Abstract
Key message
CsGPA1 interacts with CsTIP1.1 (a member of CsAQPs) and suppression of CsGPA1 results the reverse expression of CsAQPs in leaves and roots, resulting in declining water content of cucumber seedlings under salt stress.
Abstract
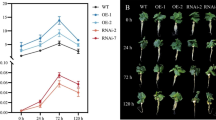
Salt stress seriously affects cucumber growth and development. Whether the G-protein alpha subunit functions in cucumber during salt stress and its regulation mechanism remains unknown. We interrogated CsGPA1-RNAi lines to identify the role of CsGPA1 during salt stress. Phenotypically, compared with wild type, leaves were severely withered, and root cells showed signs of senescence under salt stress for RNAi lines. Compared with WT, SOD and CAT activity, soluble protein and proline contents all decreased in RNAi lines, while malondialdehyde and relative electrical conductivity increased. Through screening the yeast two-hybrid library and combined with yeast two-hybrid and GST pull-down, the interaction of CsGPA1 with CsTIP1.1 was found the first time in a plant. Then, the expression of aquaporin (AQP) family genes was detected. The expression of CsAQP genes in leaves and roots was primarily up-regulated in WT under salt stress. However, interference by CsGPA1 resulted in enhanced expression of CsAQPs except for CsTIP3.2 in leaves, but reduced expression of some CsAQPs in roots under salt stress. Furthermore, principal component analysis of CsAQP expression profiles and linear regression analysis between CsGPA1 and CsAQPs revealed that CsGPA1 reversely regulated the expression of CsAQPs in leaves and roots under salt stress. Moreover, the water content in leaves and roots of RNAi seedlings significantly decreased compared with WT under salt stress. Overall, CsGPA1 interacts with CsTIP1.1 and suppression of CsGPA1 results in opposite patterns of expression of CsAQPs in leaves and roots, resulting in declining water content of cucumber under salt stress.










Similar content being viewed by others
Change history
18 September 2021
A Correction to this paper has been published: https://doi.org/10.1007/s00299-021-02782-6
References
Aebi H (1984) Catalase in vitro. Method Enzymol 105:121–126
Aharon R, Shahak Y, Wininger S, Bendov R, Kapulnik Y, Galili G (2003) Overexpression of a plasma membrane aquaporin in transgenic tobacco improves plant vigor under favorable growth conditions but not under drought or salt stress. Plant Cell 15:439–447
Alexandersson E, Danielson JÅ, Råde J, Moparthi VK, Fontes M, Kjellbom P, Johanson U (2010) Transcriptional regulation of aquaporins in accessions of Arabidopsis in response to drought stress. The Plant J 61:650–660
Alexieva V, Sergiev I, Mapelli S, Karanov E (2001) The effect of drought and ultraviolet radiation on growth and stress markers in pea and wheat. Plant Cell Environ 24:1337–1344
Bao G, Tang W, He F et al (2019) Physiological response in the leaf and stolon of white clover under acid precipitation and freeze–thaw stress. Funct Plant Biol 47(1):50–57
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Beyer JR, Wayne F, Fridovich I (1987) Assaying for superoxide dismutase activity: some large consequences of minor changes in conditions. Anal Biochem 161:559–566
Cao YY, Yang MT, Chen SY, Zhou ZQ, Li X, Wang XJ, Bai JG (2015) Exogenous sucrose influences antioxidant enzyme activities and reduces lipid peroxidation in water-stressed cucumber leaves. Biol Plant 59:147–153
Chaumont F, Tyerman SD (2014) Aquaporins: highly regulated channels controlling plant water relations. Plant Physiol 164:1600–1618
