Abstract
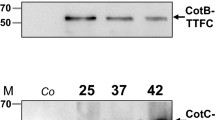
Human growth hormone (hGH) is the major and important hormone component of human being. At present, hGH for clinical uses is mostly produced in Escherichia coli, which requires costly denaturation and refolding to recover functionality. To obtain long-term bioactive hormone, we used hGH as a foreign gene and constructed a recombinant plasmid pJS700-hGH which carries a recombinant gene cotC-hgh with an enterokinase site under the control of cotC promoter. Plasmid pJS700-hGH was transformed into Bacillus subtilis by double crossover and an amylase-inactivated mutant was produced. After spore formation, Western blot and fluorescence immunoassay were used to monitor hGH surface expression on spores. Oral administration to silkworm with spores displaying hGH further showed that the recombinant spores may have potential ability to be digested and absorbed into the silkworm’s hemolymph due to both the resistant characters of spores and the addition of enterokinase site.





Similar content being viewed by others
References
Apte-Deshpande A, Rewanwar S, Kotwal P, Raiker VA, Padmanabhan S (2009) Efficient Expression and Secretion of Recombinant Human Growth Hormone in the Methylotrophic Yeast Pichia Pastoris: Potential Applications for other Proteins. Biotechnol Appl Biochem 54:197–205
Baumans V (2004) Use of Animals in Experimental Research: an Ethical Dilemma? Gene Ther 11(Suppl 1):64–66
Bernal A, Kimbrell DA (2000) Drosophila Thor Participates in Host Immune Defense and Connects a Translational Regulator with Innate Immunity. Proc Natl Acad Sci U S A 97:6019–6024
Bidlingmaier M, Strasburger CJ (2010) Growth Hormone. Handb Exp Pharmacol 195:187–200
Bradford MM (1976) A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-dye Binding. Anal Biochem 72:248–254
Ciabattini A, Parigi R, Isticato R, Oggioni MR, Pozzi G (2004) Oral Priming of Mice by Recombinant Spores of Bacillus subtilis. Vaccine 22:4139–4143
Demain AL, Vaishnav P (2009) Production of Recombinant Proteins by Microbes and Higher Organisms. Biotechnol Adv 27:297–306
Donovan W, Zheng LB, Sandman K, Losick R (1987) Genes Encoding Spore Coat Polypeptides from Bacillus subtilis. J Mol Biol 196:1–10
Driks A (1999) Bacillus subtilis Spore Coat. Microbiol Mol Biol Rev 63:1–20
Hamamoto H, Kurokawa K, Kaito C, Kamura K, Manitra Razanajatovo I, Kusuhara H, Santa T, Sekimizu K (2004) Quantitative Evaluation of the Therapeutic Effects of Antibiotics using Silkworms Infected with Human Pathogenic Microorganisms. Antimicrob Agents Chemother 48:774–779
Hamamoto H, Tonoike A, Narushima K, Horie R, Sekimizu K (2009) Silkworm as a Model Animal to Evaluate Drug Candidate Toxicity and Metabolism. Comp Biochem Physiol C 149:334–339
Henriques AO, Moran CP Jr (2000) Structure and Assembly of the Bacterial Endospore Coat. Methods 20:95–110
Huo J, Shi H, Yao Q, Chen H, Wang L, Chen K (2010) Cloning and Purification of Recombinant Silkworm Dihydrolipoamide Dehydrogenase Expressed in Escherichia coli. Protein Expr Purif 72:95–100
Isaksson OG, Eden S, Jansson JO (1985) Mode of Action of Pituitary Growth Hormone on Target Cells. Annu Rev Physiol 47:483–499
Isticato R, Esposito G, Zilhao R, Nolasco S, Cangiano G, De Felice M, Henriques AO, Ricca E (2004) Assembly of Multiple CotC Forms into the Bacillus subtilis Spore Coat. J Bacteriol 186:1129–1135
Johny S, Lange CE, Solter LF, Merisko A, Whitman DW (2007) New Insect System for Testing Antibiotics. J Parasitol 93:1505–1511
Kaito C, Akimitsu N, Watanabe H, Sekimizu K (2002) Silkworm Larvae as an Animal Model of Bacterial Infection Pathogenic to Humans. Microb Pathog 32:183–190
Kaito C, Kurokawa K, Matsumoto Y, Terao Y, Kawabata S, Hamada S, Sekimizu K (2005) Silkworm Pathogenic Bacteria Infection Model for Identification of Novel Virulence Genes. Mol Microbiol 56:934–944
