Abstract
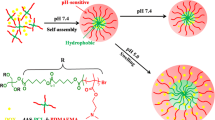
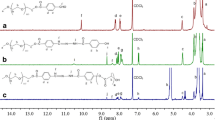
Biodegradable micelles based on disulfide-linked monomethoxy poly(ethylene glycol)-b-poly(methyl methacrylate)2 (mPEG-SS-PMMA2) miktoarm star block copolymers were synthesized through atom transfer radical polymerization procedure and applied to solubilization and delivery of methotrexate (MTX). The synthesized miktoarm star block copolymers were characterized by 1H nuclear magnetic resonance, Fourier transform infrared spectra, and gel permeation chromatography. The block copolymers were able to self-assemble into spherical micelles in water with an average diameter up to 130 nm by transmission electron microscopy observation and dynamic light scattering measurements and have a critical micelle concentration of 0.91 mg/L. When MTX was incorporated into obtained micelles, high drug loading capacity was found attributed to the designed miktoarm star-shaped nanostructure. Interestingly, in presence of dithiothreitol, the micelles could be degraded into polymer chains by cleavage of the disulfide linkages. The in vitro release studies revealed that these micelles released over 95 % of MTX within 48 h under a reductive environment analogous to that of the cell comparing to minimal drug release (<22 %) under nonreductive conditions. Cell cytoxicity experiments showed that the obtained micelles exhibited nontoxic, and the drug-loaded micelles exhibited high anticancer efficacy and better biocompatibility as compared to free MTX. All of these results showed that mPEG-SS-PMMA2 micelles are promising carriers for delivery of hydrophobic antitumor drugs.










Similar content being viewed by others
References
Scarano W, Duong HTT, Lu H, De Souza PL, Stenzel MH (2013) Folate conjugation to polymeric micelles via boronic acid ester to deliver platinum drugs to ovarian cancer cell lines. Biomacromolecules 14:962–975
Liu J, Pang Y, Huang W, Huang X, Meng L, Zhu X, Zhou Y, Yan D (2011) Bioreducible micelles self-assembled from amphiphilic hyperbranched multiarm copolymer for glutathione-mediated intracellular drug delivery. Biomacromolecules 12:1567–1577
Laouini A, Koutroumanis KP, Charcosset C, Georgiadou S, Fessi H, Holdich RG, Vladisavljević GT (2013) pH-Sensitive micelles for targeted drug delivery prepared using a novel membrane contactor method. ACS Appl Mater Inter 5:8939–8947
Zhou Q, Lin J, Wang J, Li F, Tang F, Zhao X (2009) A designed amphiphilic peptide containing the silk fibroin motif as a potential carrier of hydrophobic drugs. Prog Nat Sci 19:1529–1536
Jin X, Mo R, Ding Y, Zheng W, Zhang C (2014) Paclitaxel-loaded n-octyl-o-sulfate chitosan micelles for superior cancer therapeutic efficacy and overcoming drug resistance. Mol Pharm 11:145–157
Zhang L, Eisenberg A (1995) Multiple morphologies of “Crew-Cut” aggregates of polystyrene-b-poly (acrylic acid) block copolymers. Science 268:1728–1731
Zhang L, Yu K, Eisenberg A (1996) Ion-induced morphological changes in “Crew-Cut” aggregates of amphiphilic block copolymers. Science 272:1777–1779
Torchilin VP (2006) Multifunctional nanocarriers. Adv Drug Delivery Rev 58:1532–1555
Kwon GS, Kataoka K (1995) Block copolymer micelles as long-circulating drug vehicles. Drug Delivery Rev 16:295–309
Kataoka K, Harada A, Nagasaki Y (2001) Block copolymer micelles for drug delivery: design, characterization and biological significance. Adv Drug Delivery Rev 47:113–131
Lin J, Luo J-B, Yang S-T, Zhou Q-H (2013) Template-directed self-assembly of a designed amphiphilic hexapeptide on mica surface. Colloid Polym Sci 291:2263–2270