Chaumont F, Barrieu F, Wojcik E, Chrispeels MJ, Jung R (2001) Aquaporins constitute a large and highly divergent protein family in maize. Plant Physiol 125:1206–1215
Cheng Z, Li JF, Niu Y et al (2015) Pathogen-secreted proteases activate a novel plant immune pathway. Nature 521:213
Chu Y, Liu Y, Li J, Gong Q (2019) Effects of elevated pCO2 and nutrient enrichment on the growth photosynthesis and biochemical compositions of the brown alga Saccharina japonica (Laminariaceae Phaeophyta). PeerJ 7:e8040
Colaneri AC, Tunc-Ozdemir M, Huang JP, Jones AM (2014) Growth attenuation under saline stress is mediated by the heterotrimeric G protein complex. BMC Plant Biolo 14:129
Coursol S, Fan LM, Le Stunff H, Spiegel S, Gilroy S, Assmann SM (2003) Sphingolipid signalling in Arabidopsis guard cells involves heterotrimeric G proteins. Nature 423:651
Danielson JÅ, Johanson U (2008) Unexpected complexity of the aquaporin gene family in the moss Physcomitrella patens. BMC Plant Biolo 8:45
Ferrero-Serrano Á, Assmann SM (2016) The α-subunit of the rice heterotrimeric G protein RGA1, regulates drought tolerance during the vegetative phase in the dwarf rice mutant d1. J Exp Bot 67:3433–3443
Fetter K, Van Wilder V, Moshelion M, Chaumont F (2004) Interactions between plasma membrane aquaporins modulate their water channel activity. Plant Cell 16:215–228
Gao Y, Li T, Liu Y, Ren C, Zhao Y, Wang M (2010) Isolation and characterization of gene encoding G protein α subunit protein responsive to plant hormones and abiotic stresses in Brassica napus. Mol Biol Rep 37:3957–3965
Guo L, Devaiah SP, Narasimhan R, Pan X, Zhang Y, Zhang W, Wang X (2012) Cytosolic glyceraldehyde-3-phosphate dehydrogenases interact with phospholipase Dδ to transduce hydrogen peroxide signals in the arabidopsis response to stress. Plant Cell 24:2200–2212
Guo L, Wang ZY, Lin H et al (2006) Expression and functional analysis of the rice plasma-membrane intrinsic protein gene family. Cell Res 16:277
Hachez C, Veselov D, Ye Q, Reinhardt H, Knipfer T, Fricke W, Chaumont F (2012) Short-term control of maize cell and root water permeability through plasma membrane aquaporin isoforms. Plant Cell Environ 35:185–198
Hachez C, Moshelion M, Zelazny E, Cavez D, Chaumont F (2006) Localization and quantification of plasma membrane aquaporin expression in maize primary root: a clue to understanding their role as cellular plumbers. Plant Mol Bio 62:305–323
Hao HP, Li H, Jiang CD, Tang YD, Shi L (2018) Ion micro-distribution in varying aged leaves in salt-treated cucumber seedlings. Plant Physiol Bioch 129:71–76
Hu X, Qian Q, Xu T et al (2013) The U-box E3 ubiquitin ligase TUD1 functions with a heterotrimeric G α subunit to regulate brassinosteroid-mediated growth in rice. PLOS Genet 9:e1003391
Hu W, Yuan Q, Wang Y et al (2012) Overexpression of a wheat aquaporin gene TaAQP8, enhances salt stress tolerance in transgenic tobacco. Plant Cell Physiol 53:2127–2141
Huang J, Taylor JP, Chen JG et al (2006) The plastid protein THYLAKOID FORMATION1 and the plasma membrane G-protein GPA1 interact in a novel sugar-signaling mechanism in Arabidopsis. Plant Cell 18:1226–1238
Jangam AP, Pathak RR, Raghuram N (2016) Microarray analysis of rice d1 (RGA1) mutant reveals the potential role of G-protein alpha subunit in regulating multiple abiotic stresses such as drought salinity heat and cold. Front plant Sci 7:11
Jiang M, Zhang J (2001) Effect of abscisic acid on active oxygen species antioxidative defence system and oxidative damage in leaves of maize seedlings. Plant Cell Physiol 42:1265–1273