Kim H, Hahn M, Grabowski P, McPherson DC, Otte MM, Wang R, Ferguson CC, Eichenberger P, Driks A (2006) The Bacillus subtilis Spore Coat Protein Interaction Network. Mol Microbiol 59:487–502
Kim J, Schumann W (2009) Display of Proteins on Bacillus subtilis Endospores. Cell Mol Life Sci 66:3127–3136
Kunst F, Ogasawara N, Moszer I, Albertini AM, Alloni G, Azevedo V, Bertero MG, Bessieres P, Bolotin A, Borchert S, Borriss R, Boursier L, Brans A, Braun M, Brignell SC, Bron S, Brouillet S, Bruschi CV, Caldwell B, Capuano V, Carter NM, Choi SK, Codani JJ, Connerton IF, Danchin A et al (1997) The Complete Genome Sequence of the Gram-positive Bacterium Bacillus subtilis. Nature 390:249–256
Lee SY, Choi JH, Xu Z (2003) Microbial Cell-surface Display. Trends Biotechnol 21:45–52
Li Q, Ning D, Wu C (2010) Surface Display of GFP using CotX as a Molecular Vector on Bacillus subtilis Spores. Sheng Wu Gong Cheng Xue Bao 26:264–269
Mahajan-Miklos S, Tan MW, Rahme LG, Ausubel FM (1999) Molecular Mechanisms of Bacterial Virulence Elucidated using a Pseudomonas aeruginosa-Caenorhabditis Elegans Pathogenesis Model. Cell 96:47–56
Mao L, Jiang S, Li G, He Y, Chen L, Yao Q, Chen K (2012) Surface Display of Human Serum Albumin on Bacillus subtilis Spores for Oral Administration. Curr Microbiol 64:545–551
Mauriello EM, le Duc H, Isticato R, Cangiano G, Hong HA, De Felice M, Ricca E, Cutting SM (2004) Display of Heterologous Antigens on the Bacillus subtilis Spore Coat using CotC as a Fusion Partner. Vaccine 22:1177–1187
Mesnage S, Tosi-Couture E, Mock M, Fouet A (1999) The S-layer Homology Domain as a means for Anchoring Heterologous Proteins on the Cell Surface of Bacillus Anthracis. J Appl Microbiol 87:256–260
Nicholson WL, Munakata N, Horneck G, Melosh HJ, Setlow P (2000) Resistance of Bacillus Endospores to Extreme Terrestrial and Extraterrestrial Environments. Microbiol Mol Biol Rev 64:548–572
Ozin AJ, Samford CS, Henriques AO, Moran CP Jr (2001) SpoVID guides SafA to the Spore Coat in Bacillus subtilis. J Bacteriol 183:3041–3049
Riddick L, Alter C, Davis DA, Frane J, Lippe B, Bakker B (2009) A Stepwise Increase in Recombinant Human Growth Hormone Dosing During Puberty Achieves Improved Pubertal Growth: A National Cooperative Growth Study Report. J Pediatr Endocrinol Metab 22:623–628
Russell AD (1990) Bacterial Spores and Chemical Sporicidal Agents. Clin Microbiol Rev 3:99–119
Shao X, Ni H, Lu T, Jiang M, Li H, Huang X, Li L (2012) An Improved System for the Surface Immobilisation of Proteins on Bacillus Thuringiensis Vegetative Cells and Spores through a New Spore Cortex-lytic Enzyme Anchor. N Biotechnol 29:302–310
Wang N, Chang C, Yao Q, Li G, Qin L, Chen L, Chen K (2011) Display of Bombyx Mori Alcohol Dehydrogenases on the Bacillus subtilis Spore Surface to Enhance Enzymatic Activity under Adverse Conditions. PLoS ONE 6:e21454
Zilhao R, Serrano M, Isticato R, Ricca E, Moran CP Jr, Henriques AO (2004) Interactions Among CotB, CotG, and CotH during Assembly of the Bacillus subtilis Spore Coat. J Bacteriol 186:1110–1119
Acknowledgments
We honestly thank Professor Degang Ning for kindly gifting plasmid pJS700, and Professor Hengchuan Xia for language revision of the text. This study was supported by the National Basic Research Program of China “973” under Grant No. 2012CB114604, the Natural Science Research General Project of Education Office of Anhui Province (No. KJ2012B108), and Postgraduate Research and Innovation Project of Jiangsu Province (Nos. CXZZ12_0703 and CXZZ13_0699). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lian, C., Zhou, Y., Feng, F. et al. Surface Display of Human Growth Hormone on Bacillus subtilis Spores for Oral Administration. Curr Microbiol 68, 463–471 (2014). https://doi.org/10.1007/s00284-013-0500-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-013-0500-9