 31 patients with different prognosis were investigated. HS expression was demonstrated in 50–55% of the GBM tumours by immunohistochemistry (IHC), while almost no HS content was detected in the surrounding paratumourous brain tissues. Heterogeneous HS distribution in the HS-positive tumours was more related to the necrosis or glandular-like brain zones rather than glioma cells with high or low Ki-67 index. According the Kaplan–Meier curves, HS accumulation in glioma cells was associated with low relapse-free survival (RS) of the GBM patients (p < 0.05) and was likely to be due to the increased transcriptional activity of HSPG core proteins (syndecan-1, 2–3 fold; glypican-1, 2,5 fold; perlecan/HSPG2, 13–14 fold). Activation of perlecan/HSPG2 expression correlated with the patients’ survival according Kaplan–Meier (p = 0.0243) and Cox proportional-hazards regression (HR = 3.1; P(Y) = 0.03) analyses, while up-regulation of syndecan-1 and glypican-1 was not associated with the patients survival. Taken together, the results indicate that increase of HS content and up-regulation of perlecan/HSPG2 expression in glioblastoma tissues contribute to tumour development through the transformation of brain extracellular matrix into tumour microenvironment, and represent negative prognostic factors for glioblastoma progression.




Similar content being viewed by others
References
Bellail AC, Hunter SB, Brat DJ, Tan C, Van Meir EG (2004) Microregional extracellular matrix heterogeneity in brain modulates glioma cell invasion. Int J Biochem Cell Biol 36(6):1046–1069. https://doi.org/10.1016/j.biocel.2004.01.013
Chintala SK, Sawaya R, Gokaslan ZL, Fuller G, Rao JS (1996) Immunohistochemical localization of extracellular matrix proteins in human glioma, both in vivo and in vitro. Cancer Lett 101(1):107–114
Farach-Carson MC, Warren CR, Harrington DA, Carson DD (2014) Border patrol: insights into the unique role of perlecan/heparan sulfate proteoglycan 2 at cell and tissue borders. Matrix Biol 34:64–79. https://doi.org/10.1016/j.matbio.2013.08.004
Ford-Perriss M, Turner K, Guimond S, Apedaile A, Haubeck HD, Turnbull J, Murphy M (2003) Localisation of specific heparan sulfate proteoglycans during the proliferative phase of brain development. Dev Dyn 227(2):170–184. https://doi.org/10.1002/dvdy.10298
Gubbiotti MA, Neill T, Iozzo RV (2017) A current view of perlecan in physiology and pathology: a mosaic of functions. Matrix Biol 57–58:285–298. https://doi.org/10.1016/j.matbio.2016.09.003
Hagihara K, Watanabe K, Chun J, Yamaguchi Y (2000) Glypican-4 is an FGF2-binding heparan sulfate proteoglycan expressed in neural precursor cells. Dev Dyn 219(3):353–367. https://doi.org/10.1002/1097-0177(2000)9999:9999%3C::AID-DVDY1059%3E3.0.CO;2-%23
Ho IAW, Shim WSN (2017) Contribution of the microenvironmental niche to glioblastoma heterogeneity. Biomed Res Int 9634172. https://doi.org/10.1155/2017/9634172
Ilhan-Mutlu A, Siehs C, Berghoff AS, Ricken G, Widhalm G, Wagner L, Preusser M (2016) Expression profiling of angiogenesis-related genes in brain metastases of lung cancer and melanoma. Tumour Biol 37(1):1173–1182. https://doi.org/10.1007/s13277-015-3790-7
Inatani M, Irie F, Plump AS, Tessier-Lavigne M, Yamaguchi Y (2003) Mammalian brain morphogenesis and midline axon guidance require heparan sulfate. Science 302(5647):1044–1046. https://doi.org/10.1126/science.1090497