Khalil HB, Wang Z, Wright JA, Ralevski A, Donayo AO, Gulick PJ (2011) Heterotrimeric Gα subunit from wheat (Triticum aestivum), GA3, interacts with the calcium-binding protein Clo3, and the phosphoinositide-specific phospholipase C PI-PLC1. Plant Mol Biol 77:145
Lapik YR, Kaufman LS (2003) The Arabidopsis cupin domain protein AtPirin1 interacts with the G protein α-subunit GPA1 and regulates seed germination and early seedling development. Plant Cell 15:1578–1590
Li G, Santoni V, Maurel C (2014) Plant aquaporins: roles in plant physiology. Acta Gen Subj 1840:1574–1582
Li Y, Jia Y, Bian Y, Tong H, Qu J, Wang K, Wan XP (2019) Autocrine motility factor promotes endometrial cancer progression by targeting GPER-1. Cell Commun Signal 17:22
Ma Y, Dai X, Xu Y et al (2015) COLD1 confers chilling tolerance in rice. Cell 160:1209–1221
Matsumoto T, Lian HL, Su WA, Tanaka D, Liu CW, Iwasaki I, Kitagawa Y (2008) Role of the aquaporin PIP1 subfamily in the chilling tolerance of rice. Plant Cell Physiol 50:216–229
Maurel C, Verdoucq L, Luu DT, Santoni V (2008) Plant aquaporins: membrane channels with multiple integrated functions. Annu Rev Plant Biol 59:595–624
Mishra G, Zhang W, Deng F, Zhao J, Wang X (2006) A bifurcating pathway directs abscisic acid effects on stomatal closure and opening in Arabidopsis. Science 312:264–266
Misra S, Wu Y, Venkataraman G, Sopory SK, Tuteja N (2007) Heterotrimeric G-protein complex and G-protein-coupled receptor from a legume (Pisum sativum): role in salinity and heat stress and cross-talk with phospholipase C. The Plant J 51:656–669
Moshelion M, Hachez C, Ye Q, Cavez D, Bajji M, Jung R, Chaumont F (2009) Membrane water permeability and aquaporin expression increase during growth of maize suspension cultured cells. Plant cell environ 32:1334–1345
Murshed R, Lopez-Lauri F, Sallanon H (2008) Microplate quantification of enzymes of the plant ascorbate–glutathione cycle. Anal Biochem 383:320–322
Nguyen MX, Moon S, Jung KH (2013) Genome-wide expression analysis of rice aquaporin genes and development of a functional gene network mediated by aquaporin expression in roots. Planta 238:669–681
Oliviero T, Verkerk R, Van Boekel M, Dekker M (2014) Effect of water content and temperature on inactivation kinetics of myrosinase in broccoli (Brassica oleracea var italica). Food Chem 163:197–201
Postaire O, Tournaire-Roux C, Grondin A, Boursiac Y, Morillon R, Schäffner AR, Maurel C (2010) A PIP1 aquaporin contributes to hydrostatic pressure-induced water transport in both the root and rosette of Arabidopsis. Plant physiol 152:1418–1430
Roy Choudhury S, Pandey S (2017) Phosphatidic acid binding inhibits RGS 1 activity to affect specific signaling pathways in Arabidopsis. The Plant J 90:466–477
Rubinstein B (2000) Regulation of cell death in flower petals. In: Lam E, Fukuda H, Greenberg J (eds) Programmed cell death in higher plants. Springer, Dordrecht
Sade N, Vinocur BJ, Diber A et al (2009) Improving plant stress tolerance and yield production: is the tonoplast aquaporin SlTIP2; 2 a key to isohydric to anisohydric conversion. New Phytol 181:651–661
Secchi F, Zwieniecki MA (2013) The physiological response of Populus tremula x alba leaves to the down-regulation of PIP1 aquaporin gene expression under no water stress. Front Plant Sci 4:507
Sreedharan S, Shekhawat UK, Ganapathi TR (2013) Transgenic banana plants overexpressing a native plasma membrane aquaporin M usa PIP 1; 2 display high tolerance levels to different abiotic stresses. Plant Biotechnol J 11:942–952
