Abstract
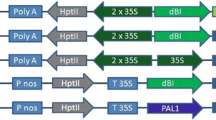
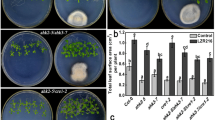
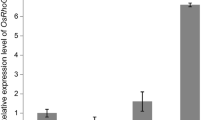
To study plant roots in response to ipt gene activation, the transcriptional fusions of ipt-GUS and GUS-ipt were expressed in roots or in whole plants of Arabidopsis under the control of a root-specific promoter TobRT7 or a CaMV35S promoter through the pOp/LhG4 system in noninvasive conditions. The transgenic plants with constitutive expression of ipt-GUS or GUS-ipt showed 15–25-fold or 1–2-fold increased cytokinin levels, respectively. ipt-GUS-expressing Arabidopsis had severe root inhibition, enlarged shoot apical parts, serrated leaves, and no or few sterile flowers, whereas GUS-ipt-expressing Arabidopsis grew faster, flowered early, and had more lateral shoots. However, when ipt-GUS and GUS-ipt were specially expressed in roots under the control of TobRT7, neither cytokinin content in roots or shoots nor phenotypes were altered. In cytokinin-overproducing, ipt-GUS-expressing Arabidopsis, the light and aerial parts of plants played an important role for cytokinin synthesis and root inhibition, and the ipt gene was vigorously expressed at the shoot apical parts. Meanwhile, calli were induced at the shoot apical parts of some cytokinin-overproducing, ipt-GUS-expressing Arabidopsis.




Similar content being viewed by others
Abbreviations
- ipt :
-
Isopentenyl transferase gene
- GUS :
-
β-1,3-glucuronidase gene
- cZ:
-
cis-zeatin
- cZR:
-
cis-zeatin riboside
- cZRP:
-
cis-zeatin riboside phosphate
- DHZ:
-
Dihydrozeatin
- DH Z7G:
-
Dihydrozeatin-7-glucoside
- DHZR:
-
Dihydrozeatin riboside
- DHZRP:
-
Dihydrozeatin riboside phosphate
- DHZROG:
-
Dihydrozeatin riboside O-glucoside
- DH Z9G:
-
Dihydrozeatin-9-glucoside
- iP:
-
N 6-Isopentenyladenine
- iP7G:
-
Isopentenyl adenine-7-glucoside
- iPR:
-
Isopentenyl riboside
- iPRP:
-
Isopentenyl riboside phosphate
- Z:
-
Zeatin
- Z7G:
-
Zeatin-7-glucoside
- Z9G:
-
Zeatin-9-glucoside
- ZOG:
-
Zeatin-O-glucoside
- ZOX:
-
Zeatin oxidase
- ZR:
-
Zeatin riboside
- ZROG:
-
Zeatin riboside O-glucoside
- ZRP:
-
Zeatin riboside phosphate
References
Akiyoshi DE, Klee H, Amasino RM, Nester EW, Gordon MP (1984) T-DNA of Agrobacterium tumefaciens encodes an enzyme of cytokinin biosynthesis. Proc Natl Acad Sci USA 81:5994–5998
Barry GF, Rogers SG, Fraley RT, Brand L (1984) Identification of a cloned cytokinin biosynthetic gene. Proc Natl Acad Sci USA 81:4776–4780
Beisberger SEI, Valcke RLM, Deblaere RY, Clijsters HMM, de Greef JA, van Onckelen HA (1991) Effects of the introduction of Agrobacterium tumefaciens T-DNA ipt gene in Nicotiana tabacum L. cv. Petit Havana SR1 plant cells. Plant Cell Physiol 32:489–496
Bevan M (1984) Binary Agrobacterium vectors for plant transformation. Nucleic Acids Res 12:8711–8721
Chibnall AN (1939) Protein metabolism in plants. Yale University Press, New Haven, CT
Faiss M, Zalubilová J, Strnad M, Schmülling T (1997) Conditional transgenic expression of the ipt gene indicates a functional for cytokinins in paracrine signaling in whole tobacco plants. Plant J 12:401–415
Flores S, Tobin EM (1986) Benzyladenine modulation of the expression of two genes for nuclear-encoded chloroplast proteins in Lemna gibba: apparent post-transcriptional regulation. Planta 168:340–349
Gan S, Amasino RM (1995) Inhibition of leaf senescence by auto-regulated production of cytokinin. Science 270:1986–1988
Geng S, Ma M, Ye HC, Liu BY, Li GF, Chong K (2001) Effects of ipt gene expression on the physiological and chemical characteristics of Artemisia annua L. Plant Sci 160:691–698
Geng S, Ma M, Ye HC, Li GF (2002) Anther-specific expression of ipt gene in transgenic tobacco and its effect on plant development. Transgenic Res 11(3):269–278
Guo JC, Hu XW, Duan RJ (2005) Interactive effects of cytokinins, light and sucrose on the phenotypes and the syntheses of anthocyanins, lignins in cytokinin over-producing transgenic Arabidopsis. J Plant Growth Regul 24:93–101
Hewelt A, Prinsen E, Schell J, Van Onckelen H, Schmülling T (1994) Promoter tagging with a promoterless ipt gene leads to cytokinin-induced phenotypic variability in transgenic tobacco plants: implication of gene dosage effects. Plant J 6:879–891
Kozak M (1978) How do eucaryotic ribosomes select initiation regions in messenger RNA? Cell 15:1109–1123
Letham DS (1994) Cytokinin as phytohormones—sites of biosynthesis, translocation, and function of translocated cytokinins. In: Mok DWS, MOK MC (eds) Cytokinins: chemistry, activity and function. CRC Press, Boca Raton, FL, pp 57–80
Li Y, Hagen G, Guilfoyle TJ (1992) Altered morphology in transgenic tobacco plants that overproduce cytokinins in specific tissues and organs. Dev Biol 153:386–395