Jiang X, Couchman JR (2003) Perlecan and tumor angiogenesis. J Histochem Cytochem 51(11):1393–1410. https://doi.org/10.1177/002215540305101101
Kraushaar DC, Dalton S, Wang L (2013) Heparan sulfate: a key regulator of embryonic stem cell fate. Biol Chem 394(6):741–751. https://doi.org/10.1515/hsz-2012-0353
Lander AD, Stipp CS, Ivins JK (1996) The glypican family of heparan sulfate proteoglycans: major cell-surface proteoglycans of the developing nervous system. Perspect Dev Neurobiol 3(4):347–358
Logun MT, Bisel NS, Tanasse EA, Zhao W, Gunasekera B, Mao L, Karumbaiah L (2016) Glioma cell invasion is significantly enhanced in composite hydrogel matrices composed of chondroitin 4- and 4,6-sulfated glycosaminoglycans. J Mater Chem B Mater Biol Med 4(36):6052–6064. https://doi.org/10.1039/C6TB01083K
Lundmark K, Tran PK, Kinsella MG, Clowes AW, Wight TN, Hedin U (2001) Perlecan inhibits smooth muscle cell adhesion to fibronectin: role of heparan sulfate. J Cell Physiol 188(1):67–74. https://doi.org/10.1002/jcp.1094
Mercier F (2016) Fractones: extracellular matrix structures controlling stem cell fate and growth factor activity in the brain in health and disease. Cell Mol Life Sci 73(24):4661–4674. https://doi.org/10.1007/s00018-016-2314-y
Nevo I, Woolard K, Cam M, Li A, Webster JD, Kotliarov Y, Kim HS, Ahn S, Walling J, Kotliarova S, Belova G, Song H, Bailey R, Zhang W, Fine HA (2014) Identification of molecular pathways facilitating glioma cell invasion in situ. PLoS One 9(11):e111783. https://doi.org/10.1371/journal.pone.0111783
Nicholson C, Hrabětová S (2017) Brain extracellular space: the final frontier of neuroscience. Biophys J. https://doi.org/10.1016/j.bpj.2017.06.052
Onishi M, Ichikawa T, Kurozumi K, Date I (2011) Angiogenesis and invasion in glioma. Brain Tumor Pathol 28(1):13–24. https://doi.org/10.1007/s10014-010-0007-z
Park JB, Kwak HJ, Lee SH (2008) Role of hyaluronan in glioma invasion. Cell Adh Migr 2(3):202–207
Phillips JJ (2012) Novel therapeutic targets in the brain tumor. microenvironment Oncotarget 3(5):568–575. https://doi.org/10.18632/oncotarget.493
Qazi MA, Vora P, Venugopal C, Sidhu SS, Moffat J, Swanton C, Singh SK (2017) Intratumoral heterogeneity: pathways to treatment resistance and relapse in human glioblastoma. Ann Oncol Apr 12. https://doi.org/10.1093/annonc/mdx169
Quail DF, Joyce JA (2017) The microenvironmental landscape of brain tumors. Cancer Cell 31(3):326–341. https://doi.org/10.1016/j.ccell.2017.02.009
Radotra B, McCormick D (1997) Glioma invasion in vitro is mediated by CD44-hyaluronan interactions. J Pathol 181(4):434–438. https://doi.org/10.1002/(SICI)1096-9896(199704)181:4<434::AID-PATH797>3.0.CO;2-S
Roberts J, Kahle MP, Bix GJ (2012) Perlecan and the blood-brain barrier: beneficial proteolysis? Front Pharmacol 3:155. https://doi.org/10.3389/fphar.2012.00155
Saito T, Sugiyama K, Hama S, Yamasaki F, Takayasu T, Nosaka R, Onishi S, Muragaki Y, Kawamata T, Kurisu K (2017) High expression of glypican-1 predicts dissemination and poor prognosis in glioblastomas. World Neurosurg. https://doi.org/10.1016/j.wneu.2017.05.165
Senner V, Ratzinger S, Mertsch S, Grässel S, Paulus W (2008) Collagen XVI expression is upregulated in glioblastomas and promotes tumor cell adhesion. FEBS Lett 582(23–24):3293–3300. https://doi.org/10.1016/j.febslet.2008.09.017
Shao C, Shi X, Phillips JJ, Zaia J (2013) Mass spectral profiling of glycosaminoglycans from histological tissue surfaces. Anal Chem 85(22):10984–10991. https://doi.org/10.1021/ac402517s