Sui XL, Meng FZ, Wang HY et al (2012) Molecular cloning characteristics and low temperature response of raffinose synthase gene in Cucumis sativus L. J Plant Physiol 169:1883–1891
Swain DM, Sahoo RK, Srivastava VK, Tripathy BC, Tuteja R, Tuteja N (2017) Function of heterotrimeric G-protein γ subunit RGG1 in providing salinity stress tolerance in rice by elevating detoxification of ROS. Planta 245:367–383
Urano D, Jones AM (2014) Heterotrimeric G protein–coupled signaling in plants. Annu Rev Plant Biol 65:365–384
Wang X, Li Y, Ji W et al (2011) A novel Glycine soja tonoplast intrinsic protein gene responds to abiotic stress and depresses salt and dehydration tolerance in transgenic Arabidopsis thaliana. J Plant Physiol 168:1241–1248
Wang Y, Wang Y, Deng D (2019) Multifaceted plant G protein: interaction network agronomic potential and beyond. Planta 249:1259–1266
Wei W, Alexandersson E, Golldack D, Miller AJ, Kjellbom PO, Fricke W (2007) HvPIP1; 6, a barley (Hordeum vulgare L) plasma membrane water channel particularly expressed in growing compared with non-growing leaf tissues. Plant Cell Physiol 48:1132–1147
Yadav DK, Shukla D, Tuteja N (2013) Rice heterotrimeric G-protein alpha subunit (RGA1): in silico analysis of the gene and promoter and its upregulation under abiotic stress. Plant Physiol Biochem 63:262–271
Yan Y, Zhang W, Li Y, He C, Gao L, Yu X (2018) Functions of CsGPA1 on the hypocotyl elongation and root growth of cucumbers. Sci Rep 8:15583
Yu Q, Hu Y, Li J, Wu Q, Lin Z (2005) Sense and antisense expression of plasma membrane aquaporin BnPIP1 from Brassica napus in tobacco and its effects on plant drought resistance. Plant Sci 169:647–656
Zarrouk O, Garcia-Tejero I, Pinto C et al (2016) Aquaporins isoforms in cv Touriga Nacional grapevine under water stress and recovery-regulation of expression in leaves and roots. Agric Water Manage 164:167–175
Zhu Y, Guo J, Feng R, Jia J, Han W, Gong H (2016) The regulatory role of silicon on carbohydrate metabolism in Cucumis sativus L under salt stress. Plant Soil 406:231–249
Zhu YX, Yang L, Liu N et al (2019) Genome-wide identification structure characterization and expression pattern profiling of aquaporin gene family in cucumber. BMC plant biol 19:345
Acknowledgements
This work was supported by the earmarked fund for The National Key Research and Development Program of China (2018YFD1000800), National Nature Science Foundation of China (31601796), Central Public-interest Scientific Institution Basal Research Fund (No.IVF-BRF2020010) and Science and Technology Innovation Program of the Chinese Academy of Agricultural Sciences (CAAS-ASTIP-IVFCAAS) and the support by the Key Laboratory of Horticultural Crop Biology and Germplasm Innovation, Ministry of Agriculture, China. We thank Shelley Robison, PhD, from Liwen Bianji, Edanz Editing China (www.liwenbianji.cn/ac) for editing the English text of a draft of this manuscript.
Author information
Authors and Affiliations
Contributions
XCY, CXH and YY designed the experiments. YY and MTS performed the experiments. MTS and YY wrote the paper. YY and MTS analyzed the data. XCY, CXH, YY, MTS, YSL and JW revised the paper. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Communicated by Neal Stewart.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yan, Y., Sun, M., Li, Y. et al. The CsGPA1-CsAQPs module is essential for salt tolerance of cucumber seedlings. Plant Cell Rep 39, 1301–1316 (2020). https://doi.org/10.1007/s00299-020-02565-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-020-02565-5