Martineau B, Houck CM, Sheehy RE, Hiatt WR (1994) Fruit-specific expression of the A. tumefaciens isopentenyl transferase gene in tomato: effects on fruit ripening and defense-related gene expression in leaves. Plant J 5:11–19
Mckenzie MJ, Mett V, Reynolds PHS, Jameson PE (1998) Controlled cytokinin production in transgenic tobacco using a copper-inducible promoter. Plant Physiol 116:969–977
Moore I, Gälweiler L, Grosskopf D, Schell J, Palme K (1998) A transcription activation system for regulated gene expression in transgenic plant. Proc Natl Acad Sci USA 95:376–381
Mothes K, Engelbrech L (1962) A revised medium for the rapid growth and bioassay with tobacco tissue culture. Plant Physiol 15:473–496
Mustilli AC, Fenzi F, Ciliento R, Alfano F, Bowler C (1999) Phenotype of the tomato high pigment-2 mutant is caused by a mutation in the tomato homolog of DEETIOLATED1. Plant Cell 11:145–157
Peabody DS, Subramani S, Berg P (1986) Effect of upstream reading frames on translated efficiency in Simian Virus 40 recombinants. Mol Cell Biol 6:2704–2711
Redig P, Schmülling T, van Onckelen H (1996) Analysis of cytokinin metabolism in ipt transgenic tobacco by liquid chromatography-tandem mass spectrometry. Plant Physiol 122:141–148
Richmond AE, Lang A (1957) Effect of kinetin on the protein content and survival of detached Xanthium leaf. Science 125:650–651
Rogers SG, Fraley RT, Horsch RB, Levine AD, Flick JS, Brand LA, Fink CL, Moor T, O’Connell K, Sanders PR (1985) Evidence for ribosome scanning during translation initiation of mRNAs in transformed plant cells. Plant Mol Biol Rep 3:111–116
Rupp HM, Frank M, Werner T, Stand M, Schmülling T (1999) Increased steady state mRNA levels of the STM and KNAT1 homeobox genes in cytokinin over-producing Arabidopsis thaliana indicate a role for cytokinin in the shoot apical meristem. Plant J 18(5):557–563
Schmülling T, Beinsberger S, de Greef J, Schell J, van Onckelen H, Spena A (1998) Construction of a heat-inducible chimeric gene to increase the cytokinin content in transgenic plant tissue. FEBS Lett 249:401–406
Singh S, Letham DS, Palni LMS (1992a) Cytokinin biochemistry in relation to leaf senescence. J Plant Physiol 139:279–283
Singh S, Letham DS, Palni LMS (1992b) Cytokinin biochemistry in relation to leaf senescence. J Plant Physiol 86:398–406
Smigocki AC (1991) Cytokinin content and tissue distribution in plants transformed by a reconstructed isopentenyl transferase gene. Plant Mol Biol 16:105–115
Smigocki AC (1995) Expression of a wound-inducible cytokinin biosynthesis gene in transgenic tobacco: correlation of root expression with induction of cytokinin effects. Plant Sci 109:153–163
Smigocki AC, Owens LD (1989) Cytokinin-to-auxin ratios and morphology of shoots and tissues transformed by a chimeric isopentenyl transferase gene. Plant Physiol 91:808–811
Smigocki AC, Neal JW, McCanna I, Douglass L (1993) Cytokinin-mediated insect resistance in Nicotiana plants transformed with the ipt gene. Plant Mol Biol 23:325–335
van der Graaff EE, Auer CA, Hooykaas PJJ (2001) Altered development of Arabidopsis thaliana carrying the Agrobacterium tumefaciens ipt gene partially due to ethylene effects. Plant Grow Regul 34:305–315
van Duijn LP, Holsappel S, Kasperaitis M, Bunschoten H, Konings D, Voorma HO (1988) Secondary structure and expression in vivo and in vitro of messenger RNA into which upstream AUG codon have been inserted. Eur J Biochem 172:59–66
van Loven K, Beinsberger SEL, Valcke RLM, van Onckelen HA, Clijsters HMM (1993) Morphometric analysis of the growth of phsp7-ipt transgenic tobacco plants as a model for the investigation of plant gene expression in response to phytohormonal stress. Plant Mol Biol 17:825–836
Wang J, Letham DS, Cornish E, Stevenson KR (1997a) Studies of cytokinin action and metabolism using tobacco plants expressing either the ipt or the GUS gene controlled by a chalcone synthase promoter. I. developmental features of the transgenic plants. Aust J Plant Physiol 24:661–672
Wang J, Letham DS, Cornish E, Wei K, Hocart CH, Michael M, Stevenson KR (1997b) Studies of cytokinin action and metabolism using tobacco plants expressing either the ipt or the GUS gene controlled by a chalcone synthase promoter. II. ipt and GUS gene expression, cytokinin levels and metabolism. Aust J Plant Physiol 24:673–683
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No 30360008), 973 Pre-Program of China (No 2006CB708204), and National Basic Research Program of China 2007CB108903.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, J., Hu, X. Noninvasive Expressions of ipt in Whole Plants or Roots through pOp/LhG4 Indicate a Role of Plant Aerial Parts and Light in Cytokinin Synthesis and Root Inhibition. J Plant Growth Regul 27, 251–262 (2008). https://doi.org/10.1007/s00344-008-9052-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-008-9052-9