Smith PD, Coulson-Thomas VJ, Foscarin S, Kwok JC, Fawcett JW (2015) “GAG-ing with the neuron”: the role of glycosaminoglycan patterning in the central nervous system. Exp Neurol 274(Pt B):100–114. https://doi.org/10.1016/j.expneurol.2015.08.004
Steck PA, Moser RP, Bruner JM, Liang L, Freidman AN, Hwang TL, Yung WK (1989) Altered expression and distribution of heparan sulfate proteoglycans in human gliomas. Cancer Res 49(8):2096–2103
Su JH, Cummings BJ, Cotman CW (1992) Localization of heparan sulfate glycosaminoglycan and proteoglycan core protein in aged brain and Alzheimer’s disease. Neuroscience 51(4):801–813
Tran VM, Wade A, McKinney A, Chen K, Lindberg OR, Engler JR, Persson AI, Phillips JJ (2017) Heparan sulfate glycosaminoglycans in glioblastoma promote tumor invasion. Mol Cancer Res. https://doi.org/10.1158/1541-7786.MCR-17-0352
Ushakov VS, Tsidulko AY, de La Bourdonnaye G, Kazanskaya GM, Volkov AM, Kiselev RS, Kobozev VV, Kostromskaya DV, Gaytan AS, Krivoshapkin AL, Aidagulova SV, Grigorieva EV (2017) Heparan sulfate biosynthetic system is inhibited in human glioma due to EXT1/2 and HS6ST1/2 down-regulation. Int J Mol Sci 18(11):E2301. https://doi.org/10.3390/ijms18112301
Wade A, Robinson AE, Engler JR, Petritsch C, James CD, Phillips JJ (2013) Proteoglycans and their roles in brain cancer. FEBS J 280(10):2399–2417. https://doi.org/10.1111/febs.12109
Watanabe A, Mabuchi T, Satoh E, Furuya K, Zhang L, Maeda S, Naganuma H (2006) Expression of syndecans, a heparan sulfate proteoglycan, in malignant gliomas: participation of nuclear factor-kappaB in upregulation of syndecan-1 expression. J Neurooncol 77(1):25–32. https://doi.org/10.1007/s11060-005-9010-3
Whitelock JM, Melrose J, Iozzo RV (2008) Diverse cell signaling events modulated by perlecan. Biochemistry 47(43):11174–11183. https://doi.org/10.1021/bi8013938
Winkler S, Stahl RC, Carey DJ, Bansal R (2002) Syndecan-3 and perlecan are differentially expressed by progenitors and mature oligodendrocytes and accumulate in the extracellular matrix. J Neurosci Res 69(4):477–487. https://doi.org/10.1002/jnr.10311
Xiong A, Kundu S, Forsberg-Nilsson K (2014) Heparan sulfate in the regulation of neural differentiation and glioma development. FEBS J 281(22):4993–5008. https://doi.org/10.1111/febs.13097
Yamaguchi Y, Inatani M, Matsumoto Y, Ogawa J, Irie F (2010) Roles of heparan sulfate in mammalian brain development current views based on the findings from Ext1 conditional knockout studies. Prog Mol Biol Transl Sci 93:133–152. https://doi.org/10.1016/S1877-1173(10)93007-X
Acknowledgements
The authors thank Natalya Brueva for the help with immunohistochemistry analysis and Ivan Murashov for the help with microscopy. This study was funded by Russian Science Foundation (Grant number 16-15-10243).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Galina M. Kazanskaya, Alexandra Y. Tsidulko, and Alexander M. Volkov have contributed equally.
Rights and permissions
About this article
Cite this article
Kazanskaya, G.M., Tsidulko, A.Y., Volkov, A.M. et al. Heparan sulfate accumulation and perlecan/HSPG2 up-regulation in tumour tissue predict low relapse-free survival for patients with glioblastoma. Histochem Cell Biol 149, 235–244 (2018). https://doi.org/10.1007/s00418-018-1631-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-018-1631-7