Abstract
Purpose
The purpose of our research is to evaluate the mechanism of PD-1 in the promotion of HBV transmission.
Methods
HBV was used to infect two human choriocarcinoma cell line, including JEG-3, as well as BeWo. We used PCR and western blotting to detect PD-1 gene and protein expression levels in cells. Stable knockdown of the PD-1 gene in JEG-3 cells was obtained by lentiviral transfection. Trophoblast cell proliferation was evaluated using CCK8 and flow cytometry. The concentration of HBV antibody in the cell supernatant was measured by ELISA. DNA was then extracted from the cells and the copy number of the HBV virus was detected by PCR. Finally, ERK1/2 expression was detected by western blot.
Results
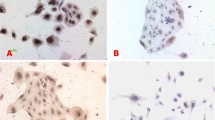
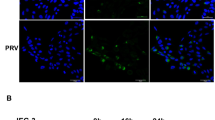
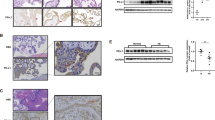
High PD-1 gene expression in HBV-infected trophoblasts and the knockdown of PD-1 gene can, respectively, improve the proliferation of HBV-infected trophoblasts and reduce viral replication in trophoblasts. In addition, PD-1 and ERK1/2 proteins were co-expressed in HBV-infected trophoblasts and inhibited the activation of ERK1/2 pathway in HBV-infected trophoblasts. ERK1/2 expression significantly increased after PD-1 knockdown. Therefore, PD-1 might be an important protein in trophoblast cells infected with HBV.
Conclusions
PD-1 promoted HBV transmission through regulating ERK1/2-mediated trophoblasts differentiation. Therefore, our research may provide new ideas and methods for preventing mother-to-child transmission of HBV infection during pregnancy.




Similar content being viewed by others
References
Trépo C, Chan HL, Lok A (2014) Hepatitis B virus infection. Lancet 384:2053–2063
Busch K, Thimme R (2015) Natural history of chronic hepatitis B virus infection. Med Microbiol Immunol 204:5–10
Ling Z, Liu X, Cheng Y, Jiang X, Jiang H, Wang Y et al (2015) Decreased diversity of the oral microbiota of patients with hepatitis B virus-induced chronic liver disease: a pilot project. Sci Rep 5:17098
Yin Y, Wu L, Zhang J, Zhou J, Zhang P, Hou H (2013) Identification of risk factors associated with immunoprophylaxis failure to prevent the vertical transmission of hepatitis B virus. J Infect 66:447–452
Chen Y, Wang L, Xu Y, Liu X, Li S, Qian Q et al (2013) Role of maternal viremia and placental infection in hepatitis B virus intrauterine transmission. Microbes Infect 15:409–415
Delorme-Axford E, Donker RB, Mouillet JF, Chu T, Bayer A, Ouyang Y et al (2013) Human placental trophoblasts confer viral resistance to recipient cells. Proc Natl Acad Sci USA 110:12048–12053
Xu DZ, Yan YP, Zou S, Choi BC, Wang S, Liu P et al (2001) Role of placental tissues in the intrauterine transmission of hepatitis B virus. Am J Obstet Gynecol 185:981–987
Bhat P, Anderson DA (2007) Hepatitis B virus translocates across a trophoblastic barrier. J Virol 81:7200–7207
Zhang X, Schwartz JC, Guo X, Bhatia S, Cao E, Lorenz M et al (2004) Structural and functional analysis of the costimulatory receptor programmed death-1. Immunity 20:337–347
Li M, Sun XH, Zhu XJ, Jin SG, Zeng ZJ, Zhou ZH et al (2012) HBcAg induces PD-1 upregulation on CD4 + T cells through activation of JNK, ERK and PI3K/AKT pathways in chronic hepatitis-B-infected patients. Lab Invest 92:295–304
Darmochwal-Kolarz D, Kludka-Sternik M, Kolarz B, Chmielewski T, Tabarkiewicz J, Rolinski J et al (2013) The expression of B7-H1 and B7-H4 co-stimulatory molecules on myeloid and plasmacytoid dendritic cells in pre-eclampsia and normal pregnancy. J Reprod Immunol 99:33–38
Evans A, Riva A, Cooksley H, Phillips S, Puranik S, Nathwani A et al (2008) Programmed death 1 expression during antiviral treatment of chronic hepatitis B: impact of hepatitis B e-antigen seroconversion. Hepatology 48:759–769
Fisicaro P, Valdatta C, Massari M, Loggi E, Biasini E, Sacchelli L et al (2010) Antiviral intrahepatic T-cell responses can be restored by blocking programmed death-1 pathway in chronic hepatitis B. Gastroenterology 138(682–693):693.e1–693.e4