Yang J, Yan J, Zhou Z, Amsden BG (2014) Dithiol-PEG-PDLLA micelles: preparation and evaluation as potential topical ocular delivery vehicle. Biomacromolecules 15:1346–1354
Ma R, Yang H, Li Z, Liu G, Sun X, Liu X, An Y, Shi L (2012) Phenylboronic acid-based complex micelles with enhanced glucose-responsiveness at physiological ph by complexation with glycopolymer. Biomacromolecules 13:3409–3417
Wang F, Bronich TK, Kabanov AV, Rauh RD, Roovers J (2005) Synthesis and evaluation of a star amphiphilic block copolymer from poly (ε-caprolactone) and poly (ethylene glycol) as a potential drug delivery carrier. Bioconjugate Chem 16:397–405
Wang F, Bronich TK, Kabanov AV, Rauh RD, Roovers J (2008) Synthesis and characterization of star poly (ε-caprolactone)-b-poly (ethylene glycol) and poly (l-lactide)-b-poly (ethylene glycol) copolymers: evaluation as drug delivery carriers. Bioconjugate Chem 19:1423–1429
Pavlov GM, Knop K, Okatova OV, Schubert US (2013) Star-brush-shaped macromolecules: peculiar properties in dilute solution. Macromolecules 46:8671–8679
Rao J, Zhang Y, Zhang J, Liu S (2008) Facile preparation of well-defined AB2 Y-shaped miktoarm star polypeptide copolymer via the combination of ring-opening polymerization and click chemistry. Biomacromolecules 9:2586–2593
Cao W, Zhou J, Mann A, Wang Y, Zhu L (2011) Folate-functionalized unimolecular micelles based on a degradable amphiphilic dendrimer-like star polymer for cancer cell-targeted drug delivery. Biomacromolecules 12:2697–2707
Poree DE, Giles MD, Lawson LB, He J, Grayson SM (2011) Synthesis of amphiphilic star block copolymers and their evaluation as transdermal carriers. Biomacromolecules 12:898–906
Ni C, Wu G, Zhu C, Yao B (2010) The Preparation and characterization of amphiphilic star block copolymer nano micelles using silsesquioxane as the core. J Phys Chem C 114:13471–13476
Lin J, Zhou Q-H, L-d L, Li Z-N (2014) Synthesis and self-assembly in bulk of star-shaped block copolymers based on helical polypeptides. Colloid Polym Sci 292:3177–3185
Junnila S, Houbenov N, Karatzas A, Hadjichristidis N, Hirao A, Iatrou H, Ikkala O (2012) Side-chain-controlled self-assembly of polystyrene–polypeptide miktoarm star copolymers. Macromolecules 45:2850–2856
Ostermann J, Merkl J-P, Flessau S, Wolter C, Kornowksi A, Schmidtke C, Pietsch A, Kloust H, Feld A, Weller H (2013) Controlling the physical and biological properties of highly fluorescent aqueous quantum dots using block copolymers of different size and shape. ACS Nano 7:9156–9167
Ye C, Zhao G, Zhang M, Du J, Zhao Y (2012) Precise synthesis of ABCDE star quintopolymers by combination of controlled polymerization and azide–alkyne cycloaddition reaction. Macromolecules 45:7429–7439
Liu H, Li S, Zhang M, Shao W, Zhao Y (2012) Facile synthesis of ABCDE-type H-shaped quintopolymers by combination of ATRP, ROP, and click chemistry and their potential applications as drug carriers. J Polym Sci A-Polym Chem 50:4705–4716
McRae Page S, Martorella M, Parelkar S, Kosif I, Emrick T (2013) Disulfide cross-linked phosphorylcholine micelles for triggered release of camptothecin. Mol Pharm 10:2684–2692
Sun H, Guo B, Cheng R, Meng F, Liu H, Zhong Z (2009) Biodegradable micelles with sheddable poly (ethylene glycol) shells for triggered intracellular release of doxorubicin. Biomaterials 30:6358–6366
Sun Y, Huang Y, Bian S, Liang J, Fan Y, Zhang X (2013) Reduction-degradable PEG-b–PAA-b–PEG triblock copolymer micelles incorporated with MTX for cancer chemotherapy. Colloids Surf B 112:197–203
Zhang Y, Jin T, Zhuo R-X (2005) Methotrexate-loaded biodegradable polymeric micelles: preparation, physicochemical properties and in vitro drug release. Colloids Surf B Biointerfaces 44:104–109
