Abstract
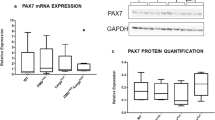
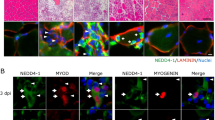
Regenerating muscle fibers emerge from quiescent satellite cells, which differentiate into mature multinuclear myofibers upon activation. It has recently been found that ATOH8, a bHLH transcription factor, is regulated during myogenic differentiation. In this study, expression and localization of ATOH8, the other well-described regeneration markers, vimentin, nestin and neonatal myosin, and the satellite cell marker Pax7 were analyzed on protein level in human myopathy samples by immunofluorescence studies. On mRNA level, expression levels of ATOH8 and vimentin were studied by quantitative real-time PCR. ATOH8 is expressed in activated satellite cells and proliferating myoblasts of human skeletal muscle tissue. Quantitative analyses of ATOH8+, Pax7+, vimentin+, nestin+ and neonatal myosin+ muscle fibers showed the highest amount of regenerating muscle fibers in inflammatory myopathies, followed by muscular dystrophy. The relative co-expression of ATOH8 with the above-mentioned markers did not vary among the disorders. These results show that the novel regeneration marker ATOH8 contributes to muscle cell differentiation in healthy and diseased human muscle tissue.





Similar content being viewed by others
Abbreviations
- DAPI:
-
4′-6-Diamidino-2-phenylindole
- DM:
-
Dermatomyositis
- dsRNA:
-
Double-stranded ribonucleic acid
- LGMD:
-
Limb-girdle muscular dystrophy
- MD:
-
Muscular dystrophy
- MV:
-
Mean value
- PCR :
-
Polymerase chain reaction
- PM:
-
Polymyositis
- SEM:
-
Standard error of the mean
- IBM:
-
Inclusion body myositis
References
Amato AA, Barohn RJ (1997) Idiopathic inflammatory myopathies. Neurol Clin 15:615–648
Asakura A, Seale P, Girgis-Gabardo A, Rudnicki MA (2002) Myogenic specification of side population cells in skeletal muscle. J Cell Biol 159:123–134. doi:10.1083/jcb.200202092
Askanas V, Engel WK, Nogalska A (2009) Inclusion body myositis: a degenerative muscle disease associated with intra-muscle fiber multi-protein aggregates, proteasome inhibition, endoplasmic reticulum stress and decreased lysosomal degradation. Brain Pathol 19:493–506. doi:10.1111/j.1750-3639.2009.00290.x
Balakrishnan-Renuka A, Morosan-Puopolo G, Yusuf F et al (2014) ATOH8, a regulator of skeletal myogenesis in the hypaxial myotome of the trunk. Histochem Cell Biol 141:289–300. doi:10.1007/s00418-013-1155-0
Chargé SBP, Rudnicki MA (2004) Cellular and molecular regulation of muscle regeneration. Physiol Rev 84:209–238. doi:10.1152/physrev.00019.2003
Chen J, Dai F, Balakrishnan-Renuka A et al (2011) Diversification and molecular evolution of ATOH8, a gene encoding a bHLH transcription factor. PLoS One 6:e23005. doi:10.1371/journal.pone.0023005
Cornelison DD, Olwin BB, Rudnicki MA, Wold BJ (2000) MyoD(−/−) satellite cells in single-fiber culture are differentiation defective and MRF4 deficient. Dev Biol 224:122–137. doi:10.1006/dbio.2000.9682
Dalakas MC (2006) Mechanisms of disease: signaling pathways and immunobiology of inflammatory myopathies. Nat Clin Pract Rheumatol 2:219–227. doi:10.1038/ncprheum0140
De Paepe B, Creus KK, De Bleecker JL (2008) Chemokines in idiopathic inflammatory myopathies. Front Biosci 13:2548–2577
Dhawan J, Rando TA (2005) Stem cells in postnatal myogenesis: molecular mechanisms of satellite cell quiescence, activation and replenishment. Trends Cell Biol 15:666–673. doi:10.1016/j.tcb.2005.10.007
Dimachkie MM (2011) Idiopathic inflammatory myopathies. J Neuroimmunol 231:32–42. doi:10.1016/j.jneuroim.2010.10.013
Ejarque M, Altirriba J, Gomis R, Gasa R (2013) Characterization of the transcriptional activity of the basic helix–loop–helix (bHLH) transcription factor Atoh8. Biochim Biophys Acta 1829:1175–1183. doi:10.1016/j.bbagrm.2013.08.003