Das D, Sengupta I, Sarkar N, Pal A, Saha D, Bandopadhyay M et al (2017) Anti-hepatitis B virus (HBV) response of imiquimod based toll like receptor 7 ligand in hbv-positive human hepatocelluar carcinoma cell line. BMC Infect Dis 17:76
Pötgens AJ, Bolte M, Huppertz B, Kaufmann P, Frank HG (2001) Human trophoblast contains an intracellular protein reactive with an antibody against CD133—a novel marker for trophoblast. Placenta 22:639–645
Cui H, Chen J, Na Q (2016) Effect of hepatitis B virus infection on trophoblast cell line (HTR-8/SVneo) and choriocarcinoma cell line (JEG3) is linked to CD133-2 (AC141) expression. Am J Transl Res 8:3235–3240
Tian T, Sun D, Wang P, Wang H, Bai X, Yang X et al (2015) Roles of toll-like receptor 7 and 8 in prevention of intrauterine transmission of hepatitis B virus. Cell Physiol Biochem 37:445–453
Klaffenbach D, Rascher W, Röllinghoff M, Dötsch J, Meissner U, Schnare M (2005) Regulation and signal transduction of toll-like receptors in human chorioncarcinoma cell lines. Am J Reprod Immunol 53:77–84
Ding Y, Ma L, Wang XZ, Zhang J, Zhao GZ, Wang ZQ et al (2011) In vitro study on hepatitis B virus infecting human choriocarcinoma JEG3 cells and its mechanism. Intervirology 54:276–281
Boni C, Fisicaro P, Valdatta C, Amadei B, Di VP, Giuberti T et al (2007) Characterization of hepatitis B virus (HBV)-specific T-cell dysfunction in chronic HBV infection. J Virol 81:4215–4225
Boettler T, Panther E, Bengsch B, Nazarova N, Spangenberg HC, Blum HE et al (2006) Expression of the interleukin-7 receptor alpha chain (CD127) on virus-specific CD8 + T cells identifies functionally and phenotypically defined memory T cells during acute resolving hepatitis B virus infection. J Virol 80:3532–3540
Otano I, Escors D, Schurich A, Singh H, Robertson F, Davidson BR et al (2018) Molecular recalibration of PD-1 + antigen-specific T cells from blood and liver. Mol Ther 26:2553–2566
Kleffel S, Posch C, Barthel SR, Mueller H, Schlapbach C, Guenova E et al (2015) Melanoma cell-intrinsic PD-1 receptor functions promote tumor growth. Cell 162:1242–1256
Okazaki T, Honjo T (2006) The PD-1-PD-L pathway in immunological tolerance. Trends Immunol 27:195–201
Maier H, Isogawa M, Freeman GJ, Chisari FV (2007) PD-1:PD-L1 interactions contribute to the functional suppression of virus-specific CD8 + T lymphocytes in the liver. J Immunol 178:2714–2720
Dai S, Jia R, Zhang X, Fang Q, Huang L (2014) The PD-1/PD-Ls pathway and autoimmune diseases. Cell Immunol 290:72–79
Cho H, Kikuchi M, Li Y, Nakamoto N, Amorosa VK, Valiga ME et al (2014) Induction of multiple immune regulatory pathways with differential impact in HCV/HIV coinfection. Front Immunol 5:265
Noh H, Hu J, Wang X, Xia X, Satelli A, Li S (2015) Immune checkpoint regulator PD-L1 expression on tumor cells by contacting CD11b positive bone marrow derived stromal cells. Cell Commun Signal 13:14
Chen J, Ji T, Zhao J, Li G, Zhang J, Jin R et al (2016) Sorafenib-resistant hepatocellular carcinoma stratified by phosphorylated ERK activates PD-1 immune checkpoint. Oncotarget 7:41274–41284
Chen N, Fang W, Lin Z, Peng P, Wang J, Zhan J et al (2017) KRAS mutation-induced upregulation of PD-L1 mediates immune escape in human lung adenocarcinoma. Cancer Immunol Immunother 66:1175–1187
Funding
The study was not supported by any funding.
Author information
Authors and Affiliations
Contributions
JY, JL, ML: project development, data collection, manuscript writing. MS, XZ: data collection.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Human or animal studies are not covered in this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, J., Liu, J., Sheng, M. et al. Programmed cell death protein 1 promotes hepatitis B virus transmission through the regulation of ERK1/2-mediated trophoblasts differentiation. Arch Gynecol Obstet 301, 551–558 (2020). https://doi.org/10.1007/s00404-019-05401-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-019-05401-8