Chassepot A, Gao L, Nguyen I, Dochter A, Fioretti F, Menu P, Kerdjoudj H, Baehr C, Schaaf P, Voegel J-C (2012) Chemically detachable polyelectrolyte multilayer platform for cell sheet engineering. Chem Mater 24:930–937
Sui X, Zhang Z, Guan S, Xu Y, Li C, Lv Y, Chen A, Yang L, Gao L (2015) A facile strategy for the synthesis of block copolymers bearing an acid-cleavable junction. Polym Chem 6:2777–2782
Jäger M, Wilke A (2003) Comprehensive biocompatibility testing of a new PMMA-HA bone cement versus conventional PMMA cement in vitro. J Biomater Sci Polymer Edn 14:1283–1298
Ding X, Liu Y, Li J, Luo Z, Hu Y, Zhang B, Liu J, Zhou J, Cai K (2014) Hydrazone-bearing PMMA-functionalized magnetic nanocubes as pH-responsive drug carriers for remotely targeted cancer therapy in vitro and in vivo. ACS Appl Mater Interfaces 6:7395–7407
Streubel A, Siepmann J, Bodmeier R (2003) Multiple unit gastroretentive drug delivery systems: a new preparation method for low density microparticles. J Microencapsul 20:329–347
Sparnacci K, Laus M, Tondelli L, Bernardi C, Magnani L, Corticelli F, Marchisio M, Ensoli B, Castaldello A, Caputo A (2005) Core–shell microspheres by dispersion polymerization as promising delivery systems for proteins. J Biomater Sci Polym Ed 16:1557–1574
Zhou Q-H, Zheng J-K, Shen Z, Fan X-H, Chen X-F, Zhou Q-F (2010) Synthesis and hierarchical self-assembly of rod-rod block copolymers via click chemistry between mesogen-jacketed liquid crystalline polymers and helical polypeptides. Macromolecules 43:5637–5646
Matyjaszewski K, Miller PJ, Pyun J, Kickelbick G, Diamanti S (1999) Synthesis and characterization of star polymers with varying arm number, length, and composition from organic and hybrid inorganic/organic multifunctional initiators. Macromolecules 32:6526–6535
Hanson JA, Li Z, Deming TJ (2010) Nonionic block copolypeptide micelles containing a hydrophobic rac-leucine core. Macromolecules 43:6268–6269
Ren T-B, Xia W-J, Dong H-Q, Li Y-Y (2011) Sheddable micelles based on disulfide-linked hybrid PEG-polypeptide copolymer for intracellular drug delivery. Polymer 52:3580–3586
Hu Y, Zhang L, Cao Y, Ge H, Jiang X, Yang C (2004) Degradation behavior of poly (ε-caprolactone)-b-poly (ethylene glycol)-b-poly (ε-caprolactone) micelles in aqueous solution. Biomacromolecules 5:1756–1762
Kang H, Kim J-D, Han S-H, Chang I-S (2002) Self-aggregates of poly (2-hydroxyethyl aspartamide) copolymers loaded with methotrexate by physical and chemical entrapments. J Control Release 81:135–144
Ashwanikumar N, Kumar NA, Nair SA, Kumar GSV (2014) Dual drug delivery of 5-fluorouracil (5-FU) and methotrexate (MTX) through random copolymeric nanomicelles of PLGA and polyethylenimine demonstrating enhanced cell uptake and cytotoxicity. Colloids Surf B Biointerfaces 122:520–528
Baranello MP, Bauer L, Benoit DSW (2014) Poly (styrene-alt-maleic anhydride)-based diblock copolymer micelles exhibit versatile hydrophobic drug loading, drug-dependent release, and internalization by multidrug resistant ovarian cancer cells. Biomacromolecules 15:2629–2641
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Grant Nos. 21404086) and the Project of Postgraduate Degree Construction, Southwest University for Nationalities (No. 2015XWD-S0703).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhou, QH., Lin, J., Li, LD. et al. Biodegradable micelles self-assembled from miktoarm star block copolymers for MTX delivery. Colloid Polym Sci 293, 2291–2300 (2015). https://doi.org/10.1007/s00396-015-3610-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-015-3610-z