Hawke TJ, Garry DJ (2001) Myogenic satellite cells: physiology to molecular biology. J Appl Physiol 91:534–551
Kerst B, Mennerich D, Schuelke M et al (2000) Heterozygous myogenic factor 6 mutation associated with myopathy and severe course of Becker muscular dystrophy. Neuromuscul Disord 10:572–577
Mauro A (1961) Satellite cell of skeletal muscle fibers. J Biophys Biochem Cytol 9:493–495
Muntoni F, Brown S, Sewry C, Patel K (2002) Muscle development genes: their relevance in neuromuscular disorders. Neuromuscul Disord 12:438–446
Onofre-Oliveira PCG, Santos ALF, Martins PM et al (2012) Differential expression of genes involved in the degeneration and regeneration pathways in mouse models for muscular dystrophies. Neuromol Med 14:74–83. doi:10.1007/s12017-012-8172-3
Pinkus JL, Amato AA, Taylor JP, Greenberg SA (2014) Abnormal distribution of heterogeneous nuclear ribonucleoproteins in sporadic inclusion body myositis. Neuromuscul Disord 24:611–616. doi:10.1016/j.nmd.2014.04.012
Rahimov F, Kunkel LM (2013) The cell biology of disease: cellular and molecular mechanisms underlying muscular dystrophy. J Cell Biol 201:499–510. doi:10.1083/jcb.201212142
Schmidt J, Rakocevic G, Raju R, Dalakas MC (2004) Upregulated inducible co-stimulator (ICOS) and ICOS-ligand in inclusion body myositis muscle: significance for CD8+ T cell cytotoxicity. Brain 127:1182–1190. doi:10.1093/brain/awh148
Seale P, Sabourin LA, Girgis-Gabardo A et al (2000) Pax7 is required for the specification of myogenic satellite cells. Cell 102:777–786
Tajbakhsh S (2009) Skeletal muscle stem cells in developmental versus regenerative myogenesis. J Intern Med 266:372–389. doi:10.1111/j.1365-2796.2009.02158.x
Vaittinen S, Lukka R, Sahlgren C et al (2001) The expression of intermediate filament protein nestin as related to vimentin and desmin in regenerating skeletal muscle. J Neuropathol Exp Neurol 60:588–597
Wang Y, Chen K, Yao Q et al (2009) Phylogenetic analysis of zebrafish basic helix–loop–helix transcription factors. J Mol Evol 68:629–640. doi:10.1007/s00239-009-9232-7
Wanschitz JV, Dubourg O, Lacene E et al (2013) Expression of myogenic regulatory factors and myo-endothelial remodeling in sporadic inclusion body myositis. Neuromuscul Disord 23:75–83. doi:10.1016/j.nmd.2012.09.003
Weise C, Dai F, Pröls F et al (2006) Myogenin (Myf4) upregulation in trans-differentiating fibroblasts from a congenital myopathy with arrest of myogenesis and defects of myotube formation. Anat Embryol (Berl) 211:639–648. doi:10.1007/s00429-006-0117-x
Winter A, Bornemann A (1999) NCAM, vimentin and neonatal myosin heavy chain expression in human muscle diseases. Neuropathol Appl Neurobiol 25:417–424
Zammit PS, Heslop L, Hudon V et al (2002) Kinetics of myoblast proliferation show that resident satellite cells are competent to fully regenerate skeletal muscle fibers. Exp Cell Res 281:39–49
Acknowledgments
Beate Brand-Saberi was supported by the MYORES Project (511978) funded by the EU’s Sixth Framework Program, FoRUM F647-09 and F732N-2011 of the Faculty of Medicine, Ruhr University Bochum and MERCUR PR 2012-0058. Anne-K. Güttsches was supported by FoRUM K069-12 of the Faculty of Medicine, Ruhr University Bochum. We sincerely thank J. Mertens-Rill and A. Schreiner for excellent technical support.
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Güttsches, AK., Balakrishnan-Renuka, A., Kley, R.A. et al. ATOH8: a novel marker in human muscle fiber regeneration. Histochem Cell Biol 143, 443–452 (2015). https://doi.org/10.1007/s00418-014-1299-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-014-